��Ŀ����
��������Ҫ�ĵ��ʣ��Dz������Ļ�����Ʒ֮һ���¹��˹�����1905�귢���˺ϳɰ��ķ�������ϳ�ԭ��Ϊ��N2��g��+3H2��g��?2NH3��g����H=-92.4kJ?mol-1������˻����1918��ŵ������ѧ����
���ܱ������У�ʹ2mol N2��6mol H2��Ϸ������з�Ӧ��
N2��g��+3H2��g��?2NH3��g��������ӦΪ���ȷ�Ӧ��
��1������Ӧ�ﵽƽ��ʱ��N2��H2��Ũ�ȱ��� ��N2��H2��ת���ʱ��� ��
��2������ƽ����ϵ���¶ȣ�����������䣩����������ƽ����Է������� ���ܶ� ����������С�����䡱����
��3�����ﵽƽ��ʱ�����������������ѹǿ���䣬ƽ�⽫ ����������������ƶ���
��4�����������ݡ����ȣ�����ʹ�������¶�Ѹ������ԭ����2����ƽ�⽫ ��������ƶ����������ƶ������ƶ��������ﵽ��ƽ����������¶� ������ڡ���С�ڡ����ڡ���ԭ����2����
��5������1molN2��3molH2�����ܱ������У�����Ӧ��������ѹǿΪԭ����90%����ų�������Ϊ KJ��
���ܱ������У�ʹ2mol N2��6mol H2��Ϸ������з�Ӧ��
N2��g��+3H2��g��?2NH3��g��������ӦΪ���ȷ�Ӧ��
��1������Ӧ�ﵽƽ��ʱ��N2��H2��Ũ�ȱ���
��2������ƽ����ϵ���¶ȣ�����������䣩����������ƽ����Է�������
��3�����ﵽƽ��ʱ�����������������ѹǿ���䣬ƽ�⽫
��4�����������ݡ����ȣ�����ʹ�������¶�Ѹ������ԭ����2����ƽ�⽫
��5������1molN2��3molH2�����ܱ������У�����Ӧ��������ѹǿΪԭ����90%����ų�������Ϊ
���㣺��ѧƽ��ļ���,��ѧƽ���Ӱ������
ר�⣺��ѧƽ��ר��
��������1������ĵ��������������ʵ���֮�ȵ����仯ѧ������֮�ȣ����۷�Ӧ��ʲô�̶ȣ�������������Ũ��֮����ʼ����1��3��ת����֮��1��1��
��2��ƽ����Է�������=
����=
��
��3���������������ѹǿ���䣬������
��4��������������ԭ�����
��5���������ʵ���֮�ȵ���ѹǿ֮�Ƚ������ʽ���㼴�ɣ�
��2��ƽ����Է�������=
| ���������ܺ� |
| �������ʵ����ܺ� |
| m |
| V |
��3���������������ѹǿ���䣬������
��4��������������ԭ�����
��5���������ʵ���֮�ȵ���ѹǿ֮�Ƚ������ʽ���㼴�ɣ�
���
�⣺��1������ĵ��������������ʵ���֮�ȵ����仯ѧ������֮�ȣ����۷�Ӧ��ʲô�̶ȣ��õ�����������ʾ���������ȵ��ڻ�ѧ������֮�ȣ����Ե�����������Ũ��֮����ʼ����1��3��ת����֮��1��1��
�ʴ�Ϊ��1��3��1��1��
��2�����£�ƽ�������ƶ����������ʵ��������������䣬�����ƽ����Է�������=
�����ԣ���������ƽ����Է���������С��
���������û�䣬�����������䣬�����ܶȲ��䣬
�ʴ�Ϊ����С�����䣻
��3���������������ѹǿ���䣬�������������ķ�ѹ��С��ƽ�����������������ƶ����ʴ�Ϊ������
��4�����£�ƽ�������ȵ������ƶ���ʹ��ϵ�¶Ƚ��ͣ��ʴ�Ϊ�������ƶ���С�ڣ�
��5��N2��g��+3H2��g��?2NH3��g����H=-92.4kJ?mol-1��
��ʼ����1 3 0
�仯����x 3x 2x 92.4x
ƽ������1-x 3-3x 2x 92.4x
��Ӧ��������ѹǿΪԭ����90%����
=90%�����x=0.2����ų���������92.4��0.2kJ=18.48kJ���ʴ�Ϊ��18.48��
�ʴ�Ϊ��1��3��1��1��
��2�����£�ƽ�������ƶ����������ʵ��������������䣬�����ƽ����Է�������=
| ���������ܺ� |
| �������ʵ����ܺ� |
���������û�䣬�����������䣬�����ܶȲ��䣬
�ʴ�Ϊ����С�����䣻
��3���������������ѹǿ���䣬�������������ķ�ѹ��С��ƽ�����������������ƶ����ʴ�Ϊ������
��4�����£�ƽ�������ȵ������ƶ���ʹ��ϵ�¶Ƚ��ͣ��ʴ�Ϊ�������ƶ���С�ڣ�
��5��N2��g��+3H2��g��?2NH3��g����H=-92.4kJ?mol-1��
��ʼ����1 3 0
�仯����x 3x 2x 92.4x
ƽ������1-x 3-3x 2x 92.4x
��Ӧ��������ѹǿΪԭ����90%����
| 1-x+3-3x+2x |
| 4 |
���������⿼���˷�Ӧ�����뻯ѧ�������Ĺ�ϵ�������ƽ����Է����������ܶȡ���ѧƽ����ƶ�����Ŀ�ѶȲ���

��ϰ��ϵ�д�
�����Ŀ
�������ʵ�����A��B�����2L���ܱ������У��������·�Ӧ��
3A��g��+B��g��?xC��g��+2D��g����
5minʱ���D��Ũ��Ϊ0.5mol/L��c��A����c��B��=3��5��C��ƽ����Ӧ������0.1mol/��L?min��������˵����ȷ���ǣ�������
3A��g��+B��g��?xC��g��+2D��g����
5minʱ���D��Ũ��Ϊ0.5mol/L��c��A����c��B��=3��5��C��ƽ����Ӧ������0.1mol/��L?min��������˵����ȷ���ǣ�������
| A����ʼA�����ʵ���Ϊ2mol |
| B��5minʱB��ת����Ϊ16.67% |
| C��5minʱA��Ũ��Ϊ1.5 mol/L |
| D��x=3 |
��֪�����Ȼ�ѧ����ʽ��Zn��s��+
O2��g��=ZnO��s����H=-351.1kJ?mol-1 Hg��l��+
O2��g��=HgO��s����H=-90.7kJ?mol-1�ɴ˿�֪��ӦZn��s��+HgO��s��=ZnO��s��+Hg��l�����ʱ�Ϊ��������
| 1 |
| 2 |
| 1 |
| 2 |
| A��-260.4 kJ?mol-1 |
| B��-441.8 kJ?mol-1 |
| C��260.4 kJ?mol-1 |
| D��441.8 kJ?mol-1 |
�������û�з��������ʱ��Ե��ǣ�������
| A���ø������ֽ��ƶ���걾 |
| B����պ����������Ϊ75%�ľƾ������Ƥ�� |
| C���������ߵƹ����䲡�� |
| D��������Һ�мӱ���ʳ��ˮ����� |
����������ָ���������ܴ���������ǣ�������
| A����������Һ�У�Ba2+��K+��SO42-��Cl- |
| B�����д���Al3+����Һ�У�K+��Na+��NH4+��SO42- |
| C��ʹ���ȱ�Ƶ���Һ�У�Na+��I-��CO32-��OH- |
| D����ɫ��Һ�У�K+��Cl-��Cr2O72-��HCO3- |

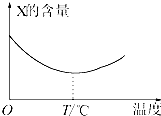 ������̬����X��Y��ֱ�ӻ�������Z�����淴Ӧ�����罫X��Y��һ��������ϲ�ѹ���ܱ������У��ڲ�ͬ�¶��¾���һ��ʱ���Ӧ�������X�ĺ����仯��ͼ��ʾ���ش��������⣺
������̬����X��Y��ֱ�ӻ�������Z�����淴Ӧ�����罫X��Y��һ��������ϲ�ѹ���ܱ������У��ڲ�ͬ�¶��¾���һ��ʱ���Ӧ�������X�ĺ����仯��ͼ��ʾ���ش��������⣺