ћвƒњƒЏ»Ё
оў(Pd)іяїѓ≈ЉЅ™Јі”¶ «љьƒкјі”–їъЇѕ≥…µƒ—–Њњ»»µг÷Ѓ“ї°£јэ»з£Ї

їѓЇѕќпҐсњ…”…“‘ѕ¬Їѕ≥…¬Јѕяїсµ√£Ї

(1)їѓЇѕќпҐс”лBr2ЈҐ…ъЉ”≥…Јі”¶Ћщµ√≤ъќпµƒљбєєЉт љќ™______________°£
(2)їѓЇѕќпҐтµƒЈ÷„” љќ™________°£
(3)їѓЇѕќпҐу”лїѓЇѕќпҐт‘ЏЋбіяїѓѕ¬ЈҐ…ъх•їѓЈі”¶µƒїѓ—ІЈљ≥ћ љќ™_____________________________________________________ („Ґ√чћхЉю)°£
(4)їѓЇѕќпҐфµƒљбєєЉт љќ™______________°£
(5)Ґх «їѓЇѕќпҐтµƒЌђЈ÷“мєєће°£ҐхµƒЈ÷„”÷–±љїЈ…ѕ”–»эЄц»°іъїщ£ђƒ№ЈҐ…ъ“шЊµЈі”¶£ђ«“±љїЈ…ѕµƒ“ї¬»іъќп”–2÷÷°£ҐхµƒљбєєЉт љњ…ƒ№ «______________°£
(6)1Ј÷„” ”л1Ј÷„”
”л1Ј÷„” ‘Џ“їґ®ћхЉюѕ¬њ…ЈҐ…ъјаЋ∆Јі”¶ҐўµƒЈі”¶£ђ…ъ≥…”–їъїѓЇѕќпҐц£ђҐцµƒљбєєЉт љќ™________________°£
‘Џ“їґ®ћхЉюѕ¬њ…ЈҐ…ъјаЋ∆Јі”¶ҐўµƒЈі”¶£ђ…ъ≥…”–їъїѓЇѕќпҐц£ђҐцµƒљбєєЉт љќ™________________°£
°°(1)CH2Br°™CHBr°™COOH
(2)C9H8O2
(3) 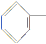 CH==CH°™COOH
CH==CH°™COOH
£ЂCH2==CH°™CH2OH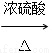
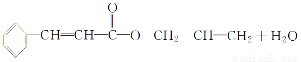
(4)CH2==CH°™CHO
(5) 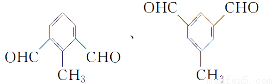
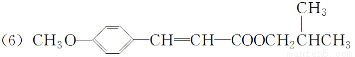
°Њљвќц°њ°°(2)їѓЇѕќпҐтµƒЈ÷„” љќ™C9H8O2°£(3)їѓЇѕќпҐу «CH2==CHCH2ClЋЃљвµ√µљµƒіЉCH2==CHCH2OH°£(5)±љїЈ…ѕ”–»эЄц»°іъїщ£ђ«“±љїЈ…ѕ“ї¬»іъќп”–Ѕљ÷÷£ђ‘т»эЄц»°іъїщ÷–”–ЅљЄцѕаЌђ≤Ґ«“ґ‘≥∆£їƒ№єїЈҐ…ъ“шЊµЈі”¶£ђ‘т”–»©їщ£ђє ҐхµƒљбєєЉт љњ…ƒ№ «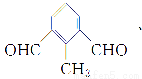

 ‘ƒґЅњм≥µѕµЅ–ір∞Є
‘ƒґЅњм≥µѕµЅ–ір∞ЄSiO2°ҐSO2ЇЌCO2ґЉ «Ћб–‘—хїѓќп£ђЋь√«µƒїѓ—І–‘÷ Њя”–“їґ®µƒѕаЋ∆–‘£їMgЇЌNaµƒїѓ—І–‘÷ “≤Њя”–“їґ®ѕаЋ∆–‘°£
ƒ≥–Ћ»§–°„й”√»зЌЉЋщ Њ„∞÷√љш––Mg”лSO2Јі”¶µƒ µ—й°£

(1)—°‘с÷∆»°SO2µƒЇѕ ‘ЉЅ________(ћо±аЇ≈)°£
Ґў≈®HCl°°ҐЏ≈®H2SO4°°ҐџNa2SO3єћће°°Ґ№CaSO3єћће
(2)…ѕ ц„∞÷√їєњ…”≈їѓ£ђ”≈їѓµƒЈљЈ® «________________________________________£ђ„∞÷√C÷–NaOH»№“Їµƒ„ч”√ «___________________________________________________________
(3)Љ„Ќђ—ІЌ∆≤вMg”лSO2µƒЈі”¶ЇЌMg”лCO2µƒЈі”¶ѕаЋ∆£ђ‘тЄ√Јі”¶Јљ≥ћ љќ™_________________________________________£ї
““Ќђ—ІµƒЌ∆≤в «£Ї2Mg£Ђ3SO2 2MgSO3£ЂS£ї±ыЌђ—ІµƒЌ∆≤в «£Ї3Mg£ЂSO2
2MgSO3£ЂS£ї±ыЌђ—ІµƒЌ∆≤в «£Ї3Mg£ЂSO2 2MgO£ЂMgS£ђ“™—й÷§Љ„°Ґ““°Ґ±ы»эќїЌђ—ІµƒЌ∆≤в «Јс’э»Ј£ђґ°Ќђ—І„ч»зѕ¬ µ—йћљЊњ£Ї
2MgO£ЂMgS£ђ“™—й÷§Љ„°Ґ““°Ґ±ы»эќїЌђ—ІµƒЌ∆≤в «Јс’э»Ј£ђґ°Ќђ—І„ч»зѕ¬ µ—йћљЊњ£Ї
“—÷™£ЇMgSO3ЇЌMgSґЉќҐ»№”ЏЋЃ£ђƒ№”л—ќЋбЈҐ…ъЄіЈ÷љвЈі”¶Ј≈≥ц∆шће£їH2S∆шћеЌ®»лCuSO4»№“Ї÷–≥цѕ÷ЇЏ…Ђ≥Ѕµн°£
ѕё—° ‘ЉЅ£Ї2 mol°§L£≠1—ќЋб°Ґ2 mol°§L£≠1ѕхЋб°Ґ’фЅуЋЃ°Ґ2 mol°§L£≠1 NaOH»№“Ї°Ґ∆ЈЇм»№“Ї°Ґ≥ќ«е ѓї“ЋЃ°Ґ2 mol°§L£≠1 CuSO4»№“Ї£ї“«∆чЇЌ”√∆Ј„‘—°°£
–тЇ≈ | µ—й≤љ÷и | ‘§∆Џѕ÷ѕуЇЌљб¬џ |
Ґў | »°…ўЅњЈі”¶ЇуЋщµ√єћће”Џ ‘є№÷– |
|
ҐЏ | ѕт ‘є№÷–µƒєћће¬э¬эµќЉ”____________£ђ ‘є№њЏ»ы…ѕішµЉє№µƒµ•њ„»ы£ђ≤ҐљЂµЉє№Ќ®»л Ґ”–________µƒ ‘є№÷– | »ф ‘є№÷–µƒ________£ђ‘т±ыЌђ—ІЌ∆≤в’э»Ј£ђ»ф ‘є№÷–µƒєћћеќіЌк»Ђ»№љв£ђ«“________£ђ‘т““Ќђ—ІЌ∆≤в’э»Ј |
ЄщЊЁ…ѕ ц µ—йћљЊњ£ђƒ№÷§√чЉ„Ќђ—ІЌ∆≤в’э»Јµƒ≤ў„чЇЌ‘§∆Џѕ÷ѕу «
_____________________________________________________________°£
(4)…ѕ ц µ—й–и“™100 mL 2 mol°§L£≠1µƒ—ќЋб£ђ≈д÷∆ ±—°”√________(—°ћо10 mL°Ґ25 mL°Ґ50 mLїт100 mL)ЅњЌ≤Ѕњ»°36.5%√№ґ»ќ™1.19 g°§mL£≠1µƒ≈®—ќЋбµƒћеїэќ™________mL°£


