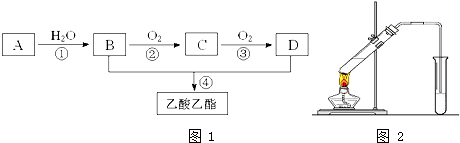
��Ŀ����
ʵ��������һ��NaCl��Na2CO3?10H2O��NaHCO3�Ļ���ijͬѧ�������ʵ�飬ͨ��������Ӧǰ��C��Dװ�������ı仯���ⶨ�û�����и���ֵ�����������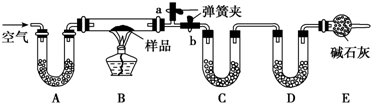
��1��ʵ��ʱ��B�з�����Ӧ�Ļ�ѧ����ʽΪ ��
��2��װ��C��D��ʢ�ŵ��Լ��ֱ�ΪC ��D ����ѡ�Լ�ΪŨ���ᡢ��ˮCaCl2����ʯ�ң���
��3��Eװ���е����������� �����ڸ�ʵ���е���Ҫ������ ��
��4������Aװ�û���ʢ��NaOH��Һ��ϴ��ƿ�����õ�NaCl������ ���ƫ�ߡ�����ƫ�͡�����Ӱ�족����
��5����Ӧǰ����Bװ����ͨ������ɼ���ʵ�������������� ��

��1��ʵ��ʱ��B�з�����Ӧ�Ļ�ѧ����ʽΪ
��2��װ��C��D��ʢ�ŵ��Լ��ֱ�ΪC
��3��Eװ���е�����������
��4������Aװ�û���ʢ��NaOH��Һ��ϴ��ƿ�����õ�NaCl������
��5����Ӧǰ����Bװ����ͨ������ɼ���ʵ��������������
���㣺̽�����ʵ���ɻ�������ʵĺ���
ר�⣺ʵ��̽�������ݴ�����,ʵ�������
������������ͨ����ʯ������CO2��H2O�����װ�ã��Գ���ų���Ӧ��B�в�����CO2��H2O����B�У�Na2CO3?10H2O��NaHCO3�����ֽ⣬������H2O��C�ܣ�ʢ��ˮCaCl2�����գ�CO2��D�ܣ�ʢ��ʯ�ң����գ�ͨ��C��D�ܵ��������ӿɼ���Na2CO3?10H2O��NaHCO3���������Ӷ�ȷ�����ɷֵ�������������һ��Ҫ��ֹ�������е�CO2��H2O����װ�ã�����Aװ�û���ʢNaOH��Һ��ϴ��ƿ����m��H2O�����ӣ�ʹNa2CO3?10H2O��NaHCO3�ĺ���ƫ�ߣ�NaCl�ĺ���ƫ�ͣ�
��1������ʱ��Na2CO3?10H2O��NaHCO3�ֽ⣻
��2��CDʢװ���壬��������ˮ��������̼���壻
��3���������ǣ����Σ�����ܣ���ʯ�������տ����еĶ�����̼��ˮ������
��4��������ͨ������������Һ�л���߲���ˮ�֣����¶�ʵ����ɸ��ţ�
��5����Bװ����ͨ���������װ���ڵ�ˮ�Ͷ�����̼�ų��������ܽ���Cװ�ã�
��1������ʱ��Na2CO3?10H2O��NaHCO3�ֽ⣻
��2��CDʢװ���壬��������ˮ��������̼���壻
��3���������ǣ����Σ�����ܣ���ʯ�������տ����еĶ�����̼��ˮ������
��4��������ͨ������������Һ�л���߲���ˮ�֣����¶�ʵ����ɸ��ţ�
��5����Bװ����ͨ���������װ���ڵ�ˮ�Ͷ�����̼�ų��������ܽ���Cװ�ã�
���
�⣺������ͨ����ʯ������CO2��H2O�����װ�ã��Գ���ų���Ӧ��B�в�����CO2��H2O����B�У�Na2CO3?10H2O��NaHCO3�����ֽ⣬������H2O��C�ܣ�ʢ��ˮCaCl2�����գ�CO2��D�ܣ�ʢ��ʯ�ң����գ�ͨ��C��D�ܵ��������ӿɼ���Na2CO3?10H2O��NaHCO3���������Ӷ�ȷ�����ɷֵ�������������һ��Ҫ��ֹ�������е�CO2��H2O����װ�ã�����Aװ�û���ʢNaOH��Һ��ϴ��ƿ����m��H2O�����ӣ�ʹNa2CO3?10H2O��NaHCO3�ĺ���ƫ�ߣ�NaCl�ĺ���ƫ�ͣ�
��1��B��Na2CO3?10H2O��NaHCO3�����ֽ⣬��Ӧ�ķ���ʽ�ֱ�ΪNa2CO3?10H2O
Na2CO3+10 H2O��2NaHCO3=Na2CO3+CO2��+H2O��
�ʴ�Ϊ��Na2CO3?10H2O
Na2CO3+10 H2O��2NaHCO3=Na2CO3+CO2��+H2O��
��2��Na2CO3?10H2O��NaHCO3�����ֽ⣬������H2O��C�ܣ�ʢ��ˮCaCl2�����գ�CO2��D�ܣ�ʢ��ʯ�ң����գ�ͨ��C��D�ܵ��������ӿɼ���Na2CO3?10H2O��NaHCO3��������
�ʴ�Ϊ����ˮCaCl2����ʯ�ң�
��3��EΪ����ܣ�ʢ�м�ʯ�ң��ɷ�ֹ�������е�CO2��H2O����װ�ã��ʴ�Ϊ������ܣ���ֹ�����е�H2O��CO2����D�У�
��4������Aװ�û���ʢ��NaOH��Һ��ϴ��ƿ����m��H2O�����ӣ�ʹNa2CO3?10H2O��NaHCO3�ĺ���ƫ�ߣ�NaCl�ĺ���ƫ�ͣ��ʴ�Ϊ��ƫ�ͣ�
��5����Ӧǰ����Bװ����ͨ�������Ӧ�رյ��ɼ�b�����ɼ�a����װ���ڵ�ˮ�Ͷ�����̼�ų��������ܽ���Cװ���У�
�ʴ�Ϊ���رյ��ɼ�b�����ɼ�a����װ����ͨ�������
��1��B��Na2CO3?10H2O��NaHCO3�����ֽ⣬��Ӧ�ķ���ʽ�ֱ�ΪNa2CO3?10H2O
| ||
�ʴ�Ϊ��Na2CO3?10H2O
| ||
��2��Na2CO3?10H2O��NaHCO3�����ֽ⣬������H2O��C�ܣ�ʢ��ˮCaCl2�����գ�CO2��D�ܣ�ʢ��ʯ�ң����գ�ͨ��C��D�ܵ��������ӿɼ���Na2CO3?10H2O��NaHCO3��������
�ʴ�Ϊ����ˮCaCl2����ʯ�ң�
��3��EΪ����ܣ�ʢ�м�ʯ�ң��ɷ�ֹ�������е�CO2��H2O����װ�ã��ʴ�Ϊ������ܣ���ֹ�����е�H2O��CO2����D�У�
��4������Aװ�û���ʢ��NaOH��Һ��ϴ��ƿ����m��H2O�����ӣ�ʹNa2CO3?10H2O��NaHCO3�ĺ���ƫ�ߣ�NaCl�ĺ���ƫ�ͣ��ʴ�Ϊ��ƫ�ͣ�
��5����Ӧǰ����Bװ����ͨ�������Ӧ�رյ��ɼ�b�����ɼ�a����װ���ڵ�ˮ�Ͷ�����̼�ų��������ܽ���Cװ���У�
�ʴ�Ϊ���رյ��ɼ�b�����ɼ�a����װ����ͨ�������
���������⿼���Ϊ�ۺϣ����ƵĻ�����Ϊ���壬�ۺϿ���ѧ��ʵ�������ͷ���������ע��������ʵ������Լ�ʵ��ԭ����Ϊ������Ĺؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
 Сѧ��ʱ��ҵȫͨ����ϵ�д�
Сѧ��ʱ��ҵȫͨ����ϵ�д� �����ÿ�ʱѵ��ϵ�д�
�����ÿ�ʱѵ��ϵ�д� ��Ԫȫ��������ϵ�д�
��Ԫȫ��������ϵ�д� �»ƸԱ����ܾ�ϵ�д�
�»ƸԱ����ܾ�ϵ�д�
�����Ŀ
�����⻯�������ȶ����ǣ�������
| A��CH4 |
| B��SiH4 |
| C��HCl |
| D��HF |
��ҵ�Ͻ���ͭ��ұ�����û�ͭ��Ϊԭ�ϣ���ͪ���ڱ��չ����з�������Ҫ��Ӧ�Ļ�����ʽΪ��2CuFeS2+O2=Cu2S+2FeS+SO2����Cu2S+O2�T2Cu+SO2 �����й�˵����ȷ���ǣ�������
| A����Ӧ���У�O2ֻ����������CuFeS2ֻ����ԭ�� |
| B������Ӧ��SO2 ������������ |
| C������1mol Cu���ɣ���������Ӧ��ת����6mol���� |
| D�����ڷ�Ӧ�٣�����1mol O2�μӷ�Ӧ����Ӧ���й�ת����4mol���� |
���еݱ��������ȷ���ǣ�������
| A��Na��Mg��Alԭ�ӵ��������������μ�С |
| B��C��N��O��ԭ�Ӱ뾶���μ�С |
| C��Si��P��SԪ�ص�������ν��� |
| D��Li��Na��K�Ľ��������μ��� |
 �黯�����ڵ������뵼�壬����ֱ�ӽ�����ת��Ϊ���ܣ��黯�ص�����������ͨ���ݵ�100����������ֻ������10%���ƹ��黯�صȷ�������ܣ�LED���������ǽ��ܼ��ŵ���Ч�ٴ룬��ͼ�黯�ؾ���������˵����ȷ���ǣ�������
�黯�����ڵ������뵼�壬����ֱ�ӽ�����ת��Ϊ���ܣ��黯�ص�����������ͨ���ݵ�100����������ֻ������10%���ƹ��黯�صȷ�������ܣ�LED���������ǽ��ܼ��ŵ���Ч�ٴ룬��ͼ�黯�ؾ���������˵����ȷ���ǣ�������| A���黯�ؾ����ṹ��NaCl��ͬ |
| B����һ�����ܣ�As��Ga |
| C���縺�ԣ�As��Ga |
| D������ض�����p��Ԫ�� |
����ɫ��ѧ��ʵ�����߽����ã��������������ϡ���ɫ��ѧ�����ǣ�������

| A��ʵ�����ռ���������ͼ1��ʾװ�� |
| B��ʵ���������������Ƶķ�Ӧʵ��ʱ����ͼ2��ʾװ�� |
| C��ʵ�������ò������ֱ�պȡŨ�����Ũ��ˮ���������ᷴӦ������ε�ʵ�� |
| D��ʵ�����в���ͼ3��ʾװ�ý���ͭ��ϡ����ķ�Ӧ |
 ijС��ͬѧ������ͼ��ʾװ��̽�����������ʣ�
ijС��ͬѧ������ͼ��ʾװ��̽�����������ʣ�