��Ŀ����
15�������ӵ�����ԼΪ6.02��1023mol-1��������������ȷ���ǣ�������| A�� | ��״���£�2.24L����Լ����3.612��1023��̼ԭ�� | |
| B�� | ���³�ѹ�£������ͳ����Ļ����16g��Լ����6.02��1023����ԭ�� | |
| C�� | 25��ʱ��1 L pH=13������������Һ��Լ����6.02��l023������������ | |
| D�� | 0.5mol CH4��Լ����3.01��1024������ |
���� A������Ħ�����ʹ�ö���Ϊ���壻
B�������ͳ�����������ԭ�ӹ��ɣ�����n=$\frac{m}{M}$������
C.25��ʱ��1 L pH=13������������Һ������������Ũ��Ϊ��0.1mol/L������N=cVNA�������������Ӹ�����
D.1��������Ӻ���10�����ӣ�
��� �⣺A������£�����Һ̬������ʹ������Ħ���������A����
B�����³�ѹ�£������ͳ����Ļ����16g��Լ������ԭ�Ӹ�����$\frac{16g}{16g/mol}$��6.02��1023mol-1=6.02��1023����B��ȷ��
C.25��ʱ��1 L pH=13������������Һ������������Ũ��Ϊ��0.1mol/L���������������Ӹ���Ϊ��0.1mol/L��1L��6.02��1023mol-1=6.02��1022����C����
D.0.5mol CH4��Լ����0.5mol��10��6.02��1023mol-1=3.01��1024�����ӣ���D��ȷ��
��ѡ��BD��
���� ���⿼���˰���٤��������Ӧ������㣬��ȷ�����ʵ���Ϊ���ļ��㹫ʽ����Ϥ����Ħ�����ʹ�������ǽ���ؼ�����Ŀ�ѶȲ���

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
8�����й��̲��漰������ԭ��Ӧ���ǣ�������
| A�� | �����̵� | B�� | ��ҵ��Ư�۾� | ||
| C�� | ��SO2Ư��ֽ�� | D�� | �칬һ������Na2O2���� |
3����N0Ϊ�����ӵ�������ֵ�������й���������ȷ���ǣ�������
| A�� | 0.5 Ħ1��3-����ϩ�����к��й��ۼ���Ϊ5.5N0 | |
| B�� | 1 mol����-CH3�������ĵ�������Ϊ9N0 | |
| C�� | 1 mol-OH��1 mol OH-������������Ϊ9N0 | |
| D�� | 1 mol̼�����ӣ�CH3+�������ĵ�������Ϊ8N0 |
10����������ɫ����Һ�У��ܴ���������������ǣ�������
| A�� | Cu2+��Na+��SO42-��Cl- | B�� | Ca2+��NH4+��HCO3-��NO3- | ||
| C�� | NO3-��Fe2+��Ca2+��H+ | D�� | Mg2+��Na+��OH-��NO3- |
7���ڱ�״���£���A����V L ��Ħ��������M g•mol-1������0.5Lˮ�У�������Һ���ܶ�Ϊa g•cm-3�������Һ�����ʵ���Ũ��Ϊ��������
| A�� | $\frac{1000Va}{MV+1120}$ mol•L -1 | B�� | $\frac{1000Va}{MV+22400}$ mol•L -1 | ||
| C�� | $\frac{1000Va}{MV+11200}$ mol•L -1 | D�� | $\frac{1000V•M}{2240��V+0.5��a}$ mol•L -1 |
4��NA��ʾ�����ӵ�������ֵ������˵���У���ȷ���ǣ�������
| A�� | �����£�4.2g��ϩ�ͱ�ϩ�Ļ�������к��еķǼ��Լ�����Ϊ0.6NA | |
| B�� | ��⾫��ͭʱ������·��ת��NA�����ӣ������ܽ�32 gͭ | |
| C�� | �ں���CH3COO-Ϊ1.0 mol•L-1�Ĵ�����Һ�У�H+��Ŀ�Դ���NA | |
| D�� | V L a mol•L-1���Ȼ�����Һ�У���Fe3+����ĿΪ NA����Cl-����Ŀ����3NA |
5�����ݻ��̶����ܱ������д������·�Ӧ��A��g��+3B��g��?2C��g����H��0��ij�о�С���о�������������ʱ���ı�ijһ������������Ӧ��Ӱ�죬������ʵ�������������й�ϵͼ��

�����ж���ȷ���ǣ�������

�����ж���ȷ���ǣ�������
| A�� | ͼI�о������¶ȶԷ�Ӧ��Ӱ�죬���ҵ��¶Ƚϸ� | |
| B�� | ͼ���о�����ѹǿ�Է�Ӧ��Ӱ�죬�Ҽ�ѹǿ�ϸ� | |
| C�� | ͼIII�о������¶ȶԷ�Ӧ��Ӱ�죬���ҵ��¶Ƚϵ� | |
| D�� | ͼIV�о����Dz�ͬ�����Է�Ӧ��Ӱ�죬�Ҽ�ʹ�õĴ���Ч�ʽϵ� |
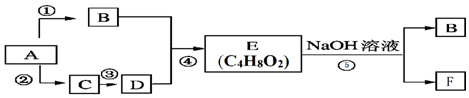

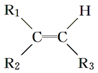 $\stackrel{����}{��}$
$\stackrel{����}{��}$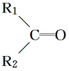 +R3COOH
+R3COOH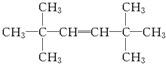 G��
G�� ��
��