��Ŀ����
���ᣨH2C2O4����Ҫ�������������غͱ�Ƭ��ҩ���Լ�����ϡ�н������ܼ���Ⱦ�ϻ�ԭ����������ȣ���֪25��ʱ������Ka1=5.9��10-2��Ka2=6.4��10-5��̼�ᣨH2CO3���ĵ���ƽ�ⳣ��ΪKa1=4.2��10-7��Ka2=5.6��10-11��
NH3?H2O��Kb=1.8��10-5��
��1����ʢ��3ml 0.1mol?L-1Na2CO3��Һ���Թ��У���μ���0.1mol?L-1H2C2O4��Һ������������д����ʼ�η�����Ӧ�����ӷ���ʽ�� ��0.1mol?L-1��H2C2O4��Һ��ˮϡ�����У����б���ʽ��ֵ������ ��
A.
B.
C.
D.
��2��25��ʱ����10mL0.2mol?L-1H2C2O4��Һ�м���10mL 0.2mol?L-1NaOH��Һʱ��������ҺpH��7����ԭ���ǣ� ������˵����ȷ����
A.10mL0.1mol?l-1H2C2O4��Һ�м���VmL0.1mol?L-1NH3?H2O��Һ��������ҺpH=7����V��20mL
B�������ʵ���Ũ�ȵ�NaHC2O4��Na2C2O4��Na2CO3��Һ��pH��С˳��Ϊ��Na2CO3��Na2C2O4��NaHC2O4
C��NaHC2O4��Һ�и�����Ũ�ȴ�С˳��c(Na+)��c(HC2O4)��c(H+)��c(OH-)��c(C2O42-)
D�������ʵ���Ũ�ȵ�Na2C2O4��NaHCO3���Һ�У�c(CO32-)+c(HCO3-)=c(C2O42-)+c(HC2O4-)
��3������Ũ�Ⱦ�Ϊ0.1mol?L-1��Na2CO3��NaHCO3�����Һ�м������������������ָ���Һ��pH�仯������ԭ���� ���ݴ˿��ж�Ũ�Ⱦ�Ϊ0.1mol?L-1�����л����Һ�У����ֿܵ���������������ҺpHӰ����� ��
A��H2C2O4��NaHC2O4 B��NH4Cl��NaCl C��NaHSO3��Na2SO3 D��Na2S2O3��KNO3
��4����֪CaCO3��CaC2O4���ܶȻ�������25�棩�ֱ�Ϊ5.0��10-9��2.5��10-9����0.3mol?L-1Na2C2O4��Һ�м������CaCO3�������Һ�е�c(CO32-)= ��
��5�����ʵ�鷽��֤����ˮ�д����ŵ���ƽ�� ��
NH3?H2O��Kb=1.8��10-5��
��1����ʢ��3ml 0.1mol?L-1Na2CO3��Һ���Թ��У���μ���0.1mol?L-1H2C2O4��Һ������������д����ʼ�η�����Ӧ�����ӷ���ʽ��
A.
| c(H+) |
| c(OH-) |
| c(H+) |
| c(H2C2O4) |
c(C2
| ||
c(HC2
|
c2(H+)?c(C2
| ||
| c(H2C2O4) |
��2��25��ʱ����10mL0.2mol?L-1H2C2O4��Һ�м���10mL 0.2mol?L-1NaOH��Һʱ��������ҺpH��7����ԭ���ǣ�
A.10mL0.1mol?l-1H2C2O4��Һ�м���VmL0.1mol?L-1NH3?H2O��Һ��������ҺpH=7����V��20mL
B�������ʵ���Ũ�ȵ�NaHC2O4��Na2C2O4��Na2CO3��Һ��pH��С˳��Ϊ��Na2CO3��Na2C2O4��NaHC2O4
C��NaHC2O4��Һ�и�����Ũ�ȴ�С˳��c(Na+)��c(HC2O4)��c(H+)��c(OH-)��c(C2O42-)
D�������ʵ���Ũ�ȵ�Na2C2O4��NaHCO3���Һ�У�c(CO32-)+c(HCO3-)=c(C2O42-)+c(HC2O4-)
��3������Ũ�Ⱦ�Ϊ0.1mol?L-1��Na2CO3��NaHCO3�����Һ�м������������������ָ���Һ��pH�仯������ԭ����
A��H2C2O4��NaHC2O4 B��NH4Cl��NaCl C��NaHSO3��Na2SO3 D��Na2S2O3��KNO3
��4����֪CaCO3��CaC2O4���ܶȻ�������25�棩�ֱ�Ϊ5.0��10-9��2.5��10-9����0.3mol?L-1Na2C2O4��Һ�м������CaCO3�������Һ�е�c(CO32-)=
��5�����ʵ�鷽��֤����ˮ�д����ŵ���ƽ��
���㣺�����ʱ�Ķ����жϼ��й�ph�ļ���,Ӱ������ˮ��̶ȵ���Ҫ����,���ܵ���ʵ��ܽ�ƽ�⼰����ת���ı���
ר�⣺����ƽ������Һ��pHר��,�����ˮ��ר��
��������1�������̼���Ʒ�Ӧ���ɲ����ƺ�̼�����ƣ���ˮϡ�Ͳ���ٽ�������룬����Һ��������Ũ�ȼ�С������������Ũ������
��2�������̼���Ƶ����ʵ�����ȣ����߷�Ӧ���ɲ���һ���ƣ���Һ�����ԣ�˵������������ӵ���̶ȴ���ˮ��̶ȣ���������غ�͵���غ���
��3������ҺΪ������Һ��
��4��C2O42-������ȫʱ����CO32- ��Ũ��Ϊa mol/L����̼����ܶȻ�������Һ��c��Ca 2+ ����������ʾ����Һ��C2O42-��Ũ�ȵı仯����ƽ��ʱC2O42-��Ũ�ȣ��ɲ�����ܶȻ��з��̼��㣻
��5����pH�Ʋ�����Һ��pH�жϣ�
��2�������̼���Ƶ����ʵ�����ȣ����߷�Ӧ���ɲ���һ���ƣ���Һ�����ԣ�˵������������ӵ���̶ȴ���ˮ��̶ȣ���������غ�͵���غ���
��3������ҺΪ������Һ��
��4��C2O42-������ȫʱ����CO32- ��Ũ��Ϊa mol/L����̼����ܶȻ�������Һ��c��Ca 2+ ����������ʾ����Һ��C2O42-��Ũ�ȵı仯����ƽ��ʱC2O42-��Ũ�ȣ��ɲ�����ܶȻ��з��̼��㣻
��5����pH�Ʋ�����Һ��pH�жϣ�
���
�⣺��1�����ݵ���ƽ�ⳣ��֪������ǿ��˳���ǣ��������������ӣ�̼�̼��������ӣ����Բ����̼���Ʒ�Ӧ���ɲ����ƺ�̼�����ƣ����ӷ���ʽΪ��H2C2O4+2CO32-=C2O42-+2HCO3-����ˮϡ�Ͳ���ٽ�������룬����Һ��������Ũ�ȼ�С������������Ũ������
A������������Ũ��������ԭ��Ũ�ȼ�С����������Ũ��������������Ũ��֮�ȼ�С���ʴ���
B����ˮϡ�ʹٽ�������룬�������Ӹ�����������Ӹ�����С������������Ũ����������Ũ��֮��������ȷ��
C����ˮϡ�ʹٽ���������ӵ��룬��������Ӹ���������������Ӹ�����С�����Բ��������Ũ��������������Ũ��֮��������ȷ��
D���¶Ȳ��䣬����ĵ���ƽ�ⳣ�����䣬�ʴ���
�ʴ�Ϊ��H2C2O4+2CO32-=C2O42-+2HCO3-��BC��
��2�������̼���Ƶ����ʵ�����ȣ����߷�Ӧ���ɲ���һ���ƣ���Һ�����ԣ�˵������������ӵ���̶ȴ���ˮ��̶ȣ�
A.10mL0.1mol?l-1H2C2O4��Һ�м���VmL0.1mol?L-1NH3?H2O��Һ��������ҺpH=7���������Һ�����ԣ�Ҫʹ��Һ�����ԣ�һˮ�ϰ������ʵ���Ӧ��������������V��20mL������ȷ��
B�������ʵ���Ũ�ȵ�NaHC2O4��Na2C2O4��Na2CO3��Һ�У��������ˮ��̶ȣ�̼������ӣ�̼��������ӣ�����������ӣ��������ˮ��̶�Խ����Һ��PHԽ��������������Һ��pH��С˳��Ϊ��Na2CO3��Na2C2O4��NaHC2O4������ȷ��
C������������Һ�в���������ӵ���̶ȴ���ˮ��̶ȣ���Һ�����ԣ�ˮ������������ӣ�����������Ũ�ȴ��ڲ��������Ũ�ȣ���Һ������������Ũ����С���ʴ���
D�������ʵ���Ũ�ȵ�Na2C2O4��NaHCO3���Һ�У����������غ��c��H2CO3��+c��CO32- ��+c��HCO3-��=c��C2O42- ��+c��HC2O4-��+c��H2C2O4�����ʴ���
�ʴ�Ϊ������������ӵ���̶ȴ���ˮ��̶ȣ�AB��
��3������Ũ�Ⱦ�Ϊ0.1mol?L-1��Na2CO3��NaHCO3�����Һ�м�������������ʱ��������̼��������ӵ��룬��������������̼�������ˮ�⣬��Һ��ҺpH�仯����
A��H2C2O4��NaHC2O4�в����ܺͼӦ�����������ܺͼӦ�������ֿܵ���������������ҺpHӰ�죬�ʴ���
B��NH4Cl��NaCl���Ȼ���ܺ��ᵫ���ܺͼӦ�����Բ��ֿܵ���������������ҺpHӰ�죬����ȷ��
C��NaHSO3��Na2SO3���ܺ����Ӧ�������ֿܵ���������������ҺpHӰ�죬�ʴ���
D��Na2S2O3��KNO3���ᷴӦ�����ͼӦ�����Բ��ֿܵ���������������ҺpHӰ�죬����ȷ��
�ʴ�Ϊ��������������ʱ�������Ӻ�̼������ӷ�Ӧ����̼��������ӣ�ʹ�����ӵ������Ӽ��٣�������������ʱ�����������Ӻ�̼�����������������̼������ӣ�ʹ�����ӵ����������Ӽ��٣���������������������Һ��pHӰ�첻��BD��
��4��C2O42-������ȫʱ����CO32- ��Ũ��Ϊa mol/L����̼����ܶȻ���֪����Һ��c��Ca 2+ ��=
mol/L����C2O42-��Ũ�ȵı仯��=��a-
��mol/L��ƽ��ʱC2O42-��Ũ��=0.3mol/L-��a-
��mol/L���ɲ�����ܶȻ���֪��[0.3-��a-
��]��
=2.5��10-9����Һ��̼���Ũ��Զ���ڸ�����Ũ�ȣ����Խ���Ϊ��0.3-a����
=2.5��10-9�����a=0.2��
�ʴ�Ϊ��0.2mol/L��
��5������������������ȫ���룬����Һ��pH=1����pH�Ʋ���0.10mol/L����������Һ��pH����1С��7����֤��HC2O4-��ˮ�д����ŵ���ƽ�⣬�ʴ�Ϊ����pH�Ʋ���0.10mol/L����������Һ��pH����1С��7��
A������������Ũ��������ԭ��Ũ�ȼ�С����������Ũ��������������Ũ��֮�ȼ�С���ʴ���
B����ˮϡ�ʹٽ�������룬�������Ӹ�����������Ӹ�����С������������Ũ����������Ũ��֮��������ȷ��
C����ˮϡ�ʹٽ���������ӵ��룬��������Ӹ���������������Ӹ�����С�����Բ��������Ũ��������������Ũ��֮��������ȷ��
D���¶Ȳ��䣬����ĵ���ƽ�ⳣ�����䣬�ʴ���
�ʴ�Ϊ��H2C2O4+2CO32-=C2O42-+2HCO3-��BC��
��2�������̼���Ƶ����ʵ�����ȣ����߷�Ӧ���ɲ���һ���ƣ���Һ�����ԣ�˵������������ӵ���̶ȴ���ˮ��̶ȣ�
A.10mL0.1mol?l-1H2C2O4��Һ�м���VmL0.1mol?L-1NH3?H2O��Һ��������ҺpH=7���������Һ�����ԣ�Ҫʹ��Һ�����ԣ�һˮ�ϰ������ʵ���Ӧ��������������V��20mL������ȷ��
B�������ʵ���Ũ�ȵ�NaHC2O4��Na2C2O4��Na2CO3��Һ�У��������ˮ��̶ȣ�̼������ӣ�̼��������ӣ�����������ӣ��������ˮ��̶�Խ����Һ��PHԽ��������������Һ��pH��С˳��Ϊ��Na2CO3��Na2C2O4��NaHC2O4������ȷ��
C������������Һ�в���������ӵ���̶ȴ���ˮ��̶ȣ���Һ�����ԣ�ˮ������������ӣ�����������Ũ�ȴ��ڲ��������Ũ�ȣ���Һ������������Ũ����С���ʴ���
D�������ʵ���Ũ�ȵ�Na2C2O4��NaHCO3���Һ�У����������غ��c��H2CO3��+c��CO32- ��+c��HCO3-��=c��C2O42- ��+c��HC2O4-��+c��H2C2O4�����ʴ���
�ʴ�Ϊ������������ӵ���̶ȴ���ˮ��̶ȣ�AB��
��3������Ũ�Ⱦ�Ϊ0.1mol?L-1��Na2CO3��NaHCO3�����Һ�м�������������ʱ��������̼��������ӵ��룬��������������̼�������ˮ�⣬��Һ��ҺpH�仯����
A��H2C2O4��NaHC2O4�в����ܺͼӦ�����������ܺͼӦ�������ֿܵ���������������ҺpHӰ�죬�ʴ���
B��NH4Cl��NaCl���Ȼ���ܺ��ᵫ���ܺͼӦ�����Բ��ֿܵ���������������ҺpHӰ�죬����ȷ��
C��NaHSO3��Na2SO3���ܺ����Ӧ�������ֿܵ���������������ҺpHӰ�죬�ʴ���
D��Na2S2O3��KNO3���ᷴӦ�����ͼӦ�����Բ��ֿܵ���������������ҺpHӰ�죬����ȷ��
�ʴ�Ϊ��������������ʱ�������Ӻ�̼������ӷ�Ӧ����̼��������ӣ�ʹ�����ӵ������Ӽ��٣�������������ʱ�����������Ӻ�̼�����������������̼������ӣ�ʹ�����ӵ����������Ӽ��٣���������������������Һ��pHӰ�첻��BD��
��4��C2O42-������ȫʱ����CO32- ��Ũ��Ϊa mol/L����̼����ܶȻ���֪����Һ��c��Ca 2+ ��=
| 5��10-9 |
| a |
| 5��10-9 |
| a |
| 5��10-9 |
| a |
| 5��10-9 |
| a |
| 5��10-9 |
| a |
| 5��10-9 |
| a |
�ʴ�Ϊ��0.2mol/L��
��5������������������ȫ���룬����Һ��pH=1����pH�Ʋ���0.10mol/L����������Һ��pH����1С��7����֤��HC2O4-��ˮ�д����ŵ���ƽ�⣬�ʴ�Ϊ����pH�Ʋ���0.10mol/L����������Һ��pH����1С��7��
���������⿼����������ʵĵ��롢��������ܽ�ƽ���֪ʶ�㣬���ݵ���ƽ�ⳣ��ȷ���������ǿ��������Խǿ�������������ˮ��̶�ԽС���ٽ���ܶȻ��������м��㣬�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
���л�ѧ����ʽ�У��������ӷ���ʽ2H++CO32-=CO2��+H2O��ʾ���ǣ�������
| A��ϡ�����еμ�С�մ���Һ |
| B��ϡ�����еμӴ�����Һ |
| C��ϡ������ʯ��ʯ��Ӧ |
| D���Ӳ�ľ������ȡ��̼������������ᷴӦ |
����ʵ��װ�ã�����������ʡ�ԣ����ܴﵽʵ��Ŀ���ǣ�������
A�� ����Cl2 |
B�� ����HCl |
C��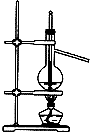 ����ʯ�� |
D��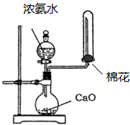 ��ȡ���� ��ȡ���� |
 ��������������Ա��Ϊ����Ӧ�ӽ�������ͼ��ʾ��־��������ǣ�������
��������������Ա��Ϊ����Ӧ�ӽ�������ͼ��ʾ��־��������ǣ�������| A���Ͼɱ�ֽ | B���Ͼɵ�� |
| C���������Ϲ� | D���ϸ��� |


 ��ϩȩ���ṹ��ʽ��CH2=CHCHO����һ����Ҫ���л��ϳ�ԭ�ϣ����Ϊ��
��ϩȩ���ṹ��ʽ��CH2=CHCHO����һ����Ҫ���л��ϳ�ԭ�ϣ����Ϊ��