��Ŀ����
��������Դ��һ����ȾС�Ŀ�������Դ�������ʵ���Ҫת��;������Ҫ������ͼ1��
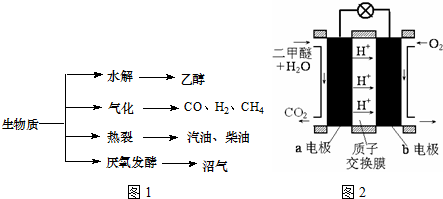
��1�������й�˵����ȷ���� ��
a���������ܣ�������������Դ��̫����
b������ά��ˮ���õ��Ҵ���ȼ������������������
c���������ѽ��õ����͡����͵����ڴ�����
d����ֲ��ո˵��������ͻ�õ���������Ҫ�ɷ��Ǽ���
��2�����������ܻ�õ�CO��H2�����Ժϳɼ״��Ͷ����ѣ�CH3OCH3���������������ʣ�������1��1����Ӧ����ԭ�������ʴ�100%���ϳɵ����ʿ����� ��
a������ b���״�c����ȩ d������
��3���״���һ����Ҫ�Ļ���ԭ�ϣ���ҵ�Ϻϳɼ״��ķ�Ӧ��CO��g��+2H2��g��CH3OH��g������H=-90.8kJ?mol-1�������¶ȡ��ݻ���ͬ��3���ܱ������У�����ͬ��ʽͶ�뷴Ӧ����ֺ��¡����ݣ���÷�Ӧ�ﵽƽ��ʱ���й��������£�
����˵����ȷ���� ��
a��c1=c2 b.2Q1=Q3 c����2+��3��100%
��4����һ���¶Ⱥ�ѹǿ�£�CO��H2���ϳɶ����ѵķ�ӦΪ��3H2��g��+3CO��g��?CH3OCH3��g��+CO2��g��
����һ����ɱ���ܱ������г���3mol H2��3mol CO��1mol CH3OCH3��1mol CO2����һ��ʱ��ﵽƽ�⣬�����ƽ��ʱ��������ܶ���ͬ��ͬѹ����ʼʱ��1.6�����ٷ�Ӧ��ʼʱ�����淴Ӧ���ʵĴ�С��v������ v���棩�����������������=������������ ��ƽ��ʱn��CH3OCH3��= mol����ͼ2Ϊ��ɫ��Դ��ֱ�Ӷ�����ȼ�ϵ�ء��Ĺ���ԭ��ʾ��ͼ��b�缫�� ����a�缫�ķ�ӦʽΪ ��

��1�������й�˵����ȷ����
a���������ܣ�������������Դ��̫����
b������ά��ˮ���õ��Ҵ���ȼ������������������
c���������ѽ��õ����͡����͵����ڴ�����
d����ֲ��ո˵��������ͻ�õ���������Ҫ�ɷ��Ǽ���
��2�����������ܻ�õ�CO��H2�����Ժϳɼ״��Ͷ����ѣ�CH3OCH3���������������ʣ�������1��1����Ӧ����ԭ�������ʴ�100%���ϳɵ����ʿ�����
a������ b���״�c����ȩ d������
��3���״���һ����Ҫ�Ļ���ԭ�ϣ���ҵ�Ϻϳɼ״��ķ�Ӧ��CO��g��+2H2��g��CH3OH��g������H=-90.8kJ?mol-1�������¶ȡ��ݻ���ͬ��3���ܱ������У�����ͬ��ʽͶ�뷴Ӧ����ֺ��¡����ݣ���÷�Ӧ�ﵽƽ��ʱ���й��������£�
| ���� | �� | �� | �� |
| ��Ӧ��Ͷ���� | 1molCO��2molH2 | 1mol CH3OH | 2molCO��4molH2 |
| CH3OH��Ũ�ȣ�mol/L�� | c1 | c2 | c3 |
| ��Ӧ�������仯 | �ų�Q1 kJ | ����Q2 kJ | �ų�Q3 kJ |
| ��Ӧ��ת���� | �� 1 | �� 2 | �� 3 |
a��c1=c2 b.2Q1=Q3 c����2+��3��100%
��4����һ���¶Ⱥ�ѹǿ�£�CO��H2���ϳɶ����ѵķ�ӦΪ��3H2��g��+3CO��g��?CH3OCH3��g��+CO2��g��
����һ����ɱ���ܱ������г���3mol H2��3mol CO��1mol CH3OCH3��1mol CO2����һ��ʱ��ﵽƽ�⣬�����ƽ��ʱ��������ܶ���ͬ��ͬѹ����ʼʱ��1.6�����ٷ�Ӧ��ʼʱ�����淴Ӧ���ʵĴ�С��v������
���㣺��ѧƽ��ļ���,����������ת����ʽ
ר�⣺
��������1��a������������Դ��̫���ܣ�b���Ҵ���Դ����ά�أ�c���������ָ�ɶ���������ɵ����ʣ�d����������Ҫ�ɷ��Ǽ��飻
��2�����������غ㶨�ɺͻ��Ϸ�Ӧ�Ķ�����ص��жϣ���Ҫ�����÷�Ӧǰ��ԭ�ӵ�����䣬����Ŀ�ر��ֲ�������жϣ�
��3��a�������Ƿ��γɵ�Чƽ����⣻b������ѹǿƽ���������С�ķ����ƶ���c�������ת���ʣ�
��4���ٷ�Ӧǰ���������������䣻���ƽ��ʱ��������ܶ���ͬ��ͬѹ����ʼʱ��1.6����˵�����ʵ����ڼ��٣��жϳ���ʼ������У��ò���������ʽ��3H2��g��+3CO��g��?CH3OCH3��g��+CO2��g��������4mol���⣻
�ڸ���ʧ���ӣ������õ��ӣ���CH3��2O-12e-+3H2O=2CO2+12H+��
��2�����������غ㶨�ɺͻ��Ϸ�Ӧ�Ķ�����ص��жϣ���Ҫ�����÷�Ӧǰ��ԭ�ӵ�����䣬����Ŀ�ر��ֲ�������жϣ�
��3��a�������Ƿ��γɵ�Чƽ����⣻b������ѹǿƽ���������С�ķ����ƶ���c�������ת���ʣ�
��4���ٷ�Ӧǰ���������������䣻���ƽ��ʱ��������ܶ���ͬ��ͬѹ����ʼʱ��1.6����˵�����ʵ����ڼ��٣��жϳ���ʼ������У��ò���������ʽ��3H2��g��+3CO��g��?CH3OCH3��g��+CO2��g��������4mol���⣻
�ڸ���ʧ���ӣ������õ��ӣ���CH3��2O-12e-+3H2O=2CO2+12H+��
���
�⣺��1��a�������ʱ���������Դ��̫���ܣ���a��ȷ��
b����ά��ˮ���õ��Ҵ���Դ����ά�أ������������ܣ���b��ȷ��
c�����͡����͵����ڻ�����c����
d����������Ҫ�ɷ��Ǽ��飬��d��ȷ��
��ѡabd��
��2��������ɫ��ѧ����ʵ�����ŷţ�����Ӧ���е�ԭ�������ʴﵽ100%��Ҳ����˵��Ӧ����һ���ǻ��Ϸ�Ӧ���ҷ�Ӧ������еĸ�ԭ����Ŀ�Ȳ��䣬CO��H2��һ�������°��ղ�ͬ�ı�����Ӧ���ɼٶ���Ӧʱ�Ļ�ѧ������֮��Ϊ1��1����ֻҪ��ѡ���еĻ�ѧʽ�ܻ�Ϊ��ʽ��CO��n��H2��n��������ȷ�ģ������ǻ����״���CH4O���ɱ�Ϊ��CO��1��H2��2����ȩ��CH2O���ɱ�Ϊ��CO��1��H2��1�����ᣨC2H4O2���ɱ�Ϊ��CO��2��H2��2���ʴ�Ϊ��cd��
��3��a��1molCO��2molH2��1mol CH3OH�ĵ�Ч�ģ�ƽ��ʱCH3OH��Ũ��c1=c2����a��ȷ��
b�����ķ�Ӧ��Ͷ�����ǼĶ������ݻ���ͬ��ѹǿ�ȼ״�ƽ�����ҽ��У��ų�������2Q1��Q3����b����
C�������ת���ʣ���c����
�ʴ�Ϊ��a��
��4���ٷ�Ӧǰ���������������䣬ͬ�¡�ͬѹ�£��ﵽƽ��ʱ�������ܶ��������������С��ƽ��ʱ��������ܶ���ͬ��ͬѹ����ʼʱ��1.6�����������ʵ�����Ϊԭ�ȵ�0.625���������ʵ���=8��0.625=5mol����Ӧǰ�������3mol��˵����ʼ������Ӧ���У�v��������v���棩���跴Ӧ���ɼ���Xmol��
3H2��g��+3CO��g��?CH3OCH3��g��+CO2��g��������4mol����
1 4
x 3
=
��ã�x=0.75mol
ƽ��ʱ��n��CH3OCH3��=1+0.75=1.75mol��
�ʴ�Ϊ��������Ӧǰ���������������䣬ͬ�¡�ͬѹ�£��ﵽƽ��ʱ�������ܶ��������������С��ƽ�����ƣ� 1.75��
�������õ��ӣ����ϼ۽��ͣ���b�缫����������ͼ֪��a�缫��������ˮ��Ӧ���ɶ�����̼�������ӣ���a�缫�ķ�ӦʽΪ��CH3��2O-12e-+3H2O=2CO2+12H+��
�ʴ�Ϊ��������CH3��2O-12e-+3H2O=2CO2+12H+��
b����ά��ˮ���õ��Ҵ���Դ����ά�أ������������ܣ���b��ȷ��
c�����͡����͵����ڻ�����c����
d����������Ҫ�ɷ��Ǽ��飬��d��ȷ��
��ѡabd��
��2��������ɫ��ѧ����ʵ�����ŷţ�����Ӧ���е�ԭ�������ʴﵽ100%��Ҳ����˵��Ӧ����һ���ǻ��Ϸ�Ӧ���ҷ�Ӧ������еĸ�ԭ����Ŀ�Ȳ��䣬CO��H2��һ�������°��ղ�ͬ�ı�����Ӧ���ɼٶ���Ӧʱ�Ļ�ѧ������֮��Ϊ1��1����ֻҪ��ѡ���еĻ�ѧʽ�ܻ�Ϊ��ʽ��CO��n��H2��n��������ȷ�ģ������ǻ����״���CH4O���ɱ�Ϊ��CO��1��H2��2����ȩ��CH2O���ɱ�Ϊ��CO��1��H2��1�����ᣨC2H4O2���ɱ�Ϊ��CO��2��H2��2���ʴ�Ϊ��cd��
��3��a��1molCO��2molH2��1mol CH3OH�ĵ�Ч�ģ�ƽ��ʱCH3OH��Ũ��c1=c2����a��ȷ��
b�����ķ�Ӧ��Ͷ�����ǼĶ������ݻ���ͬ��ѹǿ�ȼ״�ƽ�����ҽ��У��ų�������2Q1��Q3����b����
C�������ת���ʣ���c����
�ʴ�Ϊ��a��
��4���ٷ�Ӧǰ���������������䣬ͬ�¡�ͬѹ�£��ﵽƽ��ʱ�������ܶ��������������С��ƽ��ʱ��������ܶ���ͬ��ͬѹ����ʼʱ��1.6�����������ʵ�����Ϊԭ�ȵ�0.625���������ʵ���=8��0.625=5mol����Ӧǰ�������3mol��˵����ʼ������Ӧ���У�v��������v���棩���跴Ӧ���ɼ���Xmol��
3H2��g��+3CO��g��?CH3OCH3��g��+CO2��g��������4mol����
1 4
x 3
| 1 |
| x |
| 4 |
| 3 |
ƽ��ʱ��n��CH3OCH3��=1+0.75=1.75mol��
�ʴ�Ϊ��������Ӧǰ���������������䣬ͬ�¡�ͬѹ�£��ﵽƽ��ʱ�������ܶ��������������С��ƽ�����ƣ� 1.75��
�������õ��ӣ����ϼ۽��ͣ���b�缫����������ͼ֪��a�缫��������ˮ��Ӧ���ɶ�����̼�������ӣ���a�缫�ķ�ӦʽΪ��CH3��2O-12e-+3H2O=2CO2+12H+��
�ʴ�Ϊ��������CH3��2O-12e-+3H2O=2CO2+12H+��
���������⿼���Чƽ�⡢��ѧƽ��ļ��㡢��ѧ��Դ���͵�صȣ���Ŀ�ѶȽϴ�ע�⻯ѧƽ��ļ����DZ�����ѵ㣬ע���������Ӧ�ã�

��ϰ��ϵ�д�
�����Ŀ
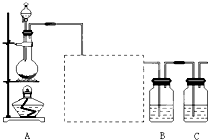
ijѧ������ʵ��������·����ֱ���ϴ����������Ӧ�á��������ܡ����ɵ��ǣ�������
| A����ϡ������ϴ����������Ӧ���Թ� |
| B����ϡ������ϴ��������طֽ�ʵ����Թ� |
| C��������������Һ��ϴʢ��������Թ� |
| D�������Ȼ�̼��ϴ�������������ձ� |
���е��뷽��ʽ������ǣ�������
| A��CaCl2=Ca2++2Cl- |
| B��Ba��OH��2=Ba2++2OH- |
| C��HNO3=H++NO3- |
| D��NaHCO3=Na++H++CO32- |
���ά����Ϊ��20������ΰ��ķ���֮һ����ʹ��Ϣ���ٹ�·��ȫ��Ѹ�ͷ�չ�����ά����Ҫ�ɷ��ǣ�������
| A��CaCO3 |
| B��Si |
| C��SiO2 |
| D��SiC |