��Ŀ����
��1��ʵ�������������������A��F��ѡ���ʵ�������������ĸ����գ�

�ٳ������ܽ�������ʵ������� ��D�������� ��
��ʵ������37%��Ũ��������500ml 1mol?L-1��ϡ����ʱ��Ҫ�õ����������е� ������ѡ����ѡ����ѡ�������֣�
��2������װ�ó��������ʵķ�����ᴿ��������ͼװ�õ���ĸ�ش�
������װ��C�е�a��������Ϊ ��
�ڳ�ȥʯ��ˮ��������CaCO3��������Ҫ�� װ�ã�
�۴Ӻ��е�����Ȼ�̼��Һ�з�������ʵ�ͻ������Ȼ�̼����Ҫ�� װ�ã�


�ٳ������ܽ�������ʵ�������
��ʵ������37%��Ũ��������500ml 1mol?L-1��ϡ����ʱ��Ҫ�õ����������е�
��2������װ�ó��������ʵķ�����ᴿ��������ͼװ�õ���ĸ�ش�
������װ��C�е�a��������Ϊ
�ڳ�ȥʯ��ˮ��������CaCO3��������Ҫ��
�۴Ӻ��е�����Ȼ�̼��Һ�з�������ʵ�ͻ������Ȼ�̼����Ҫ��

���㣺���ʵķ��롢�ᴿ�Ļ�������ѡ����Ӧ��
ר�⣺ʵ�������,��ѧʵ���������
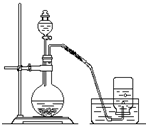
��������1�������ձ����ܽ���壬��ͼ��֪DΪ����ƿ��
����Ũ��Һ����ϡ��Һ��������Ͳ��ȡŨ���ᣬ���ձ���ϡ����ȴ��ת�Ƶ�����ƿ�У����ý�ͷ�ιܶ��ݣ�
��2����ͼ��֪��AΪ���ˣ�BΪ������CΪ����DΪ��ȡ��
����ͼ��֪C��aΪ������ƿ��
��̼��Ʋ�����ˮ��
�۴Ӻ��е�����Ȼ�̼��Һ�з�������ʵ�ͻ������Ȼ�̼�����÷е������룮
����Ũ��Һ����ϡ��Һ��������Ͳ��ȡŨ���ᣬ���ձ���ϡ����ȴ��ת�Ƶ�����ƿ�У����ý�ͷ�ιܶ��ݣ�
��2����ͼ��֪��AΪ���ˣ�BΪ������CΪ����DΪ��ȡ��
����ͼ��֪C��aΪ������ƿ��
��̼��Ʋ�����ˮ��
�۴Ӻ��е�����Ȼ�̼��Һ�з�������ʵ�ͻ������Ȼ�̼�����÷е������룮
���
�⣺��1�������ձ����ܽ���壬��ѡ����A����ͼ��֪DΪ����ƿ���ʴ�Ϊ��A������ƿ��
����Ũ��Һ����ϡ��Һ��������Ͳ��ȡŨ���ᣬ���ձ���ϡ����ȴ��ת�Ƶ�����ƿ�У����ý�ͷ�ιܶ��ݣ�����Ҫ������ΪABDF���ʴ�Ϊ��ABDF��
��2����ͼ��֪��AΪ���ˣ�BΪ������CΪ����DΪ��ȡ��
����ͼ��֪C��aΪ������ƿ���ʴ�Ϊ��������ƿ��
��̼��Ʋ�����ˮ�������ù��˷����룬ѡ��װ��A���ʴ�Ϊ��A��
�۴Ӻ��е�����Ȼ�̼��Һ�з�������ʵ�ͻ������Ȼ�̼�����÷е������룬��ѡ�������룬��ѡ��װ��C���ʴ�Ϊ��C��
����Ũ��Һ����ϡ��Һ��������Ͳ��ȡŨ���ᣬ���ձ���ϡ����ȴ��ת�Ƶ�����ƿ�У����ý�ͷ�ιܶ��ݣ�����Ҫ������ΪABDF���ʴ�Ϊ��ABDF��
��2����ͼ��֪��AΪ���ˣ�BΪ������CΪ����DΪ��ȡ��
����ͼ��֪C��aΪ������ƿ���ʴ�Ϊ��������ƿ��
��̼��Ʋ�����ˮ�������ù��˷����룬ѡ��װ��A���ʴ�Ϊ��A��
�۴Ӻ��е�����Ȼ�̼��Һ�з�������ʵ�ͻ������Ȼ�̼�����÷е������룬��ѡ�������룬��ѡ��װ��C���ʴ�Ϊ��C��
���������⿼����������ᴿ������ѡ���Ӧ�ã�Ϊ��Ƶ���㣬�������ʵ����ʡ����ʲ��켰�����������뷽����װ��Ϊ���Ĺؼ�������ʵ�����֪ʶ�ͻ������ܵĿ��飬��Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
�����ΪVaLŨ��Ϊ0.05mol?L-1CH3COOH��Һ�м������ΪVb LŨ��Ϊ0.05mol?L-1KOH��Һ�����й�ϵ������ǣ�������
| A��Va��Vbʱ��c ��CH3COOH��+c ��CH3COO-����c ��K+�� |
| B��Va=Vbʱ��c ��CH3COOH��+c ��H+��=c ��OH-�� |
| C��Va��Vbʱ��c ��CH3COO-����c ��K+����c ��OH-����c ��H+�� |
| D��Va��Vb�����ʱ��c ��K+��+c ��H+��=c ��OH-��+c ��CH3COO-�� |
��ҵ�������ڸ�¯�н��еģ���¯��������Ҫ��Ӧ�ǣ���2C����̿��+O2��������=2CO����Fe2O3+3CO=2Fe+3CO�����������У��Խ�̿��ʵ��ʹ����ҪԶԶ���ڰ��ջ�ѧ����ʽ������������Ҫԭ���ǣ���������
| A��CO���� |
| B��CO������ʯ�Ӵ������ |
| C��������¯�ĸ߶Ȳ��� |
| D��CO��Fe2O3�ķ�Ӧ��һ���� |
��֪H-H���ļ���Ϊ436kJ/mol��Cl-Cl���ļ���Ϊ243kJ/mol��H-Cl���ļ���Ϊ431kJ/mol����H2��g��+Cl2��g���T2HCl��g���ķ�Ӧ�ȣ���H�����ڣ�������
| A��862kJ/mol |
| B��183kJ/mol |
| C��-862kJ/mol |
| D��-183kJ/mol |
����������ȷ���ǣ�������
| A��ͬϵ����ܻ�Ϊͬ���칹�� |
| B�����黯ѧ���ʱȽ��ȶ������ܱ��κ����������� |
| C�������������Ӧ��������CH3Cl��CH2Cl2��CHCl3����CCl4��������ȡ����Ӧ |
| D��C4H10�������ĸ�̼ԭ����һ��ֱ���� |
 ������ͼ��ʾװ����ȡ������������ص�ʵ��̽����
������ͼ��ʾװ����ȡ������������ص�ʵ��̽����