��Ŀ����
T��ʱ����1 L���ܱ������г���2 mol CO2��6mol H2��һ�������·�����Ӧ��
CO2(g)+3H2(g)  CH3OH(g)+H2O(g)
��H=��49.0kJ/mol
CH3OH(g)+H2O(g)
��H=��49.0kJ/mol
���H2��CH3OH(g)��Ũ����ʱ��仯����ͼ��ʾ������˵������ȷ���� �� ��
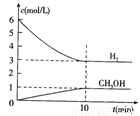
A��0~10min��v(H2)=0.3mol/(L��min)
B��T��ʱ��ƽ�ⳣ��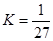 ��CO2��H2��ת�������
��CO2��H2��ת�������
C��T��ʱ��������Ӧ����64g CH3OH���ɣ�ͬʱ�ų�98.0kJ������
D���ﵽƽ��������¶Ȼ��ٳ���CO2���壬���������H2��ת����
D
��������

T��ʱ����1 L���ܱ������г���2 mol CO2��6mol H2��һ�������·�����Ӧ��
CO2(g)+3H2(g)  CH3OH(g)+H2O(g) ��H=��49.0kJ/mol
CH3OH(g)+H2O(g) ��H=��49.0kJ/mol
���H2��CH3OH(g)��Ũ����ʱ��仯����ͼ��ʾ������˵������ȷ���� �� ��
| A��0~10min��v(H2)=0.3mol/(L��min) |
B��T��ʱ��ƽ�ⳣ��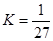 ��CO2��H2��ת������� ��CO2��H2��ת������� |
| C��T��ʱ��������Ӧ����64g CH3OH���ɣ�ͬʱ�ų�98.0kJ������ |
| D���ﵽƽ��������¶Ȼ��ٳ���CO2���壬���������H2��ת���� |
T ��ʱ����1 L���ܱ������г���2 mol CO2��6 mol H2��һ�������·�����Ӧ��CO2(g)��3H2(g) 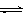 CH3OH(g)��H2O(g) ��H����49.0 kJ��mol-1���H2��CH3OH(g)��Ũ����ʱ��仯����ͼ��ʾ������˵������ȷ����:
CH3OH(g)��H2O(g) ��H����49.0 kJ��mol-1���H2��CH3OH(g)��Ũ����ʱ��仯����ͼ��ʾ������˵������ȷ����:
|
ʱ�� |
c ��H2����mol��L-1 |
c ��CH3OH����mol��L-1 |
v��������v ���棩�Ƚ� |
|
t0 |
6 |
0 |
�� |
|
t1 |
3 |
1 |
v������=v���棩 |
A��t0 ��t1ʱ������(H2)��3 /��t1 - t0�� mol��(L��min)-1
B�� t1ʱ���������¶Ȼ��ٳ���CO2���壬���������H2��ת����
C��t0ʱ��v��������v���棩
D��T ��ʱ��ƽ�ⳣ��K = 1/27��CO2��H2��ת�������
 CH3OH��g��+H2O��g�� ��H=-49.0 kJ/mol
CH3OH��g��+H2O��g�� ��H=-49.0 kJ/mol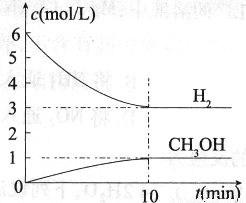
 ��H2��=0.3 mol/��L��min��
��H2��=0.3 mol/��L��min�� CH3OH(g)+H2O(g) ��H=��49.0kJ/mol
CH3OH(g)+H2O(g) ��H=��49.0kJ/mol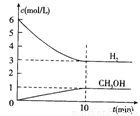
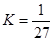 ��CO2��H2��ת�������
��CO2��H2��ת�������