��Ŀ����
B�����л���ѧ������
��1���л����й㷺������ͬ���칹����
������C5Hl2��ͬ���칹���� �֣�����ĸ����
a��2 b��3 c�� 4
������ͬ���칹���У��ܷ���������Ӧ���� ������ĸ����
a��CH3CH2CHO b��CH3CO CH3 c��CH2=CHCH2OH
������ͬ���칹���У�����������������Һ��Ӧ���� ������ĸ����

��2���л���X�Ľṹ��ʽΪCH3CHBrCH3��
��X�ĺ˴Ź�������ͼ���� �����շ壮
��X��NaOH��ˮ��Һ���ȿ������л���Y��Y�Ľṹ��ʽΪ ��
��X��NaOH�Ĵ���Һ���ȿ������л���Z���÷�Ӧ�Ļ�ѧ����ʽΪ ��
��3���л��ϳɸ߷��ӻ�����Ϊ�����ṩ�˷ḻ��ʵĺϳɲ��ϣ�
�� ��HOCH2CH2OH���ֵ����ͨ�� ��Ӧ���Ӧ���ͣ�����ϳ���ά���úϳ���ά�Ľṹ��ʽΪ ��
��HOCH2CH2OH���ֵ����ͨ�� ��Ӧ���Ӧ���ͣ�����ϳ���ά���úϳ���ά�Ľṹ��ʽΪ ��
�ھ۱�ϩ�������һ�������ĸ߷���Ϳ�ϣ���ṹ��ʽ��ͼ��
�ø߷��ӻ�����ĵ����� ����ṹ��ʽ�����õ���������������Һ��ȫ��Ӧ������ ����ṹ��ʽ����
��1���л����й㷺������ͬ���칹����
������C5Hl2��ͬ���칹����
a��2 b��3 c�� 4
������ͬ���칹���У��ܷ���������Ӧ����
a��CH3CH2CHO b��CH3CO CH3 c��CH2=CHCH2OH
������ͬ���칹���У�����������������Һ��Ӧ����

��2���л���X�Ľṹ��ʽΪCH3CHBrCH3��
��X�ĺ˴Ź�������ͼ����
��X��NaOH��ˮ��Һ���ȿ������л���Y��Y�Ľṹ��ʽΪ
��X��NaOH�Ĵ���Һ���ȿ������л���Z���÷�Ӧ�Ļ�ѧ����ʽΪ
��3���л��ϳɸ߷��ӻ�����Ϊ�����ṩ�˷ḻ��ʵĺϳɲ��ϣ�
��
 ��HOCH2CH2OH���ֵ����ͨ��
��HOCH2CH2OH���ֵ����ͨ���ھ۱�ϩ�������һ�������ĸ߷���Ϳ�ϣ���ṹ��ʽ��ͼ��

�ø߷��ӻ�����ĵ�����
���㣺�л���ĺϳ�,�л�������еĹ����ż���ṹ,�л���Ľṹ������,�л���ѧ��Ӧ���ۺ�Ӧ��
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
��������1��������C5Hl2��ͬ���칹�壬����̼���칹��
�ں�-CHO�����ʿɷ���������Ӧ��
�ۺ���-OH���л���������ԣ���Ӧ����������Ӧ��
��2����CH3CHBrCH3�к�2��λ�õ�H��
��X��NaOH��ˮ��Һ���ȿ������л���Y������ˮ�ⷴӦ��YΪ����
��X��NaOH�Ĵ���Һ���ȿ������л���Z��������ȥ��Ӧ��ZΪ��ϩ��
��3���ٺ�-OH��-COOH���л�������۷�Ӧ���ɸ߷��ӻ����
���ɽṹ��ʽ��֪���߷���Ϊ�Ӿ۷�Ӧ����������еİ���պϵõ�˫���������к��������ɷ���ˮ�ⷴӦ��
�ں�-CHO�����ʿɷ���������Ӧ��
�ۺ���-OH���л���������ԣ���Ӧ����������Ӧ��
��2����CH3CHBrCH3�к�2��λ�õ�H��
��X��NaOH��ˮ��Һ���ȿ������л���Y������ˮ�ⷴӦ��YΪ����
��X��NaOH�Ĵ���Һ���ȿ������л���Z��������ȥ��Ӧ��ZΪ��ϩ��
��3���ٺ�-OH��-COOH���л�������۷�Ӧ���ɸ߷��ӻ����
���ɽṹ��ʽ��֪���߷���Ϊ�Ӿ۷�Ӧ����������еİ���պϵõ�˫���������к��������ɷ���ˮ�ⷴӦ��
���
�⣺��1��������C5Hl2��ͬ���칹�壬����̼���칹����3��ͬ���칹�壬�ֱ�Ϊ�����顢�����顢�����飬�ʴ�Ϊ��b��
�ں�-CHO�����ʿɷ���������Ӧ��ֻ��a�ɷ���������Ӧ���ʴ�Ϊ��a��
�ۺ���-OH���л���������ԣ���Ӧ����������Ӧ��ֻ��bΪ��������Ӧ���ʴ�Ϊ��b��
��2����CH3CHBrCH3�к�2��λ�õ�H����˴Ź�������ͼ����2�����շ壬�ʴ�Ϊ��2��
��X��NaOH��ˮ��Һ���ȿ������л���Y������ˮ�ⷴӦ��YΪ����Y�Ľṹ��ʽΪCH3CHOHCH3���ʴ�Ϊ��CH3CHOHCH3��
��X��NaOH�Ĵ���Һ���ȿ������л���Z��������ȥ��Ӧ��ZΪ��ϩ���÷�Ӧ�Ļ�ѧ����ʽΪCH3CHBrCH3+NaOH
CH2=CHCH3+NaBr��
�ʴ�Ϊ��CH3CHBrCH3+NaOH
CH2=CHCH3+NaBr��
��3���� ��HOCH2CH2OH���ֵ��巢�����۷�Ӧ���ɸ߷��ӻ�����úϳ���ά�Ľṹ��ʽΪ
��HOCH2CH2OH���ֵ��巢�����۷�Ӧ���ɸ߷��ӻ�����úϳ���ά�Ľṹ��ʽΪ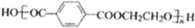 ��
��
�ʴ�Ϊ�����ۣ�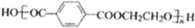 ��
��
���ɽṹ��ʽ��֪���߷���Ϊ�Ӿ۷�Ӧ����������еİ���պϵõ�˫������ �ĵ���ΪCH2=CHCOOCH3�����������ɷ���ˮ�ⷴӦ����NaOH��Ӧ��ˮ�ⷴӦΪCH2=CHCOOCH3+NaOH
�ĵ���ΪCH2=CHCOOCH3�����������ɷ���ˮ�ⷴӦ����NaOH��Ӧ��ˮ�ⷴӦΪCH2=CHCOOCH3+NaOH
CH2=CHCOONa+HOCH3������ȫ��Ӧ����CH2=CHCOONa��HOCH3��
�ʴ�Ϊ��CH2=CHCOOCH3��CH2=CHCOONa��HOCH3��
�ں�-CHO�����ʿɷ���������Ӧ��ֻ��a�ɷ���������Ӧ���ʴ�Ϊ��a��
�ۺ���-OH���л���������ԣ���Ӧ����������Ӧ��ֻ��bΪ��������Ӧ���ʴ�Ϊ��b��
��2����CH3CHBrCH3�к�2��λ�õ�H����˴Ź�������ͼ����2�����շ壬�ʴ�Ϊ��2��
��X��NaOH��ˮ��Һ���ȿ������л���Y������ˮ�ⷴӦ��YΪ����Y�Ľṹ��ʽΪCH3CHOHCH3���ʴ�Ϊ��CH3CHOHCH3��
��X��NaOH�Ĵ���Һ���ȿ������л���Z��������ȥ��Ӧ��ZΪ��ϩ���÷�Ӧ�Ļ�ѧ����ʽΪCH3CHBrCH3+NaOH
| �� |
| �� |
�ʴ�Ϊ��CH3CHBrCH3+NaOH
| �� |
| �� |
��3����
 ��HOCH2CH2OH���ֵ��巢�����۷�Ӧ���ɸ߷��ӻ�����úϳ���ά�Ľṹ��ʽΪ
��HOCH2CH2OH���ֵ��巢�����۷�Ӧ���ɸ߷��ӻ�����úϳ���ά�Ľṹ��ʽΪ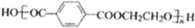 ��
���ʴ�Ϊ�����ۣ�
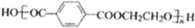 ��
�����ɽṹ��ʽ��֪���߷���Ϊ�Ӿ۷�Ӧ����������еİ���պϵõ�˫������
 �ĵ���ΪCH2=CHCOOCH3�����������ɷ���ˮ�ⷴӦ����NaOH��Ӧ��ˮ�ⷴӦΪCH2=CHCOOCH3+NaOH
�ĵ���ΪCH2=CHCOOCH3�����������ɷ���ˮ�ⷴӦ����NaOH��Ӧ��ˮ�ⷴӦΪCH2=CHCOOCH3+NaOH| ˮ |
| �� |
�ʴ�Ϊ��CH2=CHCOOCH3��CH2=CHCOONa��HOCH3��
���������⿼����ۺϣ��漰�л���ĺϳɡ��л���Ľṹ�����ʡ�ͬ���칹��ȣ�Ϊ��Ƶ���㣬���չ����������ʵĹ�ϵΪ���Ĺؼ�������±�������������ᡢ�����ʵĿ��飬��Ŀ�ѶȲ���

��ϰ��ϵ�д�
 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�
�����Ŀ
����ʡ������ʵʩ����ˮ���족���̣����в����ڡ���ˮ���족����ʵʩ���ǣ�������
| A��Ѹ��������ʡ�ļ������������� |
| B����ǿ����������ˮ�ѵ����״���������ˮ�帻Ӫ���� |
| C�������ƹ�̫���ܡ����ܡ������ܼ�ˮ�ܵȵ�ʹ�ã����ٻ�ʯȼ�ϵ�ʹ�� |
| D������ʵʩ����ȼ�ϡ������������������������������͵�����������Ⱦ |
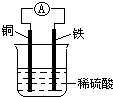 ��ͼΪͭ��ԭ���ʾ��ͼ�������й�˵����ȷ���ǣ�������
��ͼΪͭ��ԭ���ʾ��ͼ�������й�˵����ȷ���ǣ�������| A��ͭ�����ܽ� |
| B����װ���ܽ�����ת��Ϊ��ѧ�� |
| C������������ͨ����������ͭ�� |
| D��������ӦΪFe-2e-=Fe2+ |
Ϊ�˸��ƿ���������������ƴ�������Ⱦ����ŷ��������������У�δ�����ҹ���������������ǣ�������
| A���������� | B��������̼ |
| C���������� | D������������� |