��Ŀ����
��-��ѭ���ֽ�ˮ������Ҫ�漰���з�Ӧ��
�� SO2+2H2O+I2��H2SO4+2HI
�� 2HI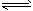 H2+I2
H2+I2
�� 2H2SO4��2SO2+O2+2H2O
��1������������Ӧ�������ж���ȷ����_____________��
a����Ӧ�����ڳ����½���
b����Ӧ����SO2�����Ա�HIǿ
c��ѭ���������貹��H2O
d��ѭ�������в���1mol O2��ͬʱ����1mol H2
��2��һ���¶��£���1L�ܱ������м���1mol HI(g)��������Ӧ��H2���ʵ�����ʱ��ı仯��ͼ��ʾ��
�� SO2+2H2O+I2��H2SO4+2HI
�� 2HI
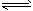 H2+I2
H2+I2 �� 2H2SO4��2SO2+O2+2H2O
��1������������Ӧ�������ж���ȷ����_____________��
a����Ӧ�����ڳ����½���
b����Ӧ����SO2�����Ա�HIǿ
c��ѭ���������貹��H2O
d��ѭ�������в���1mol O2��ͬʱ����1mol H2
��2��һ���¶��£���1L�ܱ������м���1mol HI(g)��������Ӧ��H2���ʵ�����ʱ��ı仯��ͼ��ʾ��
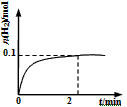
0~2 min�ڵ�ƽ����Ӧ����v(HI) ��___________�����¶��£�H2(g)+I2(g)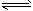 2HI(g)��ƽ�ⳣ��K��_____________����ͬ�¶��£�����ʼ����HI(g)�����ʵ�����ԭ����2������________��ԭ����2����
2HI(g)��ƽ�ⳣ��K��_____________����ͬ�¶��£�����ʼ����HI(g)�����ʵ�����ԭ����2������________��ԭ����2����
a��ƽ�ⳣ�� b��HI��ƽ��Ũ�� c���ﵽƽ���ʱ�� d��ƽ��ʱH2���������
��3��ʵ������Zn��ϡ������ȡH2����Ӧʱ��Һ��ˮ�ĵ���ƽ��__________�ƶ�(����� �����ҡ����ߡ�����)�����������������Լ��е�______________������H2�����ʽ�����
a��NaNO3 b��CuSO4 c��CH3COONa d��NaHSO3
 2HI(g)��ƽ�ⳣ��K��_____________����ͬ�¶��£�����ʼ����HI(g)�����ʵ�����ԭ����2������________��ԭ����2����
2HI(g)��ƽ�ⳣ��K��_____________����ͬ�¶��£�����ʼ����HI(g)�����ʵ�����ԭ����2������________��ԭ����2���� a��ƽ�ⳣ�� b��HI��ƽ��Ũ�� c���ﵽƽ���ʱ�� d��ƽ��ʱH2���������
��3��ʵ������Zn��ϡ������ȡH2����Ӧʱ��Һ��ˮ�ĵ���ƽ��__________�ƶ�(����� �����ҡ����ߡ�����)�����������������Լ��е�______________������H2�����ʽ�����
a��NaNO3 b��CuSO4 c��CH3COONa d��NaHSO3
��1��c
��2��0.1mol/(L��min)��64��b
��3�����ң�b
��2��0.1mol/(L��min)��64��b
��3�����ң�b

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
 ��2010?ɽ������һ��ѭ���ֽ�ˮ������Ҫ�漰���з�Ӧ��
��2010?ɽ������һ��ѭ���ֽ�ˮ������Ҫ�漰���з�Ӧ�� H2+I2
H2+I2 2HI��g����ƽ�ⳣ��K=
2HI��g����ƽ�ⳣ��K= ��һ��ѭ���ֽ�ˮ������Ҫ�漰���з�Ӧ��
��һ��ѭ���ֽ�ˮ������Ҫ�漰���з�Ӧ��












 H2+I2
H2+I2