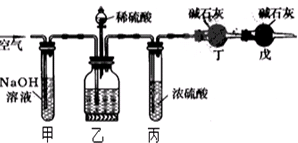
��Ŀ����
10��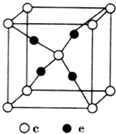 ���ڱ�ǰ�����ڵ�Ԫ��a��b��c��d��e��ԭ��������������a�ĺ����������������������ͬ��b�ļ۵��Ӳ��е�δ�ɶԵ�����3����c������������Ϊ���ڲ��������3����d��cͬ�壻e�������ֻ��1�����ӣ����������18�����ӣ��ش��������⣺
���ڱ�ǰ�����ڵ�Ԫ��a��b��c��d��e��ԭ��������������a�ĺ����������������������ͬ��b�ļ۵��Ӳ��е�δ�ɶԵ�����3����c������������Ϊ���ڲ��������3����d��cͬ�壻e�������ֻ��1�����ӣ����������18�����ӣ��ش��������⣺��1��b��c��d�е�һ������������N����Ԫ�ط��ţ���e�ļ۲�����Ų�ͼΪ��
 ��
����2��a����������Ԫ���γɵĶ�Ԫ���ۻ������У����ӳ������Σ��÷��ӵ�����ԭ�ӵ��ӻ���ʽΪsp3�������мȺ��м��Թ��ۼ����ֺ��зǼ��Թ��ۼ��Ļ�������H2O2��N2H4��C2H6�ȣ��ѧʽ��д�����֣���
��3����ЩԪ���γɵĺ������У�����������νṹ������H2SO3�����ѧʽ��
��4��e��c�γɵ�һ�����ӻ�����ľ���ṹ��ͼ��ʾ����e�����������Ϊ+1��
���� ���ڱ�ǰ�����ڵ�Ԫ��a��b��c��d��e��ԭ��������������a�ĺ����������������������ͬ����a��HԪ�أ�c������������Ϊ���ڲ��������3����ԭ��������������8������C��OԪ�أ�d��cͬ�壬��d��SԪ�أ�b�ļ۵��Ӳ��е�δ�ɶԵ�����3������ԭ������С��c����b��NԪ�أ�e�������ֻ��һ�����ӣ����������18�����ӣ���e��CuԪ�أ��ٽ��ԭ�ӽṹ�����ʽṹ��Ԫ�������ɽ��
��� �⣺���ڱ�ǰ�����ڵ�Ԫ��a��b��c��d��e��ԭ��������������a�ĺ����������������������ͬ����a��HԪ�أ�
c������������Ϊ���ڲ��������3����ԭ��������������8������C��OԪ�أ�d��cͬ�壬��d��SԪ�أ�
b�ļ۵��Ӳ��е�δ�ɶԵ�����3������ԭ������С��c����b��NԪ�أ�
e�������ֻ��һ�����ӣ����������18�����ӣ���e��CuԪ�أ�
��1��ͬһ����Ԫ�أ�Ԫ�ص�һ����������ԭ��������������������ƣ�����IIA��͵�VA��Ԫ�ص�һ�����ܴ���������Ԫ�أ�ͬһ����Ԫ���У�Ԫ�ص�һ����������ԭ�������������С������b��c��dԪ�ص�һ������������NԪ�أ�e�ļ۲����Ϊ3d��4s���ӣ���۲�����Ų�ͼΪ ��
��
�ʴ�Ϊ��N�� ��
��
��2��a��HԪ�أ�a������Ԫ���γɵĶ�Ԫ���ۻ������У����ӳ������Σ��÷���Ϊ���������������е�ԭ�Ӻ���3�����ۼ���һ���µ��Ӷԣ����Ը÷��ӵ�����ԭ�ӵ��ӻ���ʽΪsp3�������мȺ��м��Թ��ۼ����ֺ��зǼ��Թ��ۼ��Ļ�������H2O2��N2H4��
�ʴ�Ϊ��sp3��H2O2��N2H4��
��3����ЩԪ���γɵĺ������У�H2SO3��Sԭ���γ���3���� �����������ṹ���ʴ�Ϊ��H2SO3��
��4��e��c�γɵ�һ�����ӻ�����ľ���ṹ��ͼ1��c���Ӹ���=1+8��$\frac{1}{8}$=2��e���Ӹ���=4�����Ըû�����ΪCu2O����e���ӵĵ��Ϊ+1���ʴ�Ϊ��+1��
���� ���⿼������λ�á��ṹ�����ʵ��ۺ�Ӧ�ã���Ŀ�Ѷ��еȣ��ƶ�Ԫ��Ϊ���ؼ������ؿ���ѧ���ռ�����������֪ʶ�����������漰�������㡢ԭ�ӽṹ��֪ʶ�㣬�ۺ��Խ�ǿ�����þ�̯�����۲���ӶԻ������۵����۷������

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�| A�� | ���³�ѹ�£�11.2 L��������ԭ����ĿΪNA | |
| B�� | 2 L 0.2 mol/L K2SO4��ҺSO42-���ʵ���Ũ��Ϊ0.4 mol/L | |
| C�� | 1 mol Na����ԭ�����ṩ������ΪNA | |
| D�� | ͬ�¡�ͬѹ��NA��CO2���Ӻ�NA��O2���ӵ������ͬ |

| A�� | �ù����ĩ��һ��������BaCl2 | |
| B�� | �ù����ĩ��һ������KNO3 | |
| C�� | ������ɿ�����CaCO3��BaCl2��CuSO4 | |
| D�� | �������һ����CaCO3��Na2SO4��KNO3 |
��1���״������Ǽ��Է��ӣ�����ԡ��Ǽ��ԡ�����
��2����ҵ��һ��ɲ������·�Ӧ���ϳɼ״���CO��g��+2H2��g��?CH3OH��g����H=-86.6KJ/mol����T��ʱ����һ������̶�Ϊ1L���ܱ������м���1mol CO��2mol H2����Ӧ�ﵽƽ��ʱ�������ڵ�ѹǿ�ǿ�ʼʱ��$\frac{3}{5}$��
�ٴﵽƽ��ʱ��CO��ת����Ϊ60%
������ѡ�����жϸ÷�Ӧ�ﵽƽ��״̬�����ݵ���ce
a��2v��H2��=v��CH3OH��
b��CO���������ʵ���CH3OH����������
c�������ڵ�ѹǿ���ֲ���
d�����������ܶȱ��ֲ���
e����������ƽ����Է�����������ʱ����仯
��3����֪�ڳ��³�ѹ�£�
��2CH3OH��1��+3O2��g���T2CO2��g��+4H2O��g����H=-akJ•mol-1
��2CO��g��+O2��g���T2CO2��g����H=-bkl•mol-1
��H2O��g���TH2O��1����H=-ckJ•mol-1
��CH3OH��1��+O2��g���TCO��g��+2H2O��1����H=$\frac{b-a-4c}{2}$kJ•mol-1
��4���ɼ״���������NaOH��Һ���ɵ������ֻ���أ���ʹ�ֻ�����ʹ��һ���²ų�һ�ε磮
�ٸõ�ظ����ĵ缫��ӦʽΪCH3OH-6e-+8OH-=CO32-+6H2O��
�����Ըõ��Ϊ��Դ����ʯī���缫���200mL����������ӵ���Һ��
| ���� | Cu2+ | H+ | Cl- | SO42- |
| Ũ�ȣ�c/mol•L-1�� | 0.5 | 2 | 2 | 0.5 |
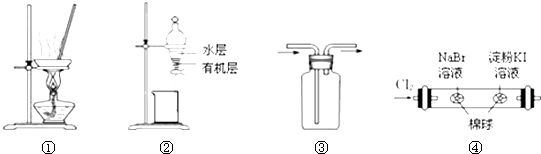
| A�� | ��ͼ����ʾװ�ã�����NH4Cl������Һ�Ʊ�NH4Cl���� | |
| B�� | ��ͼ����ʾװ�ã�����CCl4��ȡ��ˮ����л����ˮ�� | |
| C�� | ��װ�â���ʾ����������������ռ�H2��NH3�� | |
| D�� | ��ͼ����ʾװ�ã�����֤�������ԣ�Cl2��Br2��I2 |
��1����֪��CH4��g��+2O2��g���TCO2��g��+2H2O��g����H1=-820kJ/mol
CO��g��+H2O��g���TCO2��g��+H2��g����H2=-41.2kJ/mol
2CO��g��+O2��g���T2CO2��g����H3=-566kJ/mol��Ӧ��
CO2��g��+CH4��g���T2CO��g��+2H2��g����H=+229.6kJ/mol
��2����ҵ�ϣ�������̫������CO2Ϊԭ����ȡC����ԭ����ͼ1��ʾ������������FeO���Fe3O4����FeO�����Ƿ�Ӧ�Ĵ���������ϵͳ�з����ķ�ӦΪ��6FeO+CO2$\frac{\underline{\;700K\;}}{\;}$2Fe3O4+Cÿ����1mol Fe3O4��ת�Ƶ��ӵ����ʵ���Ϊ2mol��
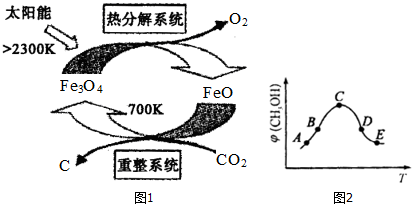
��3��CO2�������ںϳɼ״�����Ӧ����ʽΪ��CO2��g��+3H2��g��?CH3OH��g��+H2O��g����H��0
�ٸ÷�Ӧ��ƽ�ⳣ������ʽΪK=$\frac{c��C{H}_{3}OH��•c��{H}_{2}O��}{c��C{O}_{2}��•{c}^{3}��{H}_{2}��}$��
���ں��ݵ��ܱ������У�����H2��CO2�Ļ�����壬��ͬ�¶���������Ӧ��ͬʱ���ü״������������CH3OH����ͼ2��ʾ��ͼ��A��B��C��D��E������Ӧ��״̬�У�����ƽ��״̬����C��D��E������ĸ����B���E��ķ�Ӧ���ʴ�С��ϵΪv��B����v��E���������������������=������
��һ���¶��£��ڼס��������ݻ���ͬ�ĺ����ܱ������У�����H2��CO2�Ļ�����壮
| ���� | �� | �� |
| ��Ӧ��Ͷ���� | 1molCO2��3molH2 | a molCO2��b molH2��c molCH3OH��g����c molH2O��g�� |