��Ŀ����
12��ͨ����������ʵ��ⶨ���ᾧ�壨H2C2O4•xH2O���нᾧˮx��ֵ����1����ȡmg���ᾧ�壬���Ƴ�100.0mL��Һ��
��2��ȡ25.0mL������Һ������ƿ�У���������ϡ�������Ũ��Ϊa mol•L-1 KMnO4��Һ���еζ���
�䷴Ӧԭ���ǣ�2KMnO4+5H2C2O4+3H2SO4=K2SO4+2MnSO4+10CO2��+8H2O
��ش��������⣺
��ʵ�飨2���У�KMnO4Ӧװ����ʽ�ζ����У����ʽ��������ʽ�������ζ�ǰ�Ƿ���Ҫ����ƿ�ڼ���ָʾ������ǡ���������ԭ�����Ϻ�ɫKMnO4��Һ��H2C2O4��ԭΪMn2+ʱ��Һ��ɫ�������һ��KMnO4����ʱ����Һ���Ϻ�ɫ������ȥ��Ϊ�ζ��ն���
�����ζ��õ�KMnO4��Һ�����ʱ����������ֱ��ʣ�Ũ��ƫ�ͣ������ʵ�������xֵƫ�ͣ��ƫ�ߡ�����ƫ�͡�������Ӱ�족����
�����ζ��յ�ʱ������ȥKMnO4��ҺVmL���������ҺŨ��Ϊ$\frac{aV}{10}$mol•L-1��x=$\frac{50m-45aV}{9aV}$���ú���m��V�Ĵ���ʽ��ʾ��
���� ��KMnO4��Һ����ǿ�����ԣ����Ը�ʴ��Ƥ�ܣ���KMnO4��ҺӦװ����ʽ�ζ����У�
KMnO4��Һ����ɫ�����ᷴӦ��ϣ��������һ��KMnO4��Һ����ɫ����ȥ��˵���ζ����յ㣬����Ҫ���ָʾ����
���������ѧ����ʽ�����ݿɵó�X�����ζ��õ�KMnO4��Һ�����ʱ����������ֱ��ʣ�Ũ��ƫ�ͣ���������������KMnO4��Һ�����ƫ���ɴ�����n��H2C2O4��ƫ����n��H2O��ƫС��xƫ�ͣ�
�۸��ݻ�ѧ����ʽ��������֮��Ĺ�ϵ���м��㣮
��� �⣺��KMnO4��Һ����ǿ�����ԣ����Ը�ʴ��Ƥ�ܣ���KMnO4��ҺӦװ����ʽ�ζ����У�
KMnO4��Һ����ɫ�����ᷴӦ��ϣ��������һ��KMnO4��Һ����ɫ����ȥ��˵���ζ����յ㣬����Ҫ���ָʾ����
�ʴ�Ϊ������Ϻ�ɫKMnO4��Һ��H2C2O4��ԭΪMn2+ʱ��Һ��ɫ�������һ��KMnO4����ʱ����Һ���Ϻ�ɫ������ȥ��Ϊ�ζ��ն���
�����ζ��õ�KMnO4��Һ�����ʱ����������ֱ��ʣ�Ũ��ƫ�ͣ��ɴ�����n��H2C2O4��ƫ����n��H2O��ƫС��xƫ�ͣ�
�ʴ�Ϊ��ƫ�ͣ�
����2MnO2-+5H2C2O4 +6H+�T2Mn2++10CO2��+8H2O
2mol 5mol
aV��10-3mol 0.025��Cmol
$\frac{2mol}{aV��1{0}^{-3}mol}$=$\frac{5mol}{0.025L��cmol/L}$
��ã�C=$\frac{aV}{10}$mol•L-1
H2C2O4•xH2O��H2C2O4 ��xH2O
1mol 18xg
0.1aV��0.1 w-0.1aV��0.1��90
$\frac{1mol}{0.1aV��0.1}$=$\frac{18xg}{w-0.1aV��0.1��90}$
���x=$\frac{50m-45aV}{9aV}$��
�ʴ�Ϊ��$\frac{aV}{10}$��$\frac{50m-45aV}{9aV}$��
���� ���⿼���˵ζ��������ζ�Ӧ������㡢������ɺ����ⶨ�ȣ��Ѷ���Ŀ�еȣ�ע��ζ��о������ݹ�ϵʽ���м��㣬���ո��ݹ�ϵʽ���㷽����

| Ԫ�� | �����Ϣ |
| X | X���������Ϊ��Ӧ��ˮ���ﻯѧʽΪH2XO3 |
| Y | Y�ǵؿ��к�����ߵ�Ԫ�� |
| Z | Z�Ļ�̬ԭ�����������Ų�ʽΪ3s23p1 |
| W | W��һ�ֺ��ص�������Ϊ28��������Ϊ14 |
��2��Z�ĵ�һ�����ܱ�W��С�����С������XY2�ɹ�̬��Ϊ��̬����˷��������������Ƿ��Ӽ�����������Ԫ�ء�X��Y��ԭ�ӿɹ�ͬ�γɶ��ַ��ӣ�д������һ�����γ�ͬ�ַ��Ӽ���������������Ҵ��������
��3�����£���Z���������ᷴӦ�����ɫ��Һ�еμ�NaOH��Һֱ���������ܹ۲쵽�������������ɰ�ɫ��������������ܽ⣬�������ɫ��Һ��W�ĵ���������ᷴӦ����������ɫ���壬�÷�Ӧ�Ļ�ѧ����ʽ��Si+4HF=SiF4��+2H2��
��4����25�㡢101kpa�£���֪13.5g��Z���嵥����Y2��������ȫȼ�պ�ָ���ԭ״̬������419kJ���÷�Ӧ���Ȼ�ѧ����ʽ��2Al��s��+3O2��g��=2Al2O3��s����H=-3352kJ/mol��
| A�� | ���ʱ���������Һ��K+�������ƶ� | |
| B�� | �ŵ�ʱ���������Һ��K+�������ƶ� | |
| C�� | ���ʱ��������ӦΪ��Zn��OH��${\;}_{4}^{2-}$+2e-�TZn+4OH- | |
| D�� | �ŵ�ʱ����·��ͨ��2mol���ӣ���������22.4L����״���� |
| A�� | AlCl3��Һ�м��������ˮ��Al3++4OH-=AlO2-+2H2O | |
| B�� | Ư����Һ��ͨ��������SO2���壺Ca2++2ClO-+SO2+H2O=CaSO3��+2HClO | |
| C�� | ��ϡ�����ȥ�Թ��ڱ�����3Ag+4H++NO3-=3Ag++NO��+2H2O | |
| D�� | ����ʯ��ˮ������С�մ���Һ��ϣ�Ca2++2OH-+2HCO3-=CaCO3��+CO32-+2H2O |
����һ��
���϶���
| ���� | �۵�/�� | �е�/�� | �ܶ�/g•cm-3 | �ܽ��� |
| �Ҷ����� C2H6O2�� | -11.5 | 198 | 1.11 | ������ˮ���Ҵ� |
| ��������C3H8O3�� | 17.9 | 290 | 1.26 | �ܸ�ˮ���ƾ�������Ȼ��� |
A������ B����ȡ�� C�����ܽ⡢�ᾧ�����ˡ��ķ��� D����Һ��
��1����������Ȼ��ƺʹ���Ļ�����з�����������Ӧ��C��
��2�����Ҷ����ͱ�������������ѷ�����A��
��3�������л��ܼ�CCl4ʱ��Ҳ���ȡ����ķ���������ʵ��װ��ͼ�еĴ���֮���У�
���¶ȼ��¶˵�ˮ�����ܲ�����Һ��
�������ܽ���ˮ�ķ���ߵ�
�ۼ����ձ�δ����ʯ����
��ͼ������������ʱ��ʹ��ˮ�ܼ����ȵĺô������Ⱦ��ȣ����ڿ����¶ȣ�


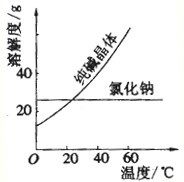
��֪ZnSO4���ܽ�������ʾ��
| �¶�/�� | 0 | 10 | 20 | 30 | 40 | 60 | 80 | 100 |
| �ܽ��/g | 41.6 | 47.5 | 53.8 | 61.3 | 70.5 | 75.4 | 71.1 | 60.5 |
��2�����������Ҫ�ɷ�ΪPbSO4��X�Ļ�ѧʽΪZnO��Zn��OH��2��
��3���ܽ�����м����ϡ������������Ʊ�ZnSO4������ZnSO4ˮ�⣮
��4���ᾧ�IJ���Ϊ����Ũ������ȴ�ᾧ��
��5���þƾ�����ϴ�ӵ�ԭ���Ǽ�������п�������ʧ�������
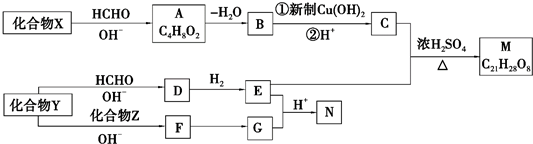
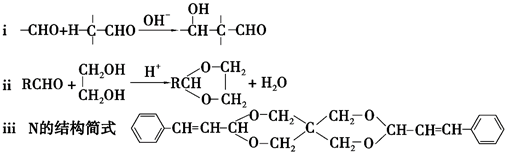
 +2Cu��OH��2+NaOH$\stackrel{��}{��}$
+2Cu��OH��2+NaOH$\stackrel{��}{��}$ +Cu2O��+3H2O��
+Cu2O��+3H2O�� ��
��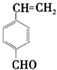 ��
�� ��
�� ��A��B��W��D��EΪ������Ԫ�أ���ԭ��������������������֮��Ϊ39��B��Wͬ���ڣ�A��Dͬ���壬A��W���γ�����Һ̬������A2W��A2W2��EԪ�ص���������������������ȣ�
��A��B��W��D��EΪ������Ԫ�أ���ԭ��������������������֮��Ϊ39��B��Wͬ���ڣ�A��Dͬ���壬A��W���γ�����Һ̬������A2W��A2W2��EԪ�ص���������������������ȣ�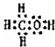 ��
��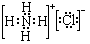 ��
��