��Ŀ����
��֪��pAg=-lg{c(Ag+)},KspAgCl=1��10-12��ͼ����10mLAgNO3��Һ������0.1 mol/L��NaCl��Һʱ����Һ��pAg���ż���NaCl��Һ���������λmL���仯��ͼ��(ʵ��)������ͼ���������н�����ȷ���ǣ� ��
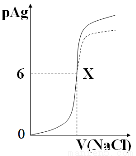
����ʾ��KspAgCl��KspAgI��
A��ԭAgNO3��Һ�����ʵ���Ũ��Ϊ0.1 mol��L-1
B��ͼ��x�������Ϊ��10��6 ��
C��ͼ��x���ʾ��Һ��Ag+ ��Cl�� Ũ����ͬ
D����0.1 mol��L-1��NaCl����0.1 mol��L-1NaI��ͼ�����յ���Ϊ���߲���
C
��������
���������A�������ͼ��֪����������Ȼ�����Һ�����Ϊ0ʱ��pAg��0������������Һ�����ʵ���Ũ����1.0 mol��L-1������B��pAg��6ʱ������ǡ�÷�Ӧ���Ȼ�����Һ�������100ml��x�������Ϊ��100��6 ��������C��KspAgCl=1��10-12��x��pAgΪ6��������Ũ��Ϊ1.0��10-6mol/L��������Ũ��Ϊ1.0��10-6mol/L��������ͬ����ȷ��D������KspAgCl��KspAgI���������0.1 mol��L-1NaI����ͼ�����յ���ΪӦ����ʵ�ߵ���࣬����
���㣺��������ܽ�ƽ�⡣

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д���11�֣�ij�о���ѧϰС������ȷ�Ӧʵ��չ���о������и��л�ѧ�̲ģ�����ѡ�ޣ��жԡ����ȷ�Ӧ��������������������������Ӧ�ų��������ȣ�������ҫ�۵Ĺ�â������ֽ©�����²����մ���������������ɳ�С������ġ���ѧ�ֲᡷ֪��Al��Al2O3��Fe��Fe2O3�۵㡢�е��������£�
���� | Al | Al2O3 | Fe | Fe2O3 |
�۵�/�� | 660 | 2054 | 1535 | 1462 |
�е�/�� | 2467 | 2980 | 2750 | - |
I.��1��ijͬѧ�Ʋ⣬���ȷ�Ӧ���õ���������Ӧ�������Ͻ������ǣ��÷�Ӧ�ų�������ʹ���ۻ����������۵�����ͣ���ʱҺ̬���������ۺ��γ������Ͻ�����Ϊ���Ľ����Ƿ��������_________________(�������������)
��2�����һ����ʵ�鷽����֤���������õĿ�״�������к��н���������ʵ�������Լ���______________,��Ӧ�����ӷ���ʽΪ_________________________________��
��3��ʵ�����ܽ������������Լ�����õ���____________(�����)��
A��Ũ���� B��ϡ����C��ϡ���� D������������Һ
II��ʵ���о����֣����ᷢ��������ԭ��Ӧʱ�������Ũ��Խϡ����Ӧ��ԭ�����е�Ԫ�صĻ��ϼ�Խ�͡�ijͬѧȡһ������������������һ������ϡ�������ַ�Ӧ����Ӧ������������ų����ڷ�Ӧ���������Һ�У���μ���4mol��L����������������Һ����������������Һ�����(mL)������ij��������ʵ���(mol)�Ĺ�ϵ��ͼ��ʾ��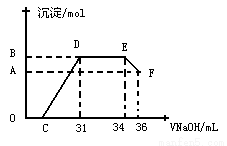 �Իش��������⣺
�Իش��������⣺
��1��ͼ��OC��û�г������ɣ��˽η�����Ӧ�����ӷ���ʽΪ________________��
��2����DE�Σ����������ʵ���û�б仯����˽η�����Ӧ�����ӷ���ʽ_______________ ��
��������˵����Һ��_________________���OH����������_______ǿ�������ӷ��ţ���
��3�� B��A�IJ�ֵΪ_________mol��B���Ӧ�ij��������ʵ���Ϊ________mol��C���Ӧ������������Һ�����Ϊ___________mL
 B��
B�� C��CH4O D��C
C��CH4O D��C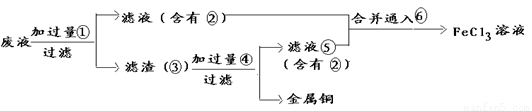
 ��Һ �ۼ�������KSCN��Һ
��Һ �ۼ�������KSCN��Һ �dz�����ѱ����֮һ������ͨ���ⶨ������ˮ��
�dz�����ѱ����֮һ������ͨ���ⶨ������ˮ��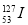 ��ͬ
��ͬ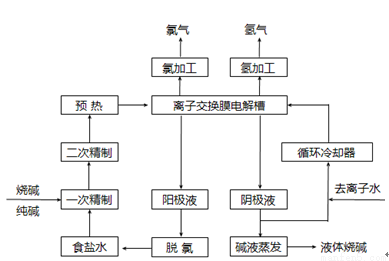
 2AlN��3CO�ϳɡ��÷�Ӧ�У�ÿ����1 mol AlN��ת��1.5 mol����
2AlN��3CO�ϳɡ��÷�Ӧ�У�ÿ����1 mol AlN��ת��1.5 mol���� 2C��2D�����ֲ�ͬ����µķ�Ӧ���ʷֱ�Ϊ��v(A)��0.15 mol/(L��s)����v(B)��0.6 mol/(L��s)����v(C)��0.4 mol/(L��s)����v(D)��0.45 mol/(L��s)���÷�Ӧ���еĿ���˳��Ϊ��>�ۣ���>��
2C��2D�����ֲ�ͬ����µķ�Ӧ���ʷֱ�Ϊ��v(A)��0.15 mol/(L��s)����v(B)��0.6 mol/(L��s)����v(C)��0.4 mol/(L��s)����v(D)��0.45 mol/(L��s)���÷�Ӧ���еĿ���˳��Ϊ��>�ۣ���>�� ��4NH
��4NH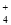 ��2H2O
��2H2O