��Ŀ����
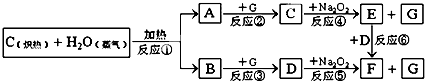
A��B��C��D��E��F��Ϊԭ��������������Ķ�����Ԫ�أ��ǽ���Ԫ��A����������������������ͬ��B��������������A������������4����B��D�г��ȼ���������������ȥBD2��E+��D2-������ͬ�ĵ�������A��F��ȼ�շ�����ɫ���棬����ƿ�ڲ�����������ش��������⣺
��1��1molC2�к��еĹ��õ��Ӷ���Ŀ�� ��NA �����İ����ӵ�����ֵ��ʵ������ȡ����F�����ӷ���ʽ ��
��2����B��DE��ɵ����ε�����Ϊ ����KI��Һ��ͨ��F���ʣ���Ӧ�����CCl4 �����л�������ɫ���ù�����֤�������Ԫ�صķǽ�����ǿ����ϵΪ ����Ԫ�ط��ţ���
��3��������A��B��D��E����Ԫ����ɵ��������ʵ���ɺͽṹ��Ϣ�����
a�ĵ���ʽΪ ������ԭ�ӵijɼ��ص�д��b�Ľṹʽ�� ��
��1��1molC2�к��еĹ��õ��Ӷ���Ŀ��
��2����B��DE��ɵ����ε�����Ϊ
��3��������A��B��D��E����Ԫ����ɵ��������ʵ���ɺͽṹ��Ϣ�����
| ���� | ��ɺͽṹ��Ϣ |
| a | ����A��D����Ԫ�صĻ���������к��м��Լ��ͷǼ��Լ� |
| b | ��ѧ���ΪBDF2�Ļ��������BԪ����������ϼ� |
���㣺λ�ýṹ���ʵ����ϵӦ��
ר�⣺
������A��B��C��D��E��F��Ϊԭ��������������Ķ�����Ԫ�أ��ǽ���Ԫ��A����������������������ͬ����AΪ��Ԫ�أ�B��������������A������������4������BΪ�ڢ�A��Ԫ�أ�B��D�г��ȼ���������������ȥBD2����BΪ̼Ԫ�أ�DΪ��Ԫ�أ�����CΪ��Ԫ�أ�E+��D2-������ͬ�ĵ���������EΪ��Ԫ�أ�A��F��ȼ�շ�����ɫ���棬����ƿ�ڲ�����������FΪ��Ԫ�أ�aΪ����A��D����Ԫ�صĻ���������к��м��Լ��ͷǼ��Լ�����aΪH2O2��bΪ��ѧ���ΪBDF2�Ļ��������BԪ����������ϼۣ���bΪCOCl2���ݴ˴��⣮
���
�⣺A��B��C��D��E��F��Ϊԭ��������������Ķ�����Ԫ�أ��ǽ���Ԫ��A����������������������ͬ����AΪ��Ԫ�أ�B��������������A������������4������BΪ�ڢ�A��Ԫ�أ�B��D�г��ȼ���������������ȥBD2����BΪ̼Ԫ�أ�DΪ��Ԫ�أ�����CΪ��Ԫ�أ�E+��D2-������ͬ�ĵ���������EΪ��Ԫ�أ�A��F��ȼ�շ�����ɫ���棬����ƿ�ڲ�����������FΪ��Ԫ�أ�aΪ����A��D����Ԫ�صĻ���������к��м��Լ��ͷǼ��Լ�����aΪH2O2��bΪ��ѧ���ΪBDF2�Ļ��������BԪ����������ϼۣ���bΪCOCl2��
��1����Ϊ�������е����μ�������1molN2�к��еĹ��õ��Ӷ���Ŀ��3NA ��ʵ������Ũ������������̼�����ȡ��������Ӧ�����ӷ���ʽΪMnO2+4H++2Cl-
Mn2++2H2O+Cl2����
�ʴ�Ϊ��3NA ��MnO2+4H++2Cl-
Mn2++2H2O+Cl2����
��2����B��DE��ɵ�����ΪNa2CO3����������Ϊ����մ���KI��Һ��ͨ����������Ӧ�����CCl4 �����л�������ɫ��˵���������û��⣬֤������Ԫ�صķǽ�����ǿ�ڵ⣬���Էǽ�����ǿ����ϵΪCl��I��
�ʴ�Ϊ������մ�Cl��I��
��3����������ķ�����֪��aΪH2O2��a�ĵ���ʽΪ ��bΪ
��bΪ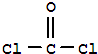 ��
��
�ʴ�Ϊ�� ��
��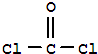 ��
��
��1����Ϊ�������е����μ�������1molN2�к��еĹ��õ��Ӷ���Ŀ��3NA ��ʵ������Ũ������������̼�����ȡ��������Ӧ�����ӷ���ʽΪMnO2+4H++2Cl-
| ||
�ʴ�Ϊ��3NA ��MnO2+4H++2Cl-
| ||
��2����B��DE��ɵ�����ΪNa2CO3����������Ϊ����մ���KI��Һ��ͨ����������Ӧ�����CCl4 �����л�������ɫ��˵���������û��⣬֤������Ԫ�صķǽ�����ǿ�ڵ⣬���Էǽ�����ǿ����ϵΪCl��I��
�ʴ�Ϊ������մ�Cl��I��
��3����������ķ�����֪��aΪH2O2��a�ĵ���ʽΪ
 ��bΪ
��bΪ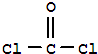 ��
���ʴ�Ϊ��
 ��
��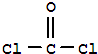 ��
��
���������⿼��Ԫ�ػ������ƶϣ���Ŀ�漰��֪ʶ��϶࣬�����ڿ���ѧ������ѧ֪ʶ���ۺ�Ӧ����������Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 �����̸�Ӯ����ٸ�Ч�����ܸ�ϰ���ϿƼ�������ϵ�д�
�����̸�Ӯ����ٸ�Ч�����ܸ�ϰ���ϿƼ�������ϵ�д� �����ҵ�����������ѧ���ӳ�����ϵ�д�
�����ҵ�����������ѧ���ӳ�����ϵ�д� ����ѧ��Ӯ�����ϵ�д�
����ѧ��Ӯ�����ϵ�д�
�����Ŀ
�������ʵĻ������÷�Һ©�����з�����ǣ�������
����������ˮ �����Ȼ�̼�ͱ� �۱��ͻ����� �ܱ���ˮ��
����������ˮ �����Ȼ�̼�ͱ� �۱��ͻ����� �ܱ���ˮ��
| A���٢� | B���ڢ� | C���٢� | D���ۢ� |
���л������������ֹ����ŵ��ǣ�������
A�� |
B�� |
C�� |
D�� |