��Ŀ����
20��ij��������¯��������Ҫ��Ӧ������ͼ��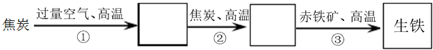
��1��д������ڢ۵Ļ�ѧ����ʽ��
��CO2+C$\frac{\underline{\;����\;}}{\;}$2CO��
��3CO+Fe2O3$\frac{\underline{\;����\;}}{\;}$2Fe+3CO2��
��2�������йظ�¯������˵����ȷ����c��
a�����Ӹ�¯�ĸ߶ȿ������CO��������
b��������Ӧ��Ϊ���ȷ�Ӧ
c���Ӹ�¯¯�������������к���û�����õ�CO����
d��ֻ�в���ڵķ�Ӧ�ǿ��淴Ӧ��
���� ��¯����������Ϊ����̿����������ڸ������������ɶ�����̼��������̼���뽹̿�ڸ�������������һ����̼������һ����̼�ѳ������е�����ԭ������
��1�����ݷ�Ӧ��Ͳ����Լ�������д��ѧ����ʽ����Ӧ��Ϊ������̼�뽹̿�ڸ��������µķ�Ӧ����Ӧ��Ϊһ����̼��ԭ�������е�����
��2����¯�ĸ߶���ƽ���ƶ��أ�ȼ�շ�ӦΪ���ȷ�Ӧ��һ����̼������ȫ��ת����������Ӧ�������森
��� �⣺��1����̿����������ڸ������������ɶ�����̼C+O2$\frac{\underline{\;����\;}}{\;}$CO2��������̼���뽹̿�ڸ�������������һ����̼������һ����̼�ѳ������е�����ԭ������
�ʲ���ڻ�ѧ����ʽΪ��CO2+C$\frac{\underline{\;����\;}}{\;}$2CO������ۻ�ѧ����ʽΪ��3CO+Fe2O3$\frac{\underline{\;����\;}}{\;}$2Fe+3CO2��
�ʴ�Ϊ��CO2+C$\frac{\underline{\;����\;}}{\;}$2CO��3CO+Fe2O3$\frac{\underline{\;����\;}}{\;}$2Fe+3CO2��
��2��a����¯�ĸ߶���ƽ���ƶ��أ����Ӹ�¯�ĸ߶Ȳ��������CO�������ʣ���a����
b����̿����������ڸ������������ɶ�����̼C+O2$\frac{\underline{\;����\;}}{\;}$CO2���÷�ӦΪ���ȷ�Ӧ����b����
c�������һ����̼����������Ӧ�����ԴӸ�¯¯�������������к���û�����õ�CO���壬��c��ȷ��
d��C+O2$\frac{\underline{\;����\;}}{\;}$CO2��CO2+C$\frac{\underline{\;����\;}}{\;}$2CO��3CO+Fe2O3$\frac{\underline{\;����\;}}{\;}$2Fe+3CO2�������ǿ��淴Ӧ����d����
��ѡc��
���� ���⿼���¯��������������̼���仯����������Լ�����ԭ���ǽ��ؼ�����Ŀ�ѶȲ���

 ����ѧ��ţ��Ӣ��ϵ�д�
����ѧ��ţ��Ӣ��ϵ�д�| A�� | ú�ĸ����������仯 | |
| B�� | ʯ���ѽ��Ŀ����Ҫ��Ϊ��������͵IJ��������� | |
| C�� | ʯ�ͷ����ܵõ���ͬ�е㷶Χ�IJ��� | |
| D�� | ʯ���ѻ���Ҫ�õ�������ϩ����ϩ�� |
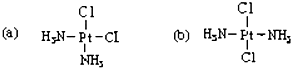 ��ѧ�ҷ��ֲ������ֻ������ͼ���в�ͬ�����ԣ�a ���п������ã��� b û�У��� a �� b ���� ��������
��ѧ�ҷ��ֲ������ֻ������ͼ���в�ͬ�����ԣ�a ���п������ã��� b û�У��� a �� b ���� ��������| A�� | ͬ���칹�� | B�� | ͬ�������� | C�� | ͬλ�� | D�� | ͬһ������ |
| A�� | Ũ���� | B�� | ϡ���� | C�� | Ũ���� | D�� | ʳ�ô� |
| A�� | ֻ�Ժ�ɫ | B�� | ����ɫ | C�� | ��ɫ����ɫ | D�� | ��ɫ����ɫ |

��1����֪A��BΪ��������Ԫ�أ���ԭ�ӵĵ�һ�����ĵ����������ʾ��
| ������/kJ•mol-1 | I1 | I2 | I3 | I4 |
| A | 578 | 1817 | 2745 | 11578 |
| B | 738 | 1451 | 7733 | 10540 |
��2��ʵ��֤����KCl��MgO��CaO��TiN��4�־���Ľṹ��NaCl����ṹ���ƣ���ͼ1��ʾ������֪3�����Ӿ���ľ��������������
| ���Ӿ��� | NaCl | KCl | CaO |
| ������/kJ•mol-1 | 786 | 715 | 3401 |
��3�����������Ӻ�δ�ɶԵ���Խ�࣬�����Խ�ż�¼����Խ�ã�������������V2O5��CrO2�У��ʺ���¼�����ŷ�ԭ�ϵ���CrO2��
��4��ij�����ķ��ӽṹ��ͼ2��ʾ��������ڲ�����AC������ţ���
A�����Ӽ� B�����Լ� C�������� D����λ�� E����� F���Ǽ��Լ���
 ��
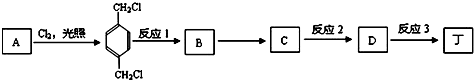
��
 ��
�� ԭ��������������缫֮������ԵIJ�ͬ���������Ʋ�Ӷ�ʹ���ӵ��������������������ſ�ѧ�����IJ��Ϸ�չ����ԭ���ԭ��Ϊ��������ѧ�������Ƴ��˸�ʽ�����Ļ�ѧ��Դ����Ӧ��ԭ������֪ʶ�ش�
ԭ��������������缫֮������ԵIJ�ͬ���������Ʋ�Ӷ�ʹ���ӵ��������������������ſ�ѧ�����IJ��Ϸ�չ����ԭ���ԭ��Ϊ��������ѧ�������Ƴ��˸�ʽ�����Ļ�ѧ��Դ����Ӧ��ԭ������֪ʶ�ش�