��Ŀ����
17��������Դ�����þ��й���ǰ������1�����辭����ѧ�仯���ܴӺ�ˮ�л�õ�������BD������ţ�
A�� Cl2 B�� ��ˮ C�� �ռ� D�� ʳ��
��2���Ӻ�ˮ����ȡ�����Ҫ��������Ũ���ĺ�ˮ��ͨ�����������������������÷�Ӧ�����ӷ���ʽ��Cl2+2Br-=Br2+2Cl-��
��3����ͼ�ǴӺ�ˮ����ȡþ�ļ����̣�
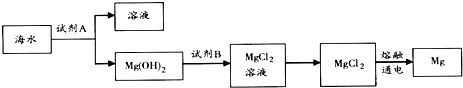
�ٹ�ҵ�ϳ����ڳ���Mg2+���Լ�A����ʯ�ң�תMg��OH��2��ΪMgCl2�����ӷ���ʽ��Mg��OH��2+2H+=Mg2++2H2O��
������ˮMgCl2��ȡMg�Ļ�ѧ����ʽ��MgCl2$\frac{\underline{\;ͨ��\;}}{\;}$Mg+Cl2����
��4���������и�����I-��ʽ���ڵĵ�Ԫ�أ�ʵ������ȡI2��;��������ʾ��
�ɺ���$\stackrel{����}{��}$������$��_{����}^{ˮ}$��Һ$��_{����������Һ}^{�ữ}$$\stackrel{CCl_{4}}{��}$$\stackrel{�ಽ����}{��}$I2
�����պ������ҽ�ʱ���õ���Ҫ����������������
�����ữ����Һ�мӹ���������Һ��д���÷�Ӧ�����ӷ���ʽ2H++2I-+H2O2�TI2+2H2O����Ӧ�������ټ���CCl4����ȡ���������ã����Թ۲쵽CCl4�����ɫ��
���� ��1���Ӻ�ˮ�Ʊ����ʵ�ԭ����֪������������ǽ���������Ҫ���û�ѧ��Ӧ����ȡ����ʳ�ο���������ԭ������ˮ��������ԭ�����õ���
��2����������������������������Ϊ�嵥�ʣ�
��3����ˮ�м�����ʯ�ҳ���þ���ӹ��˵õ�������þ�����������Լ�BΪ���ᣬ������þ�ܽ�Ϊ�Ȼ�þ��Һ��Ũ�������ᾧ�õ��Ȼ�þ���壬�缫�����Ȼ�þ�õ�����þ��������
�ٹ�ҵ�ϳ����ڳ���Mg2+���Լ�A���������ƣ�Mg��OH��2������������MgCl2����Һ��
�ڵ�������Ȼ�þ�õ�����þ��������
��4�������չ���ʱ���õ���Ҫ����������������
�������Ӻ������⽫����������Ϊ���ʵ⣻
�۵��CCl4��Һ����ɫ��
��� �⣺��1��A����ˮ����Ԫ��Ϊ�����ӣ��ǵ�ⱥ��ʳ��ˮ���������Ȼ��Ƶõ������������˻�ѧ�仯����A����
B���Ѻ�ˮ������ȷ������Եõ���ˮ������Ҫ��ѧ�仯����B��ȷ��
C����ⱥ���Ȼ�����Һ�õ��ռ���������������������ڻ�ѧ�仯����C����
D���Ѻ�ˮ��̫����ɹ������ˮ�ֺ�ʳ�Σ�����Ҫ��ѧ�仯���ܹ��Ӻ�ˮ�л�ã���D��ȷ��
�ʴ�Ϊ��BD��
��2���Ӻ�ˮ����ȡ�����Ҫ��������Ũ���ĺ�ˮ��ͨ�����������������������÷�Ӧ�����ӷ���ʽ�ǣ�Cl2+2Br-=Br2+2Cl-��
�ʴ�Ϊ��Cl2+2Br-=Br2+2Cl-��
��3����ˮ�м�����ʯ�ҳ���þ���ӹ��˵õ�������þ�����������Լ�BΪ���ᣬ������þ�ܽ�Ϊ�Ȼ�þ��Һ��Ũ�������ᾧ�õ��Ȼ�þ���壬�缫�����Ȼ�þ�õ�����þ��������
�ٹ�ҵ�ϳ����ڳ���Mg2+���Լ�A���������ƣ�Mg��OH��2������������MgCl2����Һ�����ӷ���ʽΪ��Mg��OH��2+2H+=Mg2++2H2O��
�ʴ�Ϊ����ʯ�ң�Mg��OH��2+2H+=Mg2++2H2O��
�ڵ�������Ȼ�þ�õ�����þ����������Ӧ�Ļ�ѧ����ʽΪ��MgCl2$\frac{\underline{\;ͨ��\;}}{\;}$Mg+Cl2����
�ʴ�Ϊ��MgCl2$\frac{\underline{\;ͨ��\;}}{\;}$Mg+Cl2����
��4�������չ���ʱ���õ���Ҫ�����������������ʴ�Ϊ��������
�ڼ��������Ӻ��������������Ϊ����������������ת��Ϊ���ʵ⣬���ӷ���ʽΪ2H++2I-+H2O2�TI2+2H2O���ʴ�Ϊ��2H++2I-+H2O2�TI2+2H2O��
�۵��CCl4��Һ����ɫ���ʴ�Ϊ���ϣ�
���� ���⿼���˺�ˮ��Դ���ۺ����ã����ݻ�ѧ��Ӧ�ĸ������ͼ�����ʵ��������������֪������ͼ��ÿһ���ķ�Ӧ������д��Ӧ�Ļ�ѧ����ʽ��֪����������ij�����������Ŀ�ѶȲ���

 ��ʦ����ɳ���ʱͬ��ѧ����ϵ�д�
��ʦ����ɳ���ʱͬ��ѧ����ϵ�д�| A�� | O3��O2��Ϊͬλ�� | |
| B�� | O3��O2���ת���������仯 | |
| C�� | ��ͬ״���£������O3��O2������ͬ������ | |
| D�� | O3��O2�����Ը�ǿ |
CrO42-$\underset{\stackrel{{H}^{+}}{��}}{��ת��}$Cr2O72-$\underset{\stackrel{F{e}^{2+}}{��}}{�ڻ�ԭ}$Cr3+
����˵��ת��һ����Ӧ��ƽ��״̬���ǣ�������
| A�� | Cr2O72-��CrO42-��Ũ����ͬ | B�� | 2v����Cr2O72-��=v����CrO42-�� | ||
| C�� | ��Һ����ɫ���� | D�� | ��Һ��pH���� |
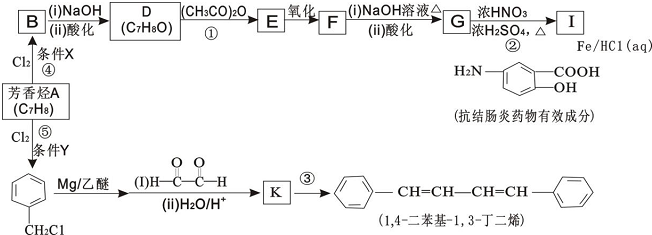
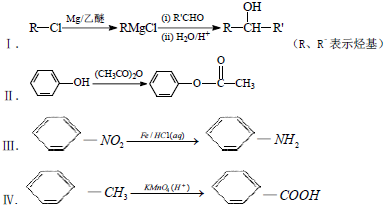
 ��
��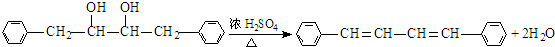 ��
�� ��
��


 ��
��