��Ŀ����
7����ͬѧ���������������ø�����ر���Һ�ζ��ķ����ⶨijҩƷ����Ԫ�صĺ�������Ӧԭ��Ϊ5Fe2++8H++MnO4-�T5Fe3++Mn2++4H2O��ȷ������ҩƷ10.00g������ȫ�������Լ�2�У����Ƴ�1000mL��Һ��ȡ��20.00mL����0.020 0mol/L��KMnO4��Һ�ζ�����ȥKMnO4��Һ12.00mL��
��1����ʵ���е��Լ�2������C�����ţ���
A������ˮ B��Ũ���� C��ϡ���� D��ϡ����
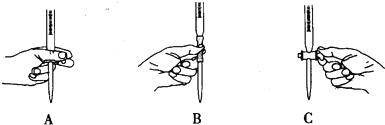
��2����ʵ��ζ������в����ζ��ܵ�ͼʾ��ȷ����A�����ţ���
��3����ͨ�����㣬˵����ҩƷ���������İٷֺ���Ϊ33.6%��
���� ��1��������ؿ����������ӣ�����������������ӣ���ѡ�������ữ��
��2��������ؾ���ǿ�����ԣ���������ѡ����ʽ�ζ��ܣ����ֿ��ƻ�����
��3�����5Fe2++8H++MnO4-�T5Fe3++Mn2++4H2O���㣮
��� �⣺��1��������ؿ����������ӣ�����������������ӣ��Լ�2Ϊ�ᣬ��Ϊ���ᣬ�ʴ�Ϊ��C��
��2��������ؾ���ǿ�����ԣ���������ѡ����ʽ�ζ��ܣ����ֿ��ƻ�����ֻ��A���ϣ��ʴ�Ϊ��A��
��3������5Fe2++8H++MnO4-�T5Fe3++Mn2++4H2O��n��Fe2+��=0.020 0 mol/L��12.00��10-3 L��5��$\frac{1000mL}{20.00mL}$=0.060 0mol��m��Fe2+��=0.0600 mol��56 g/mol=3.36g��������������$\frac{3.36g}{10.0g}$��100%=33.6%��
�ʴ�Ϊ��33.6%��
���� ���⿼�����ʺ����IJⶨ��Ϊ��Ƶ���㣬�������ʵ����ʡ��ζ�������������ʹ�á���Ϸ�Ӧ�ļ���Ϊ���Ĺؼ������ط�����ʵ�������Ŀ��飬ע��������ԭ��Ӧ��ʵ���е�Ӧ�ã���Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
12��������������ȷ���ǣ�������
| A�� | ����������������ɢϵ�ı��������ǣ���������ֱ����1-100nm֮�� | |
| B�� | һ���¶ȡ�ѹǿ�£��������������ӵĴ�С���� | |
| C�� | ������ԭ��Ӧ�ı����ǵ���ת�� | |
| D�� | ��1L2mol/L���������������������������������2NA |
13����NAΪ�����ӵ�������ֵ������˵����ȷ���ǣ�������
| A�� | ��״���£�22.4 L ���Ȼ�̼��CCl4�����е�̼ԭ����ĿΪNA | |
| B�� | CO2ͨ��Na2O2ʹ������5.6gʱ����Ӧ��ת�Ƶĵ�����Ϊ0.4 NA | |
| C�� | 0.2mol Fe��0.2 mol Cl2��Ӧʱʧ������Ϊ0.6NA | |
| D�� | ���³�ѹ�£�29.8 g KCl��NaClO�Ĺ��������������ӵ���ĿΪ0.4 NA |
10��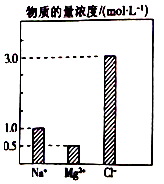 ��0.2L��NaCl��MgCl2��BaCl2��ɵĻ����Һ�У���������Ũ�ȴ�С��ͼ��ʾ�����ڸ���Һ�ɷ֣�����˵������ȷ���ǣ�������
��0.2L��NaCl��MgCl2��BaCl2��ɵĻ����Һ�У���������Ũ�ȴ�С��ͼ��ʾ�����ڸ���Һ�ɷ֣�����˵������ȷ���ǣ�������
 ��0.2L��NaCl��MgCl2��BaCl2��ɵĻ����Һ�У���������Ũ�ȴ�С��ͼ��ʾ�����ڸ���Һ�ɷ֣�����˵������ȷ���ǣ�������
��0.2L��NaCl��MgCl2��BaCl2��ɵĻ����Һ�У���������Ũ�ȴ�С��ͼ��ʾ�����ڸ���Һ�ɷ֣�����˵������ȷ���ǣ�������| A�� | NaCl�����ʵ���Ϊ0.2mol | |
| B�� | ����MgCl2������Ϊ9 g | |
| C�� | �û��Һ��BaCl2�����ʵ���Ϊ0.1mol | |
| D�� | ���û��Һ��ˮϡ�������Ϊ1 L��ϡ�ͺ���Һ�е�Ba2+���ʵ���Ũ��Ϊ0.1mol/L |
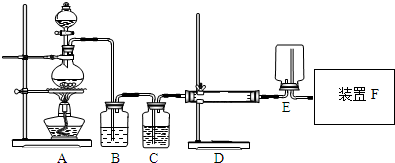
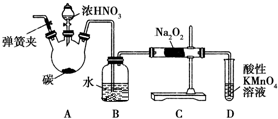 �������ƣ�NaNO2������Ϊ��ҵ�Σ���Ư�ס���Ƶȷ���Ӧ�ù㷺����ľ̿��Ũ���ᡢˮ��ͭΪԭ���Ʊ��������Ƶ�װ����ͼ��ʾ����֪�������£���2NO+Na2O2�T2NaNO2����3NaNO2+3HCl�T3NaCl+HNO3+2NO��+H2O�������������£�NO��NO2-������MnO4-��Ӧ����NO3-��Mn2+��
�������ƣ�NaNO2������Ϊ��ҵ�Σ���Ư�ס���Ƶȷ���Ӧ�ù㷺����ľ̿��Ũ���ᡢˮ��ͭΪԭ���Ʊ��������Ƶ�װ����ͼ��ʾ����֪�������£���2NO+Na2O2�T2NaNO2����3NaNO2+3HCl�T3NaCl+HNO3+2NO��+H2O�������������£�NO��NO2-������MnO4-��Ӧ����NO3-��Mn2+��

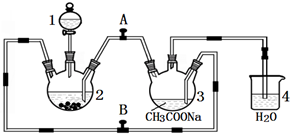 ���۸����ȶ������ױ����������������Ǹ�ˮ����{[Cr��CH3COO��2]2•2H2O����Է�������Ϊ376}��һ�����ɫ���壬��������ˮ���ѣ����������ᣬ�dz��õ��������ռ���ʵ��������п����CrCl3��Һ����������Һ������Ϊ��Ҫԭ���Ʊ������Ǹ�ˮ�����װ����ͼ��ʾ��
���۸����ȶ������ױ����������������Ǹ�ˮ����{[Cr��CH3COO��2]2•2H2O����Է�������Ϊ376}��һ�����ɫ���壬��������ˮ���ѣ����������ᣬ�dz��õ��������ռ���ʵ��������п����CrCl3��Һ����������Һ������Ϊ��Ҫԭ���Ʊ������Ǹ�ˮ�����װ����ͼ��ʾ��