��Ŀ����
(15��)
��֪G�Ǻϳ�ij�߷��ӻ������ԭ�ϣ�ֻ��C��H��O����Ԫ�أ�5.9g����G����1mol/LNaOH����Һ100mLǡ����ȫ��Ӧ��0.1mol����G�����������Ʒ�Ӧ�ڱ�״��������2.24L���������ӽṹ�к���һ������A�ķ���ʽΪC8H13O2Br���л���A��B��C��D��E��F��G֮����ת����ϵ��ͼ��ʾ��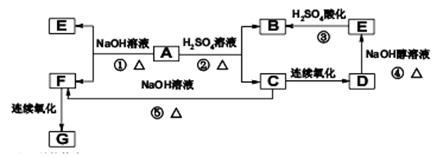
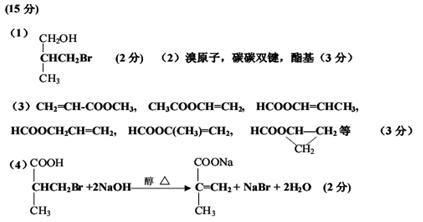
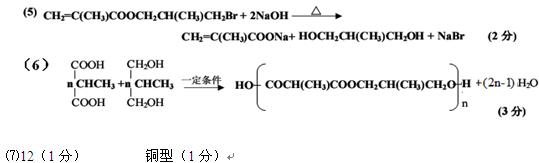
��д��C�Ľṹ��ʽ�� ��
��A�й����������ǣ� ��
��B��ͬ���칹�������������ж��֣���д����������������Ľṹ��ʽ��
�� �� ��
��д��D��E��ѧ��Ӧ����ʽ�� ��
��д����Ӧ����ѧ��Ӧ����ʽ��
��д����F��G ��һ�������ºϳɸ߷��ӻ�����Ļ�ѧ��Ӧ����ʽ��
��
����

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�(15��)������þ������ҩ��ҵ��������Ҫ����ɫ��ȼ����
������θ�����ҩ��Stmoache����Ч�ɷ�ΪMg(OH)2��
(1)��ҩ������θ��(��Ҫ�ɷ�Ϊ����)����֢ʱ��Ӧ������ʽ����ʽΪ ��
����֪:
H2O(g)=H2(g)+ O2(g) ��H1 = +242 kJ��mol-1
Mg(s)+2 H2O(g)=Mg(OH)2(s) + H2(g) ��H2 = -441 kJ��mol-1
Mg(s)+ O2(g)=MgO(s) ��H3= -602kJ��mol-1
(2)������þ�ֽ���Ȼ�ѧ����ʽΪ ��
��ij��������ˮ���Ȼ�þ�ʹ�ʯ����ȡ��������þ�������������������ʣ�ͨ���������̽����ᴿ���ƣ������ȼ��������þ��
(3)������м��뱣�շ������ã� ��
(4)��֪EDTAֻ������Һ�е���Ӧ����������ˮ�����ʣ�����
��Ӧ����Ȼ
������ˮ���������������EDTA�ļ��룬�����ܹ���
��ȥ����ô��ȸߵ�
����ӳ����ܽ�ƽ��ĽǶȼ��Խ��� ��
����Ϊ�о���ͬ�����ᴿ���������Ƶ���ȼ���Ĵ��ȴӶ�ȷ������ᴿ������ij�о�С���ȡ������������4���������Ƶõ���ȼ�����к������IJⶨ��������£�
| ������ȼ�������� | ��ȼ�������� | |||
| ��� | �ᴿ��ϵ�¶�/�� | ����EDTA����/g | ���뱣�շ�����/g | W(Fe)/(10-4g) |
| 1 | 40 | 0.05 | 0.05 | 7.63 |
| 2 | 40 | 0.05 | 0.10 | 6.83 |
| 3 | 60 | 0.05 | 0.10 | 6.83 |
| 4 | 60 | 0.10 | 0.10 | 6.51 |
(5)�����������������������ϱ����ݣ���ȡ�ߴ�����ȼ����������� (����ĸ)��
��40�� ��60�� ��EDTA����ΪO.05g
��EDTA����Ϊ0.10g �ݱ��շ�����Ϊ0.05g �ޱ��շ�����Ϊ0.10g
A���٢ۢ� B���ڢܢ� C���٢ܢ� D���ڢۢ�
(15��)������þ������ҩ��ҵ��������Ҫ����ɫ��ȼ����
������θ�����ҩ��Stmoache����Ч�ɷ�ΪMg(OH)2��
(1)��ҩ������θ��(��Ҫ�ɷ�Ϊ����)����֢ʱ��Ӧ������ʽ����ʽΪ ��
����֪:
H2O(g)=H2(g)+  O2(g)
��H1 = +242 kJ��mol-1
O2(g)
��H1 = +242 kJ��mol-1
Mg(s)+2 H2O(g)=Mg(OH)2(s) + H2(g) ��H2 = -441 kJ��mol-1
Mg(s)+  O2(g)=MgO(s)
��H3
= -602kJ��mol-1
O2(g)=MgO(s)
��H3
= -602kJ��mol-1
(2)������þ�ֽ���Ȼ�ѧ����ʽΪ ��
��ij��������ˮ���Ȼ�þ�ʹ�ʯ����ȡ��������þ�������������������ʣ�ͨ���������̽����ᴿ���ƣ������ȼ��������þ��

(3)������м��뱣�շ� �����ã�
��
�����ã�
��
(4)��֪EDTAֻ������Һ�е� ��Ӧ����������ˮ�����ʣ�����
��Ӧ����������ˮ�����ʣ����� ��Ӧ����Ȼ
��Ӧ����Ȼ ������ˮ���������������EDTA�ļ��룬�����ܹ���
������ˮ���������������EDTA�ļ��룬�����ܹ��� ��ȥ����ô��ȸߵ�
��ȥ����ô��ȸߵ� ����ӳ����ܽ�ƽ��ĽǶȼ��Խ���
��
����ӳ����ܽ�ƽ��ĽǶȼ��Խ���
��
����Ϊ�о���ͬ�����ᴿ���������Ƶ���ȼ���Ĵ��ȴӶ�ȷ������ᴿ������ij�о�С���ȡ������������4���������Ƶõ���ȼ�����к������IJⶨ��������£�
|
������ȼ�������� |
��ȼ�������� |
|||
|
��� |
�ᴿ��ϵ�¶�/�� |
����EDTA����/g |
���뱣�շ�����/g |
W(Fe)/(10-4g) |
|
1 |
40 |
0.05 |
0.05 |
7.63 |
|
2 |
40 |
0.05 |
0.10 |
6.83 |
|
3 |
60 |
0.05 |
0.10 |
6.83 |
|
4 |
60 |
0.10 |
0.10 |
6.51 |
(5)�����������������������ϱ����ݣ���ȡ�ߴ�����ȼ����������� (����ĸ)��
��40�� ��60�� ��EDTA����ΪO.05g
��EDTA����Ϊ0.10g �ݱ��շ�����Ϊ0.05g �ޱ��շ�����Ϊ0.10g
A���٢ۢ� B���ڢܢ� C���٢ܢ� D���ڢۢ�
 O2(g) ��H1 =" +242" kJ��mol-1
O2(g) ��H1 =" +242" kJ��mol-1 O2(g)="MgO(s) " ��H3 = -602kJ��mol-1
O2(g)="MgO(s) " ��H3 = -602kJ��mol-1
 �����ã� ��
�����ã� �� ��Ӧ����������ˮ�����ʣ�����
��Ӧ����������ˮ�����ʣ����� ��Ӧ����Ȼ
��Ӧ����Ȼ ������ˮ���������������EDTA�ļ��룬�����ܹ���
������ˮ���������������EDTA�ļ��룬�����ܹ��� ��ȥ����ô��ȸߵ�
��ȥ����ô��ȸߵ� ����ӳ����ܽ�ƽ��ĽǶȼ��Խ��� ��
����ӳ����ܽ�ƽ��ĽǶȼ��Խ��� ��