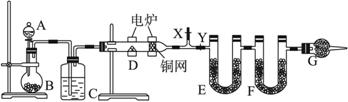
��Ŀ����
ij���ĺ���������X����Է�������С��150������������������Ϊ0.597���ҷ�������̼֧����1 mol X�����������Ʒ�Ӧ�ų�1.5 mol H2����1 molX��NaOH��Һ��ȫ�к�ʱ����2 mol NaOH���Իش�(1)X�����к��й����ŵ����ƣ�________________��
(2)X���ӵĽṹ��ʽΪ��______________________��
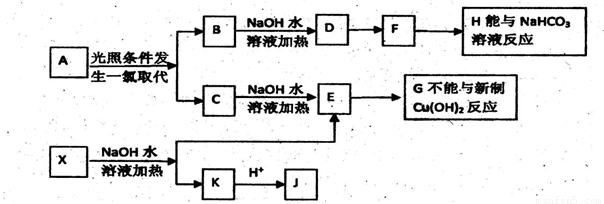
(3)X������һ�������¿ɷ������ֻ�ѧ��Ӧ(��ͼ)��
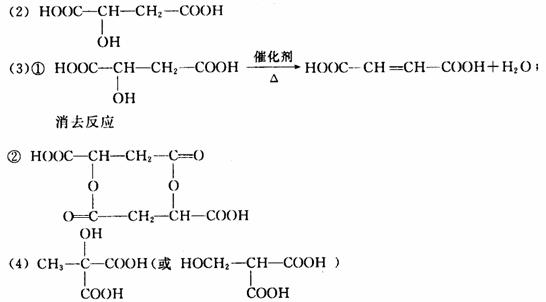
��д����Ӧ�ٵĻ�ѧ����ʽ��________________���䷴Ӧ����Ϊ��________________��
��д��Z�Ľṹ��ʽ��___________________��
(4)X��ͬ���칹���ж��֣���д����Xͬ����һ��ͬ���칹��Ľṹ��ʽ________________��
(1)�ǻ����Ȼ�


��ϰ��ϵ�д�
�����Ŀ