��Ŀ����
16��pH=0��ij��Һ�л����ܴ���Al3+��Fe2+��NH4+��Ba2+��Cl-��CO32-��SO42-��NO3-�е������֣���ȡ������Һ��������һϵ��ʵ�飺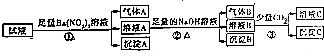
�����й��ж���ȷ���ǣ�������
| A�� | ��Һ��һ����Fe2+��SO42-��H+��NH4+��Al3+ | |
| B�� | ��Һ��һ��û��Ba2+��CO32-��NO3- | |
| C�� | ������з�Ӧ�����ӷ���ʽΪ��2AlO2-+CO2+3H2O�T2Al��OH��3��+CO32- | |
| D�� | ����A��B֮��϶����ܷ�����Ӧ |
���� ijǿ������Һ��CO32-�����ڣ�Fe2+��NO3-����ͬʱ���ڣ���Һ�м���������ᱵ��Һ����������֤��һ������Fe2+����ԭ��Һ��һ��������NO3-������ΪNO������AΪBaSO4��ԭ��Һ��һ��������Ba2+��һ������SO42-����ҺA�м����������������Һ��������BΪNH3��ȷ��ԭ��Һ�к���NH4+������BΪFe��OH��3��������ҺB�к���Ba2+��OH-����ҺB��������������̼������Ӧ�ۣ����ɵij���һ������BaCO3�������������ɳ���Al��OH��3��ԭ��Һ�в�һ������Al3+���Դ˽����⣮
��� �⣺ijǿ������Һ��CO32-�����ڣ�Fe2+��NO3-����ͬʱ���ڣ���Һ�м���������ᱵ��Һ����������֤��һ������Fe2+����ԭ��Һ��һ��������NO3-������ΪNO������AΪBaSO4��ԭ��Һ��һ��������Ba2+��һ������SO42-����ҺA�м����������������Һ��������BΪNH3��ȷ��ԭ��Һ�к���NH4+������BΪFe��OH��3��������ҺB�к���Ba2+��OH-����ҺB��������������̼������Ӧ�ۣ����ɵij���һ������BaCO3�������������ɳ���Al��OH��3��ԭ��Һ�в�һ������Al3+��
A�������Ϸ�����֪ԭ��Һ��һ����Fe2+��SO42-��H+��NH4+����һ������Al3+����A����
B������������֪��ԭ��Һ��һ��û�е�����Ϊ��Ba2+��CO32-��NO3-����B��ȷ��
C��������з�Ӧ�����ӷ���ʽ����ΪBa2++2OH-+CO2=BaCO3��+H2O����2AlO2-+CO2+3H2O=2Al��OH��3��+CO32-����C����
D������AΪNO������BΪNH3������֮���ܷ���������ԭ��Ӧ����D����
��ѡB��
���� ���⿼�����ʵ��ƶ��Լ����ӷ�Ӧ��֪ʶ��Ϊ�߿��������ͣ���Ŀ�Ѷ��еȣ�ע�����ճ������ӵ����ʼ����鷽����Ҫ��ѧ���ܹ����ݷ�Ӧ�����ж����Ӵ������������������ѧ�����Ӧ����ѧ֪ʶ��������

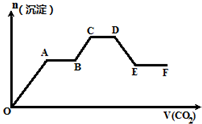 ��CO2���建����ͨ�뵽��KOH��Ba��OH��2��KAlO2�Ļ����Һ��ֱ�����������ɳ��������ʵ�������ͨCO2�������ϵ��ͼ��ʾ�����й���������Ӧ�����еĸ�����������ȷ���ǣ�������
��CO2���建����ͨ�뵽��KOH��Ba��OH��2��KAlO2�Ļ����Һ��ֱ�����������ɳ��������ʵ�������ͨCO2�������ϵ��ͼ��ʾ�����й���������Ӧ�����еĸ�����������ȷ���ǣ�������| A�� | O��A�η�Ӧ�����ӷ���ʽ��Ba2++CO32-�TBaCO3�� | |
| B�� | A��B�η�Ӧ�����ӷ���ʽ��2OH-+CO2�TCO${\;}_{3}^{2-}$+H2O | |
| C�� | A��B����C��D���������ķ�Ӧ��ͬ | |
| D�� | D��E�γ����ļ���������̼�ᱵ������CO2��ˮ��Һ��Ӧ���� |
| A�� | ���ȵĴ�����Һȥ���� | B�� | ����������Һ������ | ||
| C�� | С�մ���Һ�������� | D�� | Ũ������Һ�г�ζ |
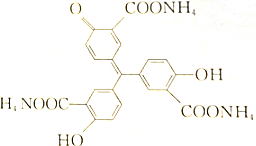 ������ͼ��ʾ���������˵���д�����ǣ�������
������ͼ��ʾ���������˵���д�����ǣ�������| A�� | �û�����Ļ�ѧʽΪC22H23O9N3 | |
| B�� | ����������3���Ȼ������кͣ�����ȫ��Ӧ1mol�û����ﻹ��2molNaOH | |
| C�� | �û��������FeCl3������ɫ��Ӧ������������Һ��Ӧ | |
| D�� | ÿ1mol�û��������5molBr2��Ӧ |
| A�� | Cl2��ˮ��Ӧ��Cl2+H2O�T2H++ClO-+Cl- | |
| B�� | CuSO4��Һ�м���Ba��OH��2��Һ��Ba2++SO42-�TBaSO4�� | |
| C�� | ���������ڴ������ܽ������ݲ�����CaCO3+2H+�TCa2++H2O+CO2�� | |
| D�� | �������ᷴӦ��Fe+2H+�TFe2++H2�� |
| A�� | 10 ��b-2a�� mol•L-1 | B�� | 10��2a-b�� mol•L-1 | C�� | 20��a-2b�� mol•L-1 | D�� | 20��b-2a�� mol•L-1 |
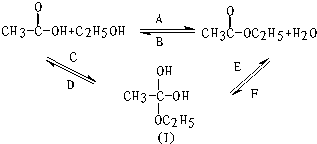 ��ͼ��ʾ��������Ҵ���Ũ������ڲ����ȵ������·���������Ӧ����ӦA�������淴Ӧ��ˮ�ⷴӦ����ӦB������Ӧ���ܾ����������м��壨����һ����
��ͼ��ʾ��������Ҵ���Ũ������ڲ����ȵ������·���������Ӧ����ӦA�������淴Ӧ��ˮ�ⷴӦ����ӦB������Ӧ���ܾ����������м��壨����һ����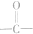 �����ǻ�������ԭ����18O��ǣ���������ˮ��ǰ��û�С������У���С���û�С���18O�����������жϵ���������������е��ǻ��봼�������ǻ�����ԭ�ӽ�ϳ�ˮ��
�����ǻ�������ԭ����18O��ǣ���������ˮ��ǰ��û�С������У���С���û�С���18O�����������жϵ���������������е��ǻ��봼�������ǻ�����ԭ�ӽ�ϳ�ˮ��