��Ŀ����
19��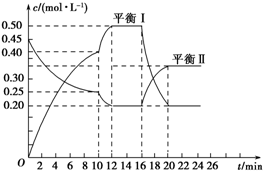 ��һ�ݻ�������ܱ������г���һ����A��B���������·�Ӧ��xA��g��+2B��s��?yC��g����H��0
��һ�ݻ�������ܱ������г���һ����A��B���������·�Ӧ��xA��g��+2B��s��?yC��g����H��0��һ�������£�������A��C�����ʵ���Ũ����ʱ��仯��������ͼ����ش��������⣺
��1����A��Ũ�ȱ仯��ʾ�÷�Ӧ0��10min�ڵ�ƽ����Ӧ����v��A��=0.02mol/��L•min����
��2������ͼʾ��ȷ��x��y=1��2��
��3��0��10min������ѹǿ����������䡱��С������
��4���Ʋ��10min�������߱仯�ķ�Ӧ���������Ǣܢޣ���16min�������߱仯�ķ�Ӧ���������Ǣܣ���д��ţ���
�ټ�ѹ��������A��Ũ�ȡ�������C�����������¡��ݽ��¡��Ӵ���
��5����ƽ����ƽ�ⳣ��ΪK1��ƽ���ƽ�ⳣ��ΪK2����K1�� K2 ���������=����������
��6����������Ϊ�жϡ�xA��g��+2B��s��?yC��g����H��0���ﵽƽ��ı�־���ǣ��ۢܢݣ�
��A������������C������������ȡ��ڵ�λʱ��������a mol A��ͬʱ����2a mol C
��B���������ٱ仯���ܻ���������ѹǿ���ٱ仯 �ݻ��������ܶȲ��ٱ仯��
���� ��1������ͼ������0��10min��A��Ũ�ȱ仯�������û�ѧ��Ӧ���ʹ�ʽ���㣻
��2������0��10min��A��C�����ʵ���Ũ�ȱ仯�ı�ֵ��ã�
��3�����ݷ�Ӧǰ�������������С�Ƚϣ�
��4����ͼ��֪����10minʱ����λʱ����A��C�����ʵ����仯���ϴ�Ӧ��������������Ӱ�����ʵ����ط��������ݵ�16minʱƽ���ƶ����������
��5����֪��16minʱ�ı���������¶ȣ������¶ȶ�ƽ���Ӱ�������
��6����A������������C������������ȣ�����֮�Ȳ����ڼ�����֮�ȣ���
�ڵ�λʱ��������a mol A��ͬʱ����2a mol C������ӳ����Ӧ����
��B���������ٱ仯��˵�����淴Ӧ������ȣ���
�ܸ÷�ӦΪ���������С�ķ�Ӧ��������������ѹǿ���ٱ仯����˵�������ʵ�Ũ�Ȳ��ٷ����仯���ݴ��жϣ�
�ݻ��������ܶȲ��ٱ仯��˵��������������䣮
��� �⣺��1��0��10min�ڡ�n��A��=0.45mol-0.25mol=0.2mol��v��A��=$\frac{��0.45-0.25��mol/L}{10min}$=0.02mol/��L•min�����ʴ�Ϊ��0.02mol/��L•min����
��2��0��10min��������A�����ʵ���Ũ�ȱ仯Ϊ��0.45mol/L-0.25mol/L=0.2mol/L��0��10min��������C�����ʵ���Ũ�ȱ仯Ϊ��0.40mol/L��x��y=0.2mol/L��0.40mol/L=1��2���ʴ�Ϊ��1��2��
��3����Ӧǰ�������ʵ���С�ڷ�Ӧ���������ʵ��������ŷ�Ӧ�Ľ��У��������ʵ�����������ѹǿ���ʴ�Ϊ�����
��4����10minʱ����λʱ����A��C�����ʵ����仯���ϴ�Ӧ��������������Ϊ�����¶Ȼ���ʹ�ô�������16minʱ��C�����ʵ�����С��A�����ʵ�������˵��ƽ�����淴Ӧ�����ƶ����÷�ӦΪ���ȷ�Ӧ��ӦΪ�����¶ȣ��ʴ�Ϊ���ܢޣ��ܣ�
��5����֪��16minʱ�ı�������������¶ȣ����÷�Ӧ�Ƿ��ȷ�Ӧ�����������¶�ƽ�����ƣ���ƽ�ⳣ����С������K1��K2���ʴ�Ϊ������
��6����A������������C������������ȣ�����֮�Ȳ����ڼ�����֮�ȣ��ʴ���
�ڵ�λʱ��������a mol A��ͬʱ����2a mol C������ӳ����Ӧ����δ����������Ĺ�ϵ���ʴ���
��B���������ٱ仯��˵�����淴Ӧ������ȣ���ƽ��״̬������ȷ����
�ܸ÷�ӦΪ���������С�ķ�Ӧ��������������ѹǿ���ٱ仯����˵�������ʵ�Ũ�Ȳ��ٷ����仯����ƽ��״̬������ȷ��
�ݻ��������ܶȲ��ٱ仯��˵��������������䣬˵����Ӧ��ƽ��״̬������ȷ��
��ѡ���ۢܢݣ�
���� ���⿼�黯ѧƽ���Լ���Ӧ���ʵ����⣬��Ŀ�Ѷ��еȣ�ע��ͼ�����߱仯�ķ�����������������Է�Ӧ���ʺ�ƽ���ƶ���Ӱ�죮

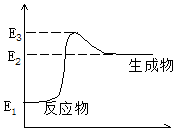 C��s��+H2O��g����CO��g��+H2��g��+Q����Ӧ������������E���ı仯��ͼ��ʾ������˵����ȷ���ǣ�������
C��s��+H2O��g����CO��g��+H2��g��+Q����Ӧ������������E���ı仯��ͼ��ʾ������˵����ȷ���ǣ�������| A�� | ��Сѹǿʱ��Q��ֵ���� | B�� | �����¶�ʱ��Q��ֵ��С | ||
| C�� | �÷�Ӧ�����ȷ�Ӧ | D�� | Q=E3-E1 |
| A�� | ҽѧ�Ͽ���̼�ᱵ�������ᱵ���������͡� | |
| B�� | ������ˮ����ҽ����Һ����Ϊ������ǿ����� | |
| C�� | ҽѧ����Һ�������������ΪҺ���ķе�ܵ� | |
| D�� | ˮ�еĸ�բ�����ӵ�Դ�������������������������������� |
| ��� | �� | �� | �� |
| A | Al2O3 | HNO3 | NaOH |
| B | N2 | O2 | NH3 |
| C | SiO2 | KOH | HF |
| D | SO2 | Ba��OH��2 | NaHCO3 |
| A�� | A | B�� | B | C�� | C | D�� | D |
| ʵ�� ��� | 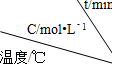 | 0 | 10 | 20 | 30 | 40 | 50 |
| 1 | 500 | 2.0 | 1.6 | 1.3 | 1.0 | 0.8 | 0.8 |
| 2 | 500 | C2 | 1.2 | 0.8 | 0.8 | 0.8 | 0.8 |
| 3 | 500 | C3 | 1.7 | 1.3 | 1.0 | 1.0 | 1.0 |
| 4 | 600 | 2.0 | 1.1 | 0.9 | 0.9 | 0.9 | 0.9 |
��1����ʵ�� 1 �У���Ӧ��10min��20min ��Aƽ������Ϊ0.03 mol/��L•min��
��2����ʵ�� 2 �У�A �ij�ʼŨ�� C2Ϊ2.0 mol/L�����Ʋ�ʵ�� 2 �л�������������ʹ�ô�����
��3����ʵ�� 3 �ķ�Ӧ����Ϊ v3��ʵ�� 1 �ķ�Ӧ����Ϊ v1���� v3��v1���������=������C3�� 2.0mol/L ���������=��
| A�� | ������ϩ����ʹ��ˮ��ɫ���ҷ�Ӧԭ����ͬ | |
| B�� | ��Ȳ�ͱ���Ϊ������������ֻ�ܷ����ӳɷ�Ӧ | |
| C�� | ��ϩ������ϩ������ͨ���ۺϷ�Ӧ�õ��߷��Ӳ��� | |
| D�� | ����ʽΪC10H14������ֻ��һ��ȡ�����ķ�����������ܵĽṹ��3�� |
 ��
��