��Ŀ����
12�� 25��ʱ���������ʵĵ���ƽ�ⳣ�������ʾ��
25��ʱ���������ʵĵ���ƽ�ⳣ�������ʾ����ش��������⣺
��1��CH3COOH�ĵ���ƽ�ⳣ������ʽ$\frac{c��C{H}_{3}CO{O}^{-}��c��{H}^{+}��}{c��C{H}_{3}COOH��}$��
��2��CH3COOH��H2CO3��HClO��������ǿ������˳��ΪCH3COOH��H2CO3��HClO��
��3�����Ϊ10mLpH=2�Ĵ�����Һ��һԪ��HX�ֱ��ˮϡ����1000mL��ϡ������pH�仯��ͼ��ʾ����HX�ĵ���ƽ�ⳣ�����ڣ�����ڡ��������ڡ���С�ڡ�������ĵ���ƽ�ⳣ����������ϡ����ͬ������HX��pH�仯��CH3COOH�Ĵ�����ǿ������ƽ�ⳣ����
���� ��1���������ƽ�ⳣ������ƽ��״̬�µ����������Ũ�ȳ˻����Է���Ũ�ȣ�
��2������ƽ�ⳣ��Խ����ĵ���̶�Խ����Һ����Խǿ��
��3����ͼ��֪��ϡ����ͬ�ı�����HX��pH�仯�̶ȴ�������HXǿ������ƽ�ⳣ����
��� �⣺��1��CH3COOH�ĵ���ƽ�⣬CH3COOH?CH3COO-+H+������ƽ�ⳣ������ʽΪ��K=$\frac{c��C{H}_{3}CO{O}^{-}��c��{H}^{+}��}{c��C{H}_{3}COOH��}$��
�ʴ�Ϊ��$\frac{c��C{H}_{3}CO{O}^{-}��c��{H}^{+}��}{c��C{H}_{3}COOH��}$��
��2�����ݱ������ݿ�֪����ĵ����ƽ�ⳣ����СΪ��CH3COOH��H2CO3��HCO3-��HClO������ƽ�ⳣ��Խ������Խǿ������������ǿ������˳��ΪΪ��CH3COOH��H2CO3��HClO��
�ʴ�Ϊ��CH3COOH��H2CO3��HClO��
��3������ͼ�����֪������ʼ��������Һ��c��H+����ͬ��c�������ᣩ��c����ǿ�ᣩ��ϡ�����н�����ĵ���̶�����������ϡ�����н������c��H+��һֱ���ڽ�ǿ���c��H+����ϡ����ͬ������HX��pH�仯��CH3COOH�Ĵ�HX����ǿ������ƽ�ⳣ����
�ʴ�Ϊ�����ڣ�ϡ����ͬ������HX��pH�仯��CH3COOH�Ĵ�����ǿ������ƽ�ⳣ����
���� ���⿼����������ʵĵ��뼰��Ӱ�죬��Ŀ�Ѷ��еȣ�����֪ʶ��϶ࡢ�ۺ��Խ�ǿ����ֿ�����ѧ���ķ������������������Ӧ����ѧ֪ʶ�����������ջ����ǽ���ؼ���

| A | B | C | D |
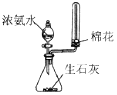 | 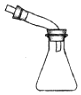 | 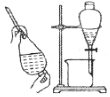 | 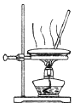 |
| �����Ʊ����ռ�һ�����İ��� | ����ʱ�Ľ���װ�� | ���ڷ���C2H5OH��ˮ����� | ֱ�������Ȼ�����Һ����Ȼ������� |
| A�� | A | B�� | B | C�� | C | D�� | D |
| A�� | 1��1 | B�� | 4��3 | C�� | 3��1 | D�� | 3��2 |
 �����ҹ�����ӵ�����ʽϿ��������ƣ�����β���ѳ�Ϊ��Ҫ�Ŀ�����Ⱦ��ش��������⣺
�����ҹ�����ӵ�����ʽϿ��������ƣ�����β���ѳ�Ϊ��Ҫ�Ŀ�����Ⱦ��ش��������⣺��1����������������ʱ������Ӧ��N2��g��+O2��g��?2NO��g�����ǵ�������β���к���NO��ԭ��֮һ��2 000 Kʱ�����ݻ�Ϊ2 L���ܱ������г���2 mol N2��2 mol O2������������Ӧ������5 min�ﵽƽ�⣬��ʱ������NO���������Ϊ0.75%����÷�Ӧ��5 min�ڵ�ƽ����Ӧ����v��O2��=0.0015mol•L-1•min-1��N2��ƽ��ת����Ϊ0.75%��2 000 Kʱ�÷�Ӧ��ƽ�ⳣ��K=2.25��10-4��
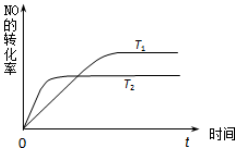
��2��һ����NO�����ֽ�Ĺ����У�NO��ת������ʱ��仯�Ĺ�ϵ��ͼ��ʾ��
�ٷ�Ӧ2NO��g��?N2��g��+O2��g��Ϊ���ȷ�Ӧ������ȡ����ȡ�����
��һ���¶��£��ܹ�˵����Ӧ2NO��g��?N2��g��+O2��g���Ѵﵽƽ�����c������ţ���
a�������ڵ�ѹǿ�������仯����������������
b�����������ܶȲ������仯
c��NO��N2��O2��Ũ�ȱ��ֲ��䡡��������
d����λʱ���ڷֽ�4mol NO��ͬʱ����2molN2
�����ĸ��ݻ����¶Ⱦ���ȫ��ͬ���ܱ������зֱ�����������ʣ���Ӧ���ʵ�����mo1�������ʾ����ͬ�����´ﵽƽ���N2���������������D�����������ţ���
| �������� | NO | N2 | O2 |
| A | 2 | 0 | 0 |
| B | 0 | 1 | 1 |
| C | 0.4 | 0.6 | 0.8 |
| D | 1 | 0.5 | 0.4 |
��֪��CH4��g��+4NO2��g���T4NO��g��+CO2��g��+2H2O��g����H=-574kJ•mol-1
CH4��g��+2NO2��g���TN2��g��+CO2��g��+2H2O��g����H=-867kJ•mol-1
��д��CH4��NO��Ӧ����N2��CO2��H2O��g�����Ȼ�ѧ����ʽ��CH4��g��+4NO��g��=2N2��g��+CO2��g��+2H2O��g����H=-1160kJ/mol
��ʹ�ô������Խ�����β������Ҫ�к��ɷ�һ����̼��CO���͵������NOx��ת��Ϊ�����壬�÷�Ӧ�Ļ�ѧ����ʽΪ2xCO+2NOx$\frac{\underline{\;����\;}}{\;}$2xCO2+N2��
| A�� | Wһ������Ԫ�� | |
| B�� | �����Ӱ뾶rz��rx��ry | |
| C�� | ��״���£�1mol Z���������������ԼΪ22.4L | |
| D�� | ��ҵ�ϳ��õ������Y���Ȼ���ķ����Ʊ�Y���� |
| A�� | �ش�����ڼ�ȼ�ŵ�������ɫ��ijЩ����Ԫ����ɫ��Ӧ�����ֳ�����ɫ�� | |
| B�� | С�մ���������ͷ������ȸ������ɼ���Ҳ������θ������һ��ҩ�� | |
| C�� | Ϊ��ֹ�±��ȸ�֬ʳƷ�����������ʣ����ڰ�װ���з�����ʯ�һ�轺 | |
| D�� | ����ʵʩ����ȼ�ϵ������������������Լ���SO2��NO2���ŷ� |