��Ŀ����
ijУ�о���ѧϰС��ͬѧ�Ծ���������������Ũ����ˮ(��Ҫ��NaCl��MgSO4)�����о���
��1��ʵ��ʱ����Ҫ����1 000 mL 0��20 mol��L-1NaOH��Һ������Ҫ��������NaOH������Ϊ������g��
��2��ȡ��������������Ũ����ˮ100 mL������200 mL 0�� 20 mol��L-1NaOH��Һ��ǡ�ð����е�Mg2+��ȫ��������Ũ����ˮ��Mg2+�����ʵ���Ũ��Ϊ��������
��3���о�С��ͬѧ������������ȫת��Ϊ��ˮMgCl2����������״̬�½��е�⣬�ɵñ�״����Cl2�����Ϊ��������L��
��1��8����2��0.2 mol��L-1����3��0.448
��������
�����������1��n(NaOH)=0.20 mol��L-1��1 L=0.20 mol��m(NaOH)=0.20 mol��40 g��mol-1=8 g��
��2������MgCl2+2NaOH====Mg(OH)2��+2NaCl��֪��n(Mg2+)= n(NaOH)=
n(NaOH)= ��0.20 mol��L-1��0.2 L
��0.20 mol��L-1��0.2 L
=0.02 mol��c(Mg2+)=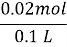 =0.2 mol��L-1��
=0.2 mol��L-1��
��3����MgCl2(����) Mg+Cl2����ϣ�2��������֪��n(Cl2)=n(MgCl2)=0.02 mol��V(Cl2)=
Mg+Cl2����ϣ�2��������֪��n(Cl2)=n(MgCl2)=0.02 mol��V(Cl2)=
0.02 mol��22.4 L��mol-1=0.448 L��
���㣺þ����ȡ��Ӧ��

��ϰ��ϵ�д�
�����Ŀ
 B
B D��
D��