��Ŀ����
5�����������������������Ҫ��Ӫ�����ʣ�Ҳ����Ҫ�Ĺ�ҵԭ�ϣ�����ˮ������ղ���Ϊ�����ǣ��Իش��������⣺I��ʵ�����е�ҩƷ��������Ʒ���ķ�����������ô�����κ�����ҩƷ���ܷ�����Һ����������Һ����
�ɽ�һ������ͨ������Һ���ܹ۲쵽һ������ͨ·��Ϊ������Һ����֮Ϊ��������Һ ������ܣ�����д����ķ�������
���������ڲ�ͬ�����¿��Ա������ɲ�ͬ���ʣ���������ش����⣺
��֪�ڼ��ȣ���ѹ�д���������2RCOOH+2CH2=CH2+O2��2RCOOCH=CH2+2H2O
��1���������ھƻ�ø�����������л���A��A��B��C��D��E���ת����ϵ��ͼ��ʾ

��B��ʯ�ͻ�ѧ��ҵ����Ҫ�Ļ���ԭ�ϣ�д��A��B�Ļ�ѧ����ʽ��CH3CH2OH$��_{Ũ����}^{170��}$CH2=CH2��+H2O
��D�Ľṹ��ʽΪ��CH3COOCH=CH2
��2�������ǵĽṹ��ʽΪHOCH2��CHOH��4CHO��F�����弡��ϸ���е���������ȱ�������½������������IJ������Ϊ���ᣮ
���� I��������Һ���ڽ��壬���ж����ЧӦ��
���������ھƻ�ø�������������л���AΪCH3CH2OH��A������ȥ��Ӧ����BΪCH2=CH2��A��������������C����������Ϣ֪��CΪCH3COOH��DΪCH3COOCH=CH2��D�����Ӿ۷�Ӧ����EΪ ��
��
��� �⣺I�����û�ѧ�Լ������õ�ˮ������ۣ�Ҳ����������Һ���������ǣ��粻�û�ѧ�Լ����ɿ��ǵ������뾶�ϴ�����Һ���н�������ʣ��ɽ�һ������ͨ������Һ���ܹ۲쵽һ������ͨ·��Ϊ������Һ����֮Ϊ��������Һ��
�ʴ�Ϊ���ɽ�һ������ͨ������Һ���ܹ۲쵽һ������ͨ·��Ϊ������Һ����֮Ϊ��������Һ��
���������ھƻ�ø�������������л���AΪCH3CH2OH��A������ȥ��Ӧ����BΪCH2=CH2��A��������������C����������Ϣ֪��CΪCH3COOH��DΪCH3COOCH=CH2��D�����Ӿ۷�Ӧ����EΪ ��
��
���Ҵ�������ȥ��Ӧ������ϩ����Ӧ����ʽΪ��CH3CH2OH$��_{Ũ����}^{170��}$CH2=CH2��+H2O��
�ʴ�Ϊ��CH3CH2OH$��_{Ũ����}^{170��}$CH2=CH2��+H2O��
��ͨ�����Ϸ���֪��DΪCH3COOCH=CH2���ʴ�Ϊ��CH3COOCH=CH2��
��2�������ǵĽṹ��ʽΪ��HOCH2��CHOH��4CHO��F�����弡��ϸ���е���������ȱ�������½������������IJ����F��CH3CH��OH��COOH������Ϊ���ᣬ
�ʴ�Ϊ��HOCH2��CHOH��4CHO�����ᣮ
���� ���⿼���л����ƶϼ��л�����𣬸��ݷ�Ӧ���������ƶϣ�ע��������Ϣ�����ã���Ŀ�ѶȲ���

 ��ְٷְټ�����Ԫ��ĩ���Ծ�ϵ�д�
��ְٷְټ�����Ԫ��ĩ���Ծ�ϵ�д� Сѧ��ĩ���Ծ�ϵ�д�
Сѧ��ĩ���Ծ�ϵ�д�| A�� | ��״���£�22.4 L CCl4����4nA��Clԭ�� | |
| B�� | 7.8 g Na2O2������ˮ��Ӧת��0.2nA������ | |
| C�� | 0.1 mol•L-1NaCl��Һ�к���0.1nA��Na+ | |
| D�� | ���³�ѹ�£�23 gNO2��N2O4��������к���nA����ԭ�� |
| A�� | �ȷ� | B�� | 1-�ȱ��� | C�� | 2-���� | D�� | 2-��-2-����� |
| A�� | 16.51 g | B�� | 23.82 g | C�� | 24.84 g | D�� | 17.02 g |
| A�� | H2O | B�� | HCl | C�� | NaOH | D�� | O2 |
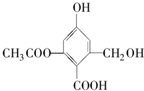
| A�� | X�Ļ�ѧʽΪC10H10O6 | |
| B�� | X��һ������������FeCl3��Һ������ɫ��Ӧ | |
| C�� | 1mol X�ֱ���������Na��NaOH��Һ��NaHCO3��Һ��Ӧ���������������ʵ����ʵ����ֱ�Ϊ3mol��5mol��1mol | |
| D�� | X�ȿ��Ժ��Ҵ��ֿ��Ժ����ᷢ��������Ӧ |
| A�� | ����һ����C��ƽ�������ƶ� | |
| B�� | �����¶Ȳ��䣬�����ݻ�ѹ����ԭ����һ�룬����B��Ũ�ȱ�Ϊԭ����1.8������ƽ�������ƶ� | |
| C�� | ���º�ѹ���ٳ���1mol�����������뷴Ӧ����A��ת����������m��n | |
| D�� | ���º�ѹ���ټ���2mol��A�������ƽ����Է���������Ϊԭ����2�� |
2Fe��OH��3+3Cl2+10KOH $\frac{\underline{\;0��30��\;}}{\;}$2K2FeO4+6KCl+8H2O
���ڸ÷�Ӧ������˵����ȷ���ǣ�������
| A�� | ˮ�ǻ�ԭ���� | B�� | Fe��OH��3������ԭ��Ӧ | ||
| C�� | Cl2�������� | D�� | ÿ����1mol K2FeO4ת��2mol���� |