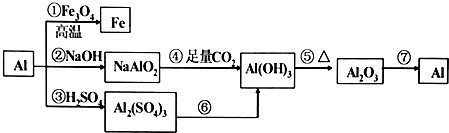
��Ŀ����
13����һƿ������Һ�����п��ܺ���NH4+��K+��Ba2+��Al3+��Fe3+��I-��NO3-��CO32-��SO42-��AlO2-��ȡ����Һ��������ʵ�飺����pH��ֽ���飬��Һ��ǿ���ԣ�
��ȡ��Һ��������������CCl4������������ˮ����CCl4����Ϻ�ɫ��
����ȡ��Һ��������μ���NaOH��Һ��
a����Һ�����Ա�Ϊ���ԣ�b����Һ����������c��������ȫ�ܽ⣻d����������Һ��������ų�����������ʹʪ��ĺ�ɫʯ����ֽ������
��ȡ�����۵õ��ļ�����Һ������Na2CO3��Һ���а�ɫ�������ɣ�
��������ʵ�����ش��������⣮
��1���ɢٿ����ų�CO32-��AlO2-�Ĵ��ڣ�
��2���ɢڿ���֤��I-�Ĵ��ڣ�ͬʱ�ų�Fe3+��NO3-�Ĵ��ڣ�
��3���ɢۿ���֤��Al3+��NH4+�Ĵ��ڣ�д��c��d���漰�Ļ�ѧ����ʽ�������ӷ�Ӧ�������ӷ���ʽ��ʾ��
cAl��OH��3+OH-�TAlO2-+2H2O��dNH3•H2O$\frac{\underline{\;����\;}}{\;}$NH3��+H2O��
��4���ɢܿ����ų�SO42-�Ĵ��ڣ�
���� ����Һ��ǿ���ԣ�˵����Һ�д���H+���������ӹ����֪һ��������CO32-��AlO2-��
��ȡ��Һ��������������CCl4������������ˮ����CCl4����Ϻ�ɫ��˵����Һ��һ������I-���������ӹ�������жϿ�֪��һ��������Fe3+��NO3-��
����ȡ��Һ��������μ���NaOH��Һ��
a����Һ�����Ա�Ϊ���ԣ��к��
b����Һ����������������������ֻ�������ӳ�����
c��������ȫ�ܽ⣬֤�����ɵij���������������ԭ��Һ��һ������Al3+��
d����������Һ��������ų�����������ʹʪ��ĺ�ɫʯ����ֽ������֤�����ɵ������ǰ�����ԭ��Һ��һ������笠����ӣ�
��ȡ�����۵õ��ļ�����Һ�������ӷ�Ӧ����ƫ��������ӣ�����Na2CO3��Һ���а�ɫ�������ɣ��������ӿ�ֻ֪�б���������̼�ᱵ������˵��ԭ��Һ�в�������������ӣ�
�����ƶ����ӵĴ�������ش����⣮
��� �⣺����pH��ֽ���飬��Һ��ǿ���ԣ���Һ�д��ڴ���H+������̼������Ӻ�ƫ��������Ӷ������������Ӻ������ӷ�Ӧ������CO32-��AlO2- ���ܴ������ڣ�
��ȡ��Һ��������������CCl4���������Ƶ���ˮ�����Ȼ�̼�����ɫ��˵����Һ�к���I-������ Fe3+��NO3-��������Һ�У�������I-ΪI2��������Һ�в�����Fe3+��NO3-��
�������������ɵij������ܽ⣬˵����Һ�д���Al3+�����ݼ�����Һ���ɵ�������ʹʪ��ĺ�ɫʯ����ֽ����֤�������ǰ�����˵��ԭ��Һ�к���NH4+��
��ȡ�����۵õ��ļ�����Һ������̼������Һ�а�ɫ�������ɣ�˵����Һ�д��ڱ����ӣ���һ����������������ӣ�
��1���ɢٿ����ų�CO32-��AlO2-�Ĵ��ڣ��ʴ�Ϊ��CO32-��AlO2-��
��2���ɢڿ���֤��I-һ���Ĵ��ڣ�CCl4����ֵⵥ�ʵ���ɫ֤����I-��Fe3+��NO3-�ڸû�������I-���ܹ��棬�������ӹ���ͬʱ�ų�Fe3+��NO3-���ӵĴ��ڣ�
�ʴ�Ϊ��I-��Fe3+��NO3-��
��3����֤����Һ��һ������Al3+��NH4+��c��d���漰�Ļ�ѧ��ӦΪ���������ܽ�������������Һ������ƫ�����ƣ�һˮ�ϰ����ȷֽ����ɰ����������ӷ�Ӧ�������ӷ���ʽ��ʾΪ��Al��OH��3+OH-=AlO2-+H2O��NH3•H2O$\frac{\underline{\;����\;}}{\;}$NH3��+H2O��
�ʴ�Ϊ��Al3+��NH4+��Al��OH��3+OH-=AlO2-+H2O��NH3•H2O$\frac{\underline{\;����\;}}{\;}$NH3��+H2O��
��4����ʵ������жϣ�ȡ�����۵õ��ļ�����Һ�������ӷ�Ӧ����ƫ��������ӣ�����Na2CO3��Һ���а�ɫ�������ɣ��������ӿ�ֻ֪�б���������̼�ᱵ������˵��ԭ��Һ�в�������������ӣ�
�ʴ�Ϊ��SO42-��
���� ���⿼�������Ӽ����ʵ�鷽���ͷ�Ӧ������жϣ��������ʺ����ӷ�Ӧ���е����ӹ�������ǽ���ؼ�����Ŀ�Ѷ��еȣ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�| A�� | 3��3-�������� | B�� | 2��3-����-4-�һ����� | ||
| C�� | 2��3��3-�������� | D�� | 2��2-����-3-�һ����� |
| A�� | 2��3-�������� | B�� | 3��3-�������� | ||
| C�� | 3-��-2-�һ����� | D�� | 2��2��3��3-�ļ����� |
| A�� | ��ʧ���ӵ�ԭ�ӣ���õ��ӵ�����һ��ǿ | |
| B�� | �õ��ӵ�ԭ�����γɵļ������ӣ��仹ԭ��һ��ǿ | |
| C�� | ���ý�������÷ǽ������ϣ����γ����Ӽ� | |
| D�� | ���Ӳ�ṹ��ͬ�IJ�ͬ���ӣ��˵����Խ�࣬�뾶Խ�� |
| A�� | ��״���£�22.4LH2O���еķ�����ΪNA | |
| B�� | 28gN2��N4��ɵĻ�������к��е�ԭ����Ϊ2NA | |
| C�� | 1mol•L-1AlCl3��Һ�к��е�Cl-��Ϊ3NA | |
| D�� | ��1mol CO2����1Lˮ�У�������Һ������������ΪNA |
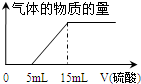 ij��ѧ��ȤС����100mL 1mol/L NaOH��Һ��ȫ������a mol CO2��õ���ҺA��Һ������ޱ仯����Ϊ��ȷ����ҺA�����ʳɷּ�aֵ������ȤС���ͬѧ����������ʵ�飮������������������Ӧʵ�����ݣ�
ij��ѧ��ȤС����100mL 1mol/L NaOH��Һ��ȫ������a mol CO2��õ���ҺA��Һ������ޱ仯����Ϊ��ȷ����ҺA�����ʳɷּ�aֵ������ȤС���ͬѧ����������ʵ�飮������������������Ӧʵ�����ݣ�