��Ŀ����
��֪A��B��C����ѧ��ѧ���������ֻ��������֮��������ת����ϵ��A
B
C
��1����A��B��C����ɫ��Ӧ���ʻ�ɫ������ˮ��Һ���Լ��ԣ���A��B��C�е������ӵĽṹʾ��ͼΪ ��D�ĽṹʽΪ ��
��2����A��B��C�������ʺ���ͬһԪ��X����B�������ԣ���
��A��C��Ӧ�����ӷ���ʽΪ ��
����֪1g X�ĵ�����MnO2��ȫ��Ӧ���ų�a kJ������д���÷�Ӧ���Ȼ�ѧ����ʽ ��
�۹�ҵ�ϳ���X��������Ŀ�ʯ�м��뽹̿����Cl2��Ӧ��X���Ȼ���ɱ�ʾΪX2O3+C+Cl2
XCl3+M�������ʵ��ȷ������M�ijɷ� ��
��3����A��C�dz������õĵ�ζƷ����
��A��B�ķ�Ӧ����ʽΪ ��
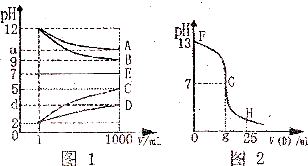
��ijͬѧ�����һ����A����Ϊȼ�ϵĵ�أ����øõ�ص��200mLһ��Ũ��NaCl��CuSO4�����Һ����װ����ͼ1��д������A���ʲ���ĵ缫��Ӧʽ �������Ϣ���������������������ʱ��仯�Ĺ�ϵ������ͼ2��ʾ����������ѻ���ɱ�״���µ����������t2ʱ������Һ��pHԼΪ ��
| D |
| D |
��1����A��B��C����ɫ��Ӧ���ʻ�ɫ������ˮ��Һ���Լ��ԣ���A��B��C�е������ӵĽṹʾ��ͼΪ
��2����A��B��C�������ʺ���ͬһԪ��X����B�������ԣ���
��A��C��Ӧ�����ӷ���ʽΪ
����֪1g X�ĵ�����MnO2��ȫ��Ӧ���ų�a kJ������д���÷�Ӧ���Ȼ�ѧ����ʽ
�۹�ҵ�ϳ���X��������Ŀ�ʯ�м��뽹̿����Cl2��Ӧ��X���Ȼ���ɱ�ʾΪX2O3+C+Cl2
| �� |
��3����A��C�dz������õĵ�ζƷ����
��A��B�ķ�Ӧ����ʽΪ
��ijͬѧ�����һ����A����Ϊȼ�ϵĵ�أ����øõ�ص��200mLһ��Ũ��NaCl��CuSO4�����Һ����װ����ͼ1��д������A���ʲ���ĵ缫��Ӧʽ

���㣺������ƶ�,ԭ��غ͵��صĹ���ԭ��
ר�⣺�ƶ���,�绯ѧר��
��������1����A��B��C����ɫ��Ӧ���ʻ�ɫ������ˮ��Һ���Լ��ԣ�A��B��C����ѧ��ѧ���������ֻ������ת����ϵ��֪AΪNaOH��BΪNa2CO3��CΪNaHCO3��DΪCO2��
��2����A��B��C�������ʺ���ͬһԪ��X����B�������ԣ�BӦΪAl��OH��3��XΪAl��A��C��Ӧ����ΪAl3+��[Al��OH��4]-��[Al��OH��4]-��Al3+��
��3����A��C�dz������õĵ�ζƷ��Ӧ�ֱ�ΪCH3CH2OH��CH3COOH��
��2����A��B��C�������ʺ���ͬһԪ��X����B�������ԣ�BӦΪAl��OH��3��XΪAl��A��C��Ӧ����ΪAl3+��[Al��OH��4]-��[Al��OH��4]-��Al3+��
��3����A��C�dz������õĵ�ζƷ��Ӧ�ֱ�ΪCH3CH2OH��CH3COOH��
���
�⣺��1����A��B��C����ɫ��Ӧ���ʻ�ɫ������ˮ��Һ���Լ��ԣ�A��B��C����ѧ��ѧ���������ֻ������ת����ϵ��֪AΪNaOH��BΪNa2CO3��CΪNaHCO3��DΪCO2����A��B��C�е������ӵĽṹʾ��ͼΪ ��������̼�ĽṹʽΪO=C=O��
��������̼�ĽṹʽΪO=C=O��
�ʴ�Ϊ�� �� O=C=O��
�� O=C=O��
��2����A��B��C�������ʺ���ͬһԪ��X����B�������ԣ�BӦΪAl��OH��3��XΪAl��A��C��Ӧ����ΪAl3+��[Al��OH��4]-��[Al��OH��4]-��Al3+��
��A��C��Ӧ�����ӷ���ʽΪAl3++4OH-=[Al��OH��4]-��[Al��OH��4]-+4H+=Al3++4H2O��
�ʴ�Ϊ��Al3++4OH-=[Al��OH��4]-��[Al��OH��4]-+4H+=Al3++4H2O��
����֪1g Al�ĵ�����MnO2��ȫ��Ӧ���ų�a kJ��������Ӧ�ķ���ʽΪ4Al��s��+3MnO2��s��=2Al2O3��s��+3Mn��s������ų�������Ϊ4��27��akJ=108kJ��
д���÷�Ӧ���Ȼ�ѧ����ʽΪ4Al��s��+3MnO2��s��=2Al2O3��s��+3Mn��s����H=-108 a kJ?mol-1��
�ʴ�Ϊ��4Al��s��+3MnO2��s��=2Al2O3��s��+3Mn��s����H=-108 a kJ?mol-1��
�۹�ҵ�ϳ���X��������Ŀ�ʯ�м��뽹̿����Cl2��Ӧ��X���Ȼ���ɱ�ʾΪX2O3+C+Cl2
XCl3+M����M����ΪCO2��CO������CO2�����ó���ʯ��ˮ�������ȵ�CuO��ĩ����CO��
�ʴ�Ϊ����������������ͨ������ʯ��ˮ�����ȵ�CuO��ĩ������Һ����ǣ���X�д���CO2�����ɫCuO��ĩ��ɺ�ɫ��ĩ����X�д���CO��
��3����A��C�dz������õĵ�ζƷ��Ӧ�ֱ�ΪCH3CH2OH��CH3COOH��
��A��B�ķ�Ӧ����ʽΪ2CH3CH2OH+O2
2CH3CHO+2H2O��
�ʴ�Ϊ��2CH3CH2OH+O2
2CH3CHO+2H2O��
���Ҵ�ȼ�ϵ���У��Ҵ����������缫����ʽΪ2CH3CH2OH+O2
2CH3CHO+2H2O��
ii��ʯī�缫Ϊ���������缫Ϊ��������ʼCl-�������ŵ���������������Cu2+�ŵ�����Cu����ͼ2�Т��ʾ�������������壬���ʾ�������������壬t1ǰ�缫��ӦʽΪ������2Cl--4e-=Cl2����t1��t2�缫��ӦʽΪ������4OH--4e-=O2��+2H2O��t2���缫��ӦʽΪ������4OH--4e-=O2��+2H2O��������t1��ʯī�缫�ϵĵ缫��ӦʽΪ4OH--4e-=O2��+2H2O��
t1��t2�缫��ӦʽΪ������4OH--4e-=O2��+2H2O������2Cu2++4e-=2Cu��t2���缫��ӦʽΪ������4OH--4e-=O2��+2H2O������4H++4e-=2H2��������t1��t2 ����������������t1��t2��������������=0.336L-0.224L=0.112L������������112mL������Ҫn��OH-��=
=0.02mol��ˮ����������ʵ����������Ӻ����������ӣ�����n��H+��=0.02mol��c��H+��=
=0.1mol/L��������Һ��pH=1��
�ʴ�Ϊ��C2H5OH-12e-+16OH-=2CO32-+11H2O�� 1��
 ��������̼�ĽṹʽΪO=C=O��
��������̼�ĽṹʽΪO=C=O���ʴ�Ϊ��
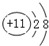 �� O=C=O��
�� O=C=O����2����A��B��C�������ʺ���ͬһԪ��X����B�������ԣ�BӦΪAl��OH��3��XΪAl��A��C��Ӧ����ΪAl3+��[Al��OH��4]-��[Al��OH��4]-��Al3+��
��A��C��Ӧ�����ӷ���ʽΪAl3++4OH-=[Al��OH��4]-��[Al��OH��4]-+4H+=Al3++4H2O��
�ʴ�Ϊ��Al3++4OH-=[Al��OH��4]-��[Al��OH��4]-+4H+=Al3++4H2O��
����֪1g Al�ĵ�����MnO2��ȫ��Ӧ���ų�a kJ��������Ӧ�ķ���ʽΪ4Al��s��+3MnO2��s��=2Al2O3��s��+3Mn��s������ų�������Ϊ4��27��akJ=108kJ��
д���÷�Ӧ���Ȼ�ѧ����ʽΪ4Al��s��+3MnO2��s��=2Al2O3��s��+3Mn��s����H=-108 a kJ?mol-1��
�ʴ�Ϊ��4Al��s��+3MnO2��s��=2Al2O3��s��+3Mn��s����H=-108 a kJ?mol-1��
�۹�ҵ�ϳ���X��������Ŀ�ʯ�м��뽹̿����Cl2��Ӧ��X���Ȼ���ɱ�ʾΪX2O3+C+Cl2
| �� |
�ʴ�Ϊ����������������ͨ������ʯ��ˮ�����ȵ�CuO��ĩ������Һ����ǣ���X�д���CO2�����ɫCuO��ĩ��ɺ�ɫ��ĩ����X�д���CO��
��3����A��C�dz������õĵ�ζƷ��Ӧ�ֱ�ΪCH3CH2OH��CH3COOH��
��A��B�ķ�Ӧ����ʽΪ2CH3CH2OH+O2
| Cu |
| �� |
�ʴ�Ϊ��2CH3CH2OH+O2
| Cu |
| �� |
���Ҵ�ȼ�ϵ���У��Ҵ����������缫����ʽΪ2CH3CH2OH+O2
| Cu |
| �� |
ii��ʯī�缫Ϊ���������缫Ϊ��������ʼCl-�������ŵ���������������Cu2+�ŵ�����Cu����ͼ2�Т��ʾ�������������壬���ʾ�������������壬t1ǰ�缫��ӦʽΪ������2Cl--4e-=Cl2����t1��t2�缫��ӦʽΪ������4OH--4e-=O2��+2H2O��t2���缫��ӦʽΪ������4OH--4e-=O2��+2H2O��������t1��ʯī�缫�ϵĵ缫��ӦʽΪ4OH--4e-=O2��+2H2O��
t1��t2�缫��ӦʽΪ������4OH--4e-=O2��+2H2O������2Cu2++4e-=2Cu��t2���缫��ӦʽΪ������4OH--4e-=O2��+2H2O������4H++4e-=2H2��������t1��t2 ����������������t1��t2��������������=0.336L-0.224L=0.112L������������112mL������Ҫn��OH-��=
| 0.112L��4mol |
| 22.4L |
| 0.02mol |
| 0.2L |
�ʴ�Ϊ��C2H5OH-12e-+16OH-=2CO32-+11H2O�� 1��
�����������ۺϿ���������ƶϣ�Ϊ�߿��������ͣ�������ѧ���ķ��������ͼ��������Ŀ��飬ע�����������ʵ����ʣ����յ��Ĺ���ԭ���Լ����㷽�����Ѷ� �еȣ�

��ϰ��ϵ�д�
�����Ŀ
�л����ұ� ��һ��ȡ�����ͬ���칹���У�������
��һ��ȡ�����ͬ���칹���У�������
 ��һ��ȡ�����ͬ���칹���У�������
��һ��ȡ�����ͬ���칹���У�������| A��5�� | B��6�� | C��7�� | D��8�� |
���й���SiO2����˵������ȷ���ǣ�������
| A��60gSiO2�к�Si-O���ĸ���Ϊ2NA |
| B�����������ѻ������ӽ��ܶѻ�ԭ����ԭ���ǹ��ۼ����б����Ժͷ����� |
| C��SiO2������۷е�ϸɱ��������ھ������Ͳ�ͬ |
| D�����ԭ��������ĸ���ԭ�ӹ���һ���������� |