��Ŀ����
ij���Ͻ�Ӳ�����к���þ��ͭ���裬Ϊ�˲ⶨ�úϽ������ĺ���������һ�ֺܲ����õķǽ������ʣ��������HF��������ᷢ����Ӧ��������������²�����飺
(1)ȡ��Ʒag����ȡʱʹ�õ���������Ϊ________��
(2)����Ʒ����������ϡ�����У����ˣ���Һ����Ҫ���е���������__________�������к���________�����ܽ����ʱʹ�õ�������______________��
(3)����Һ�м������������������Һ�����ˣ�д���ò��������йط�Ӧ�Ļ�ѧ����ʽ______________��
(4)�ڵ�(3)������Һ��ͨ������CO2�����ˣ�������������ˮϴ�����κ�ɲ��������������ټ���Ϊֹ����ȴ�����������Ϊbg���������Ʒ��������������_________��
(1)ȡ��Ʒag����ȡʱʹ�õ���������Ϊ________��
(2)����Ʒ����������ϡ�����У����ˣ���Һ����Ҫ���е���������__________�������к���________�����ܽ����ʱʹ�õ�������______________��
(3)����Һ�м������������������Һ�����ˣ�д���ò��������йط�Ӧ�Ļ�ѧ����ʽ______________��
(4)�ڵ�(3)������Һ��ͨ������CO2�����ˣ�������������ˮϴ�����κ�ɲ��������������ټ���Ϊֹ����ȴ�����������Ϊbg���������Ʒ��������������_________��
(1)������ƽ
(2)Mg2+��Al3+��ͭ�裻©�������������ձ�������̨������Ȧ������ֽ
(3)MgCl2+2NaOH=Mg(OH)2��+2NaCl��AlCl3+ 4NaOH=NaAlO2+3NaCl+2H2O
(2)Mg2+��Al3+��ͭ�裻©�������������ձ�������̨������Ȧ������ֽ
(3)MgCl2+2NaOH=Mg(OH)2��+2NaCl��AlCl3+ 4NaOH=NaAlO2+3NaCl+2H2O
(4)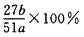

��ϰ��ϵ�д�
 �������Ͽ�ʱͬ��ѵ��ϵ�д�
�������Ͽ�ʱͬ��ѵ��ϵ�д� �������ͬ����ϰϵ�д�
�������ͬ����ϰϵ�д�
�����Ŀ