��Ŀ����
A��B��C��D��E��F��G��H��I��J��Ϊ�л�������������¿�ͼ���ش����⣺

��1��B��C��Ϊ��֧�����л������B�Ľṹ��ʽΪ ��C��Ũ���������¼��ȷ�Ӧֻ������һ��ϩ��D��D�Ľṹ��ʽΪ ���۵ķ�Ӧ����Ϊ ��
��2��G�ܷ���������Ӧ��Ҳ��ʹ������Ȼ�̼��Һ��ɫ����G�Ľṹ��ʽΪ ��
��3����Ļ�ѧ��Ӧ����ʽ ��
��4��C��C4H10O����ͬ���칹���� �֣���H������ͬ�����ŵ�H��ͬ���칹��Ľṹ��ʽΪ ��

��1��B��C��Ϊ��֧�����л������B�Ľṹ��ʽΪ
��2��G�ܷ���������Ӧ��Ҳ��ʹ������Ȼ�̼��Һ��ɫ����G�Ľṹ��ʽΪ
��3����Ļ�ѧ��Ӧ����ʽ
��4��C��C4H10O����ͬ���칹����
���㣺�л�����ƶ�
ר�⣺
����������A��B+C��ˮ�ⷴӦ�������ж�A��B��C�ֱ�����������ʹ������ɣ�1������ȷ��B��C�Ľṹ�ֱ�Ϊ��CH3��2CHCOOH�ͣ�CH3��2CHCH2OH����DΪ ��CH3��2C=CH2����D��E��ȡ����Ӧ��EΪ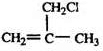 ��E����ˮ��õ���F��GΪȩ�Һ���˫��������д����ṹΪCH2=C��CH3��-CHO��������Ӧ�ߵõ�����HΪCH2=C��CH3��-COOH��H��CH3OH�õ���IΪCH2=C��CH3��-COOCH3����JΪ�Ӿ۷�Ӧ�IJ��Ϊ
��E����ˮ��õ���F��GΪȩ�Һ���˫��������д����ṹΪCH2=C��CH3��-CHO��������Ӧ�ߵõ�����HΪCH2=C��CH3��-COOH��H��CH3OH�õ���IΪCH2=C��CH3��-COOCH3����JΪ�Ӿ۷�Ӧ�IJ��Ϊ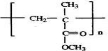 ������л���Ľṹ�������Լ���ĿҪ��ɽ����⣮
������л���Ľṹ�������Լ���ĿҪ��ɽ����⣮
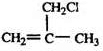 ��E����ˮ��õ���F��GΪȩ�Һ���˫��������д����ṹΪCH2=C��CH3��-CHO��������Ӧ�ߵõ�����HΪCH2=C��CH3��-COOH��H��CH3OH�õ���IΪCH2=C��CH3��-COOCH3����JΪ�Ӿ۷�Ӧ�IJ��Ϊ
��E����ˮ��õ���F��GΪȩ�Һ���˫��������д����ṹΪCH2=C��CH3��-CHO��������Ӧ�ߵõ�����HΪCH2=C��CH3��-COOH��H��CH3OH�õ���IΪCH2=C��CH3��-COOCH3����JΪ�Ӿ۷�Ӧ�IJ��Ϊ ������л���Ľṹ�������Լ���ĿҪ��ɽ����⣮
������л���Ľṹ�������Լ���ĿҪ��ɽ����⣮���
�⣺����A��B+C��ˮ�ⷴӦ�������ж�A��B��C�ֱ�����������ʹ������ɣ�1������ȷ��B��C�Ľṹ�ֱ�Ϊ��CH3��2CHCOOH�ͣ�CH3��2CHCH2OH����DΪ ��CH3��2C=CH2����D��E��ȡ����Ӧ��EΪ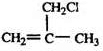 ��E����ˮ��õ���F��GΪȩ�Һ���˫��������д����ṹΪCH2=C��CH3��-CHO��������Ӧ�ߵõ�����HΪCH2=C��CH3��-COOH��H��CH3OH�õ���IΪCH2=C��CH3��-COOCH3����JΪ�Ӿ۷�Ӧ�IJ��Ϊ
��E����ˮ��õ���F��GΪȩ�Һ���˫��������д����ṹΪCH2=C��CH3��-CHO��������Ӧ�ߵõ�����HΪCH2=C��CH3��-COOH��H��CH3OH�õ���IΪCH2=C��CH3��-COOCH3����JΪ�Ӿ۷�Ӧ�IJ��Ϊ ��
��
��1�������Ϸ�����֪BΪ��CH3��2CHCOOH��DΪ��CH3��2C�TCH2����Ӧ��Ϊ��CH3��2CHCH2OH��ˮ���ɣ�CH3��2C=CH2������ȥ��Ӧ��
�ʴ�Ϊ����CH3��2CHCOOH����CH3��2C�TCH2����ȥ��Ӧ��
��2�������Ϸ�����֪GΪCH2=C��CH3��-CHO��
�ʴ�Ϊ��CH2=C��CH3��-CHO��
��3����Ӧ��ΪCH2=C��CH3��COOH��״�����������Ӧ����Ӧ����ʽΪ��CH2=C��CH3��COOH+CH3OH
CH2=C��CH3��COOCH3+H2O��
�ʴ�Ϊ��CH2=C��CH3��COOH+CH3OH
CH2=C��CH3��COOCH3+H2O��
��4��C��C4H10O����ͬ���칹����CH3CH2CH2CH2OH��CH3CH2CHOHCH3��CH3CH��CH3��CH2OH��CH3COH��CH3��2��CH3CH2CH2OCH3��CH3CH2OCH2CH3��CH3CH��CH3��OCH3����7�֣�HΪCH2=C��CH3��-COOH����H������ͬ�����ŵ�H��ͬ���칹����CH2=CHCH2COOH ��CH3CH=CHCOOH��
�ʴ�Ϊ��7��CH2=CHCH2COOH ��CH3CH=CHCOOH��
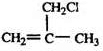 ��E����ˮ��õ���F��GΪȩ�Һ���˫��������д����ṹΪCH2=C��CH3��-CHO��������Ӧ�ߵõ�����HΪCH2=C��CH3��-COOH��H��CH3OH�õ���IΪCH2=C��CH3��-COOCH3����JΪ�Ӿ۷�Ӧ�IJ��Ϊ
��E����ˮ��õ���F��GΪȩ�Һ���˫��������д����ṹΪCH2=C��CH3��-CHO��������Ӧ�ߵõ�����HΪCH2=C��CH3��-COOH��H��CH3OH�õ���IΪCH2=C��CH3��-COOCH3����JΪ�Ӿ۷�Ӧ�IJ��Ϊ ��
����1�������Ϸ�����֪BΪ��CH3��2CHCOOH��DΪ��CH3��2C�TCH2����Ӧ��Ϊ��CH3��2CHCH2OH��ˮ���ɣ�CH3��2C=CH2������ȥ��Ӧ��
�ʴ�Ϊ����CH3��2CHCOOH����CH3��2C�TCH2����ȥ��Ӧ��
��2�������Ϸ�����֪GΪCH2=C��CH3��-CHO��
�ʴ�Ϊ��CH2=C��CH3��-CHO��
��3����Ӧ��ΪCH2=C��CH3��COOH��״�����������Ӧ����Ӧ����ʽΪ��CH2=C��CH3��COOH+CH3OH
| Ũ���� |
| �� |
�ʴ�Ϊ��CH2=C��CH3��COOH+CH3OH
| Ũ���� |
| �� |
��4��C��C4H10O����ͬ���칹����CH3CH2CH2CH2OH��CH3CH2CHOHCH3��CH3CH��CH3��CH2OH��CH3COH��CH3��2��CH3CH2CH2OCH3��CH3CH2OCH2CH3��CH3CH��CH3��OCH3����7�֣�HΪCH2=C��CH3��-COOH����H������ͬ�����ŵ�H��ͬ���칹����CH2=CHCH2COOH ��CH3CH=CHCOOH��
�ʴ�Ϊ��7��CH2=CHCH2COOH ��CH3CH=CHCOOH��
���������⿼���л�����ƶϣ����ؼ����ҽ����ͻ�ƿڣ������ۣ�������A��B+C��ˮ�ⷴӦ�������ж�A��B��C�ֱ�����������ʹ������ɣ�1������ȷ��B��C�Ľṹ���Դ˿��ƶ��������ʣ�ע���л�������ŵĽṹ�����ʣ�Ϊ��ȷ��������Ŀ�Ĺؼ�����Ŀ�Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
�л���X��Y����ʽ��ͬ������ֻ��C��H��OԪ���е����ֻ����֣�����X��Y���ۺ��ֱ�����ϣ�ֻҪ���ʵ���֮�Ͳ��䣬��ȫȼ��ʱ�������������ɵ�ˮ�����ʵ���Ҳ���䣮X��Y�ǣ�������
| A��C2H4��C2H6O |
| B��C2H2��C6H6 |
| C��CH2O��C3H6O |
| D��CH4��C2H4O2 |
��ѧƽ�ⳣ��K����ֵ��С�Ǻ�����ѧ��Ӧ���г̶ȵı�־���ڳ����£�ijЩ��Ӧ��ƽ�ⳣ����ֵ���£�
2NO��g��?N2��g��+O2��g�� K1=1��1030 2H2��g��+O2��g��?2H2O��g�� K2=2��1081
2CO2��g��?2CO��g��+O2��g�� K3=4��10-92
����˵������ȷ���ǣ�������
2NO��g��?N2��g��+O2��g�� K1=1��1030 2H2��g��+O2��g��?2H2O��g�� K2=2��1081
2CO2��g��?2CO��g��+O2��g�� K3=4��10-92
����˵������ȷ���ǣ�������
| A�������£�ˮ�ֽ����O2����ʱƽ�ⳣ����ֵԼΪ5��10-80 | ||
| B�������£����ֽ�ų�O2����ˮ | ||
C��K3=
| ||
| D�����϶�����ȷ |
 ����һ��������ζ�����ϣ��Ա�ͪΪ��Ҫԭ�Ϻϳ�3-�ǻ���ͪ�����ܽ�һ���ϳɶ��ָ߾�����о۶Ա�������-2��3-����������PBT��������͵Ĺ������ܲ���֮һ���ϳ���·���£�
����һ��������ζ�����ϣ��Ա�ͪΪ��Ҫԭ�Ϻϳ�3-�ǻ���ͪ�����ܽ�һ���ϳɶ��ָ߾�����о۶Ա�������-2��3-����������PBT��������͵Ĺ������ܲ���֮һ���ϳ���·���£�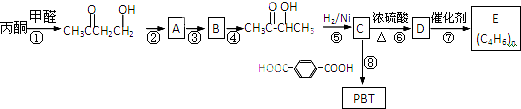


 ϵͳ����Ϊ
ϵͳ����Ϊ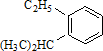 ��Ȳ���Ľṹ��ʽ
��Ȳ���Ľṹ��ʽ