��Ŀ����
16��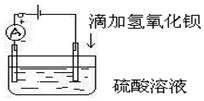 ��ͼ��ʾ��ij��ѧ��ȤС��̽����Һ������Ũ������Һ�����ԵĹ�ϵ��
��ͼ��ʾ��ij��ѧ��ȤС��̽����Һ������Ũ������Һ�����ԵĹ�ϵ��������ˮ���м���200ml��2mol/L��H2SO4��Һ���պϿ���K�������Ʒ���ƫת��Ȼ���ٽ������²������ش��������⣮
��1��������Һ����εμ�Ba��OH��2��Һ��
����Һ�з��������ӷ�Ӧ����ʽΪ��2H++SO42-+Ba2++2OH-=BaSO4��+2H2O
�ڵ�����ָ��ƫת�̶�A ������ţ�
A���ȱ�С����B�����䡢C���ȱ����С
��2����ijͬѧ��Ϊһ���¶�����Һ�����Գ���������Ũ���йأ������������������йأ�����Ϊ��ѡȡ������Щ��Һ�����о�C ������ţ���
A.0.1mol/LNaCl��Һ B.2mol/LNaCl��Һ
C.0.1mol/LMgSO4��Һ D.0.1mol/L������Һ��
���� ��1����H2SO4��Һ�еμ�Ba��OH��2��Һ�������ķ�ӦΪH2SO4+Ba��OH��2=BaSO4��+2H2O����Һ��������Һת��Ϊˮ����Һ�ܶȼ�С����Һ������Ũ�Ƚ��ͣ���Һ����������С��
��2����Һ�����Գ���������Ũ���йأ������������������ĵ���йأ�
��� �⣺��1������H2SO4��Һ�еμ�Ba��OH��2��Һ����Ӧ��ȫ�����������ӷ�Ӧ����ʽΪ2H++SO42-+Ba2++2OH-=BaSO4��+2H2O��
�ʴ�Ϊ��2H++SO42-+Ba2++2OH-=BaSO4��+2H2O��
���������������ĵ��룬��Һ�����ӵ�Ũ����С���������٣�����������������������Һǡ����ȫ��Ӧʱ����Һ�м���û�������ƶ������ӣ����Ե�����ƫת�̶���С�����Ϊ�㣬�ټ��������������õ���������������Һ����������������ǿ����ѡA��
��2����Һ�����Գ���������Ũ���йأ������������������ĵ���йأ�ѡ������Һ�е��������������ӡ������ӡ�þ���ӣ��������Ǵ�������ӡ��������Լ���������ӣ�����ѡ�����ɲ��ȵ�����þ�������飬����֤���������ĵ�ɺ���Һ��������֮��Ĺ�ϵ����ѡC��
���� ���⿼��Ԫ�ػ��������ʼ����ӷ�Ӧ����Һ�����Ե�֪ʶ�㣬Ϊ��Ƶ���㣬��ȷ������������Ũ�ȹ�ϵ�ǽⱾ��ؼ�����Ŀ�ѶȲ���

 ���ٴ�����ɽ����ϵ�д�
���ٴ�����ɽ����ϵ�д�| A�� |  ����һ�����ʵ���Ũ�ȵ�ϡ���� | B�� |  ��ȡ������Fe��OH��3���� | ||
| C�� |  ��������� | D�� |  ����NaOH���� |
| A�� | ԭ�Ӱ뾶�����������ң��� | |
| B�� | ��Ԫ�������ڱ��е�λ��Ϊ�������ڵڢ��� | |
| C�� | 1molA������B��ȫ��Ӧ��ת��1mol���� | |
| D�� | 1.0mol•L-1E��Һ���������ܵ����ʵ���С��0.1mol |
| A�� | ���� | B�� | ���� | C�� | �ᾧ | D�� | ��ȡ |
| A�� | ���³�ѹ�£�4g H2���еķ�����Ϊ 2NA | |
| B�� | 17g��������������ĿΪ8NA | |
| C�� | ��1���c1mol/L����������4���ˮ�У�ϡ��Һ��Ũ��Ϊ0.2c1mol/L | |
| D�� | ��5.85gNaCl��������100mLˮ�У��Ƶ�0.1mol/L��NaCl��Һ |