��Ŀ����
14���ֽ�һ����N2O4��NO2�Ļ������ͨ�����Ϊ2L�ĺ��º����ܱղ��������У�������Ӧ��2NO2��g��?N2O4��g������H��0��������Ũ����ʱ��仯��ϵ��ͼ��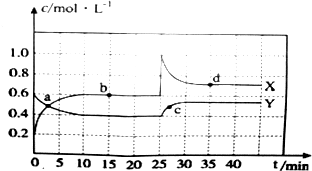
��1��ͼ������Y���X����Y������ʾc��N2O4����ʱ��ı仯��
��2��ͼ��b���ƽ�ⳣ��K��ֵΪ0.9
��3�������������䣬����ʼʱ���������ͨ��0.4mol N2O4��0.2mol NO2����v����v�������������=���������������¸÷�Ӧ��ƽ�ⳣ��K�������������С�����䡱�����ж�������N2O4�ֽ�ΪNO2�����ȷ�Ӧ���¶����ߣ�ƽ�����ƣ�ƽ�ⳣ������
��4��25minʱ���ı���������������NO2��Ũ�ȣ�ͼ����a��b��c��d�ĵ��е�c����ϵ��ɫ���
���� ��1�����ݷ�Ӧ����ʽ2NO2��g��?N2O4��g����֪��NO2��Ũ�ȱ仯��N2O4Ũ�ȱ仯����2�����ݴ˽��ͼ��������Ũ�ȵı仯���жϣ�
��2��NO2��Ũ�ȱ仯��N2O4Ũ�ȱ仯����2����0��15min����������Ũ����������������Ũ�ȼ�С��ƽ��������У�����ͼ�����ƽ��ʱ��Ũ�ȣ��ٸ���K�ı���ʽ���K��
��3�������ʱ��Ũ����Q��ƽ�ⳣ���Ƚ��жϷ�Ӧ���еķ���ӦΪN2O4��g��?2NO2��g������H��0������ƽ��������У�ƽ�ⳣ������
��4��25minʱ��������Ũ�ȼ�������ʱ����������Ũ�Ȳ��䣬�淴Ӧ���ж�������Ũ�Ƚ��У�����������Ũ������ƽ��������У�ͼ���֪��������Ũ�����ĵ�������ɫ���
��� �⣺��1����ͼ��֪10-25minƽ��״̬ʱ��X��ʾ���������Ũ�ȱ仯��Ϊ��0.6-0.2��mol/L=0.4mol/L��Y��ʾ�ķ�Ӧ���Ũ�ȱ仯��Ϊ��0.6-0.4��mol/L=0.2mol/L��X��ʾ���������Ũ�ȱ仯����Y��ʾ�ķ�Ӧ���Ũ�ȱ仯����2��������X��ʾNO2Ũ����ʱ��ı仯���ߣ�Y��ʾN2O4Ũ����ʱ��ı仯���ߣ�
�ʴ�Ϊ��Y��
��2��NO2��Ũ�ȱ仯��N2O4Ũ�ȱ仯����2����b��0��15min����ӦN2O4��g��?2NO2��g����ƽ��Ũ��Ϊc��NO2��=0.6mol•L-1��c��N2O4��=0.4mol•L-1����K=$\frac{{c}^{2}��N{O}_{2}��}{c��{N}_{2}{O}_{4}��}$=$\frac{0��{6}^{2}}{0.4}$=0.9���ʴ�Ϊ��0.9��
��3�������������䣬����ʼʱ���������ͨ��0.4mol N2O4��0.2mol NO2��NO2Ũ��Ϊ0.1mol/L��N2O4Ũ��Ϊ0.2mol/L��Qc=$\frac{0��{1}^{2}}{0.2}$=0.05��K=0.9����ƽ��������У���v����v������Ӧ���еķ��������ȷ�Ӧ����������ƽ��������У��÷�Ӧ��ƽ�ⳣ��K����
�ʴ�Ϊ����������N2O4�ֽ�ΪNO2�����ȷ�Ӧ���¶����ߣ�ƽ�����ƣ�ƽ�ⳣ������
��4��25minʱ��������Ũ�ȼ�������ʱ����������Ũ�Ȳ��䣬�淴Ӧ���ж�������Ũ�Ƚ��У�����������Ũ������ƽ��������У����Ըı���������������������Ũ�ȣ�ͼ���֪��������Ũ�����ĵ�������ɫ���c��Ķ�������Ũ�������ɫ���
�ʴ�Ϊ������NO2��Ũ�ȣ�c��
���� ���⿼���˻�ѧƽ��״̬����ѧ��Ӧ���ʵļ����Ӱ�����ط����жϣ���Ҫ�Ƿ�Ӧ���з����������ɫ�仯���жϣ���Ŀ�Ѷ��еȣ�

 ���㼤�������100�ִ��Ծ�ϵ�д�
���㼤�������100�ִ��Ծ�ϵ�д�| ���� | �����Լ� | ���뷽�� | |
| A | ����������Һ������ͭ�� | ���� | �ᾧ |
| B | BaSO4��BaCO3�� | ���� | ���� |
| C | NaCl��Һ��I2�� | �Ҵ� | ��Һ |
| D | ���ۣ�ͭ�ۣ� | ���� | ���� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | 0.03mol/L | B�� | 0.04mol/L | C�� | 0.05mol/L | D�� | 0.06mol/L |
| A�� | ��NaHA��Һ�����ԣ���H2Aһ����ǿ�� | |
| B�� | ��NaHA��Һ�ʼ��ԣ���H2Aһ�������� | |
| C�� | NaHA��Һ��Na+��HA-�ĸ���֮��һ������1��1 | |
| D�� | NaHA�����е��������Ӹ�����һ����1��1 |
| A�� | Na2O��Na2O2 | B�� | Na2O2��Na2CO3 | C�� | Na2CO3 | D�� | Na2O |
 �ҹ��нϳ��ĺ����ߣ�����ĺ�����һ�����������Դ�������ı��⣮Ŀǰ��������������о���γ�����ú�����Դ��
�ҹ��нϳ��ĺ����ߣ�����ĺ�����һ�����������Դ�������ı��⣮Ŀǰ��������������о���γ�����ú�����Դ��