��Ŀ����
7��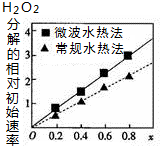 ��֪CoxNi��1-x��Fe2O4������Co��Ni��Ϊ+2�ۣ�������H2O2�ֽ�Ĵ��������нϸߵĻ��ԣ�ͨ�������ֲ�ͬ�����Ƶã���ͼΪ���ַ����ƵõĴ�������10��ʱ���ֽ�6%��H2O2��Һ����Գ�ʼ������x�仯������ͼ������˵��������ǣ�������
��֪CoxNi��1-x��Fe2O4������Co��Ni��Ϊ+2�ۣ�������H2O2�ֽ�Ĵ��������нϸߵĻ��ԣ�ͨ�������ֲ�ͬ�����Ƶã���ͼΪ���ַ����ƵõĴ�������10��ʱ���ֽ�6%��H2O2��Һ����Գ�ʼ������x�仯������ͼ������˵��������ǣ�������| A�� | �ô�������Ԫ�صĻ��ϼ�Ϊ+3�� | |
| B�� | Co��Ni��Fe��Ϊ��B��Ԫ�� | |
| C�� | ��ͼ����Ϣ��֪��Co2+��Ni2+���������д�Ч���Ϻõ���Co2+ | |
| D�� | ��ͼ����Ϣ��֪����ˮ�ȷ���ȡ�Ĵ������Ը��� |
���� A�����ݻ��ϼ۴�����Ϊ0����Fe�Ļ��ϼۣ�
B��Co��Ni��Fe��Ϊ����Ԫ�أ�
C����xֵԽ��������ķֽ�����Խ��x����Co2+�ı�������
D����������ķֽ�����Խ�������Ը��ߣ�
��� �⣺A��CoxNi��1-x��Fe2O4��Co��Ni��Ϊ+2��OΪ-2�ۣ���Fe�Ļ��ϼ�Ϊ$\frac{4��2-��2x+2-2x��}{2}$=+3����A��ȷ��
B��Co��Ni��Fe��Ϊ����Ԫ�أ�Ԫ�����ڱ���ֻ��7�����壬û�е�VIIIB�壬��B����
C����ͼ��֪����xֵԽ��������ķֽ�����Խ��x����Co2+�ı�������Co2+�Ĵ����Ը��ߣ���C��ȷ��
D����������ķֽ�����Խ�������Ը��ߣ�����ͼ���֪��x��ͬʱ����ˮ�ȷ���ʼ�ٶȴ��ڳ���ˮ�ȷ�������ˮ�ȷ��Ƶô����Ļ��Ը��ߣ���D��ȷ��
��ѡB��
���� ���⿼����Ԫ�ػ��ϼ۵��жϡ����������á�Ԫ�����ڱ��Ľṹ����ͼ��ķ��������ȣ���Ҫѧ���߱�֪ʶ�Ļ������Ѷ��еȣ������ڿ���ѧ���ķ��������Ͷ�ͼ����Ϣ��Ӧ��������

��ϰ��ϵ�д�
�����Ŀ
18��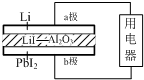 ij﮵�����LiI-Al2O3����Ϊ����ʴ������ӣ�������ṹʾ��ͼ���£�����ܷ�Ӧ�ɱ�ʾΪ��2Li+PbI2=2LiI+Pb������˵����ȷ���ǣ�������
ij﮵�����LiI-Al2O3����Ϊ����ʴ������ӣ�������ṹʾ��ͼ���£�����ܷ�Ӧ�ɱ�ʾΪ��2Li+PbI2=2LiI+Pb������˵����ȷ���ǣ�������
 ij﮵�����LiI-Al2O3����Ϊ����ʴ������ӣ�������ṹʾ��ͼ���£�����ܷ�Ӧ�ɱ�ʾΪ��2Li+PbI2=2LiI+Pb������˵����ȷ���ǣ�������
ij﮵�����LiI-Al2O3����Ϊ����ʴ������ӣ�������ṹʾ��ͼ���£�����ܷ�Ӧ�ɱ�ʾΪ��2Li+PbI2=2LiI+Pb������˵����ȷ���ǣ�������| A�� | ������b�����õ�������a�� | |
| B�� | I-��a��ͨ���������ʴ��ݵ�b�� | |
| C�� | b���ϵĵ缫��ӦʽΪ��PbI2-2e-=Pb+2I- | |
| D�� | b����������1.27 g ʱ��a��ת�Ƶĵ�����ԼΪ6.02��1021 |
19�����й��ڱ����ӵ�˵����ȷ���ǣ�������
| A�� | ����������3��C-C����3��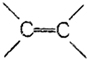 �� �� | |
| B�� | �������е�̼̼����һ�ֽ���C-C����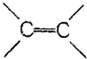 ��֮�������ļ� ��֮�������ļ� | |
| C�� | �������е�6��̼̼��������ȫ��ͬ�� | |
| D�� | �����Ӿ���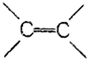 ������ʹ��ˮ��ɫ ������ʹ��ˮ��ɫ |
16�������л������У�����ԭ�Ӳ����ܴ���ͬһƽ����ǣ�������
| A�� | CH2�TCH2 | B�� | CH2�TCH-CH�TCH2 | ||
| C�� | 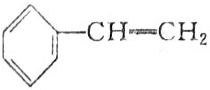 | D�� | 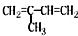 |
19�������ϡ���������þ��Ӧ��ʵ�����ݣ�
�����������ݣ��ش��������⣺
��1��ʵ��4��5���������巴Ӧ��ı�����Է�Ӧ������Ӱ�죬�����Խ��Ӧ����Խ�죬�ܱ���ͬһ���ɵ�ʵ�黹��1��2����ʵ����ţ���
��2�����н�������Ӧ��Ũ�ȶԷ�Ӧ���ʲ���Ӱ����ʵ��2��ʵ��5����ʵ����ţ���
��3����ʵ����Ӱ�췴Ӧ���ʵ��������ػ��з�Ӧ�¶ȣ��ܱ�����һ���ɵķֱ���ʵ��6��ʵ��7��ʵ��8��ʵ��9������ʵ����ţ���
��4��ʵ���е����з�Ӧ����Ӧ���������ʵ��5����ʵ����ţ�����ʵ��������ƽ����Ӧ����Ϊ0.004mol/��L•s����
| ʵ�� ��� | ��������/g | ���� ״̬ | c��H2SO4�� /mol/L | V��H2SO4��/mL | ��Һ�¶�/�� | ������ʧ ��ʱ��/s | |
| ��Ӧǰ | ��Ӧ�� | ||||||
| 1 | 0.24 | ˿ | 0.5 | 100 | 20 | 34 | 500 |
| 2 | 0.24 | ��ĩ | 0.5 | 100 | 20 | 35 | 50 |
| 3 | 0.24 | ˿ | 0.7 | 100 | 20 | 36 | 250 |
| 4 | 0.24 | ˿ | 0.8 | 100 | 20 | 35 | 200 |
| 5 | 0.24 | ��ĩ | 0.8 | 100 | 20 | 36 | 25 |
| 6 | 0.24 | ˿ | 1.0 | 100 | 20 | 35 | 125 |
| 7 | 0.24 | ˿ | 1.0 | 100 | 35 | 50 | 50 |
| 8 | 0.24 | ˿ | 1.1 | 100 | 20 | 34 | 100 |
| 9 | 0.24 | ˿ | 1.1 | 100 | 30 | 44 | 40 |
��1��ʵ��4��5���������巴Ӧ��ı�����Է�Ӧ������Ӱ�죬�����Խ��Ӧ����Խ�죬�ܱ���ͬһ���ɵ�ʵ�黹��1��2����ʵ����ţ���
��2�����н�������Ӧ��Ũ�ȶԷ�Ӧ���ʲ���Ӱ����ʵ��2��ʵ��5����ʵ����ţ���
��3����ʵ����Ӱ�췴Ӧ���ʵ��������ػ��з�Ӧ�¶ȣ��ܱ�����һ���ɵķֱ���ʵ��6��ʵ��7��ʵ��8��ʵ��9������ʵ����ţ���
��4��ʵ���е����з�Ӧ����Ӧ���������ʵ��5����ʵ����ţ�����ʵ��������ƽ����Ӧ����Ϊ0.004mol/��L•s����
16��ij��̬ԭ�ӵ�3d�ܼ�����һ�����ӣ���N���ϵĵ�����Ϊ��������
| A�� | 0 | B�� | 2 | C�� | 3 | D�� | 8 |
17���������Ǻϳ������ص�ԭ�ϣ����������ȩ�ϳɣ�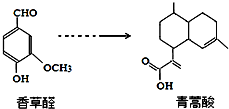 ����������ȷ���ǣ�������
����������ȷ���ǣ�������
 ����������ȷ���ǣ�������
����������ȷ���ǣ�������| A�� | �������������NaOH��Һ�з���ˮ�ⷴӦ | |
| B�� | ��һ�������£����ȩ���������16��ԭ�ӹ��� | |
| C�� | �������ʷֱ��H2��Ӧ���������H24mol��3mol | |
| D�� | ��FeCl3��Һ��NaHCO3��Һ�����Լ������ȩ�������� |
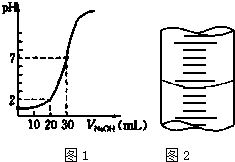 ��һ�����ʵ���Ũ�ȵ�NaOH��Һ�ζ�10.00mL��֪Ũ�ȵ����ᣬ�ζ������ͼ1��ʾ���ش��������⣺
��һ�����ʵ���Ũ�ȵ�NaOH��Һ�ζ�10.00mL��֪Ũ�ȵ����ᣬ�ζ������ͼ1��ʾ���ش��������⣺