��Ŀ����
9�����Ͽα���ʵ������ȡ����������ʵ�飬������и��⣺��1���ڴ��Թ�������һ���������Ҵ��������ŨH2SO4���Һ����ǰ����Ҫ�ڴ��Թ��м��뼸�����Ƭ�������Ƿ�ֹ���У�����Na2CO3��Һ���������ܽ��Ҵ�����Ӧ���ᣬ���������������ܽ�ȣ������ܲ��ܲ��뱥��Na2CO3��Һ����Ϊ�˷�ֹ��Һ������
��2��ʵ�������ɵ��������������ܶȱ�ˮС�����С��������д���Ҵ���������ŨH2SO4���������������������������Ļ�ѧ����ʽ��CH3COOH+CH3CH2OH
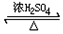 CH3COOC2H5+H2O���÷�Ӧ���л���Ӧ������ȡ������������Ӧ��
CH3COOC2H5+H2O���÷�Ӧ���л���Ӧ������ȡ������������Ӧ��
���� ��1���е�͵�Һ�����Ҫ�����Ƭ����ֹ���У�����̼������Һ�����ᷴӦ��ȥ���ᡢͬʱ���������������ܽ�ȣ����ڷֲ㣻��������Һ���¿��ܷ���������
��2�������������ܶȱ�ˮС��������Ӧ�ı���Ϊ�����ǻ��������⣬�÷�Ӧ��������������ˮ����Ϊ���淴Ӧ���÷�Ӧ����ȡ������������Ӧ��
��� �⣺��1��Һ�����ᡢ�Ҵ��е�ͣ�����Ҫ�����Ƭ����ֹ���У��Ʊ���������ʱ���ñ���̼������Һ��Ŀ�����кͻӷ����������ᣬʹ֮ת��Ϊ����������ˮ�У�������������������ζ���ܽ�ӷ��������Ҵ�����������������ˮ�е��ܽ�ȣ����ڷֲ�õ��������ܲ��ܲ�����Һ�У�����Ҫ���ڱ���̼������Һ��Һ���ϣ�����Һ���¿��ܷ���������
�ʴ�Ϊ����ֹ���У��ܽ��Ҵ�����Ӧ���ᣬ���������������ܽ�ȣ���ֹ��Һ������
��2�������������ܶȱ�ˮС��������Ӧ�ı���Ϊ�����ǻ��������⣬�������Ҵ���Ũ���������¼��ȷ���������Ӧ��������������ˮ���÷�ӦΪ���淴Ӧ����ӦΪCH3COOH+CH3CH2OH 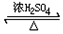 CH3COOC2H5+H2O���÷�Ӧ����ȡ����Ӧ��������Ӧ����
CH3COOC2H5+H2O���÷�Ӧ����ȡ����Ӧ��������Ӧ����
�ʴ�Ϊ��С��CH3COOH+CH3CH2OH 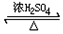 CH3COOC2H5+H2O��ȡ������������
CH3COOC2H5+H2O��ȡ������������
���� ���⿼�������������Ʊ������ʱ��ע��������Ӧ��ԭ���ͱ���̼������Һ�����ã���Ŀ�ѶȲ���

 ��У����ϵ�д�
��У����ϵ�д�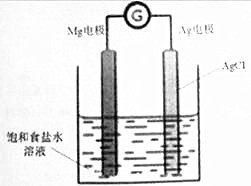 Mg/AgCl�����һ�ֻ�ѧ��Դ����þ�ͱ�����Ȼ�������Ϊ�缫������ʳ��ˮΪ���Һ����ͼ��ʾ�������ķ�Ӧ����ʽΪMg+2AgCl=MgCl2+2Ag������˵����ȷ���ǣ�������
Mg/AgCl�����һ�ֻ�ѧ��Դ����þ�ͱ�����Ȼ�������Ϊ�缫������ʳ��ˮΪ���Һ����ͼ��ʾ�������ķ�Ӧ����ʽΪMg+2AgCl=MgCl2+2Ag������˵����ȷ���ǣ�������| A�� | þ�缫�Ϸ�����ԭ��Ӧ | |
| B�� | ���缫�ĵ缫��Ӧʽ��AgCl+e-=Ag+Cl- | |
| C�� | ��װ��������ת����ʽΪ����ת��Ϊ��ѧ�� | |
| D�� | ��ع���ʱ������������þ�缫���������������缫 |
��Na+��Ba2+��C1-��HCO3-
��K+��NH4+��CH3COO-��SO42-
��Ca2+��NH4+��SO42-��SO32-
��Fe3+��Na+��SCN-��C1-
��A13+��Na+��HCO3-��NO3-��
| A�� | ֻ�Т� | B�� | �ڢ� | C�� | �٢� | D�� | �٢� |
| A | B | C | D | E | F | G | H | I | J | |
| ԭ�Ӱ뾶��10-10 m�� | 1.02 | 2.27 | 0.74 | 1.43 | 0.77 | 1.10 | 0.99 | 1.86 | 0.75 | 1.17 |
| ���̬ | +6 | +1 | - | +3 | +4 | +5 | +7 | +1 | +5 | +4 |
| ��ͼ�̬ | -2 | - | -2 | - | -4 | -3 | -1 | - | -3 | -4 |
��1������10��Ԫ�ص縺��������O����Ԫ�ط��ţ�����Ԫ��Bԭ��������7��Ԫ����Ԫ�����ڱ��е�λ�ã���4���ڣ���VIII�壬���̬ԭ�ӵĺ�������Ų�ʽ��1s22s22p63s23p63d64s2����[Ar]3d64s2����
��2��H��I��J����Ԫ�ض�Ӧ���ʵ��۵��������ߵ�˳���ǣ��õ��ʵľ��廯ѧʽ����N2��Na��Si��
��3��Ԫ��E��C����Ԫ�ؿ��γ�һ����Է�������Ϊ60��һԪ������ӣ�������й��γ�7���Ҽ���1���м���
��4��I����Ԫ���γɵ�10���ӷ���X�Ŀռ乹��Ϊ�����Σ���X����ˮ�����Һ���뵽CuSO4��Һ�����������õ��������ӵĻ�ѧʽΪ[Cu��NH3��4]2+������X��Cu2+֮������λ����ϣ�
��5����ͼ1��IԪ���γɵĺ�����Ľṹ�����Ҫ˵��������������ˮ��ԭ��
a��HNO3�Ǽ��Է��ӣ������ڼ��Ե�ˮ��
b��HNO3�����е�-OH����ˮ����֮���γ����
��6����ͼ2��J��E��ɵľ���ṹ����ÿ����J��Χ���Jԭ����ĿΪ12���������߳�Ϊa cm��������ܶ�Ϊ���ú�a��NA�Ĵ���ʽ��ʾ��$\frac{4��M��SiC��}{{a}^{3}{N}_{A}}$��$\frac{4��40}{{a}^{3}{N}_{A}}$��$\frac{160}{{a}^{3}{N}_{A}}$��g/mL��

| A�� | ����ˮ���γɵ�Al��OH��3����������ˮ�������������ˮ�ľ��� | |
| B�� | ij��ˮ��Ʒ�ɼ������һ��ʱ�䣬pH��4.68��Ϊ4.28������Ϊ��Һ�е�SO32- ����ˮ�� | |
| C�� | ������FeCl3��Һ�����ˮ�п��Ʊ�Fe��OH��3���壬���õ�������ˮ��ԭ�� | |
| D�� | ������Һ�ʼ��Ե�ԭ����CO3 2-+H2O?HCO3-+OH- |
