��Ŀ����
��8�֣��������˻ᡰ���ơ����ȼ���DZ��飨C3H8��������������˻���ȼ���DZ�ϩ��C3H6����
�ű�������ɵñ�ϩ��
��֪����C3H8(g) = CH4(g)��HC��CH(g)��H2(g)����H1��156.6kJ��mol-1
��CH3CH��CH2(g) = CH4(g)��HC��CH(g) ����H2��32.4kJ��mol-1
����ͬ�����£���ӦC3H8(g) = CH3CH��CH2 (g)��H2(g)�ġ�H�� kJ��mol-1��
���Ա���Ϊȼ����������ȼ�ϵ�أ���ص�����ͨ��O2��CO2������ͨ����飬�����������̼���Ρ���ط�Ӧ����ʽΪ ��
��̼�⻯������ȫȼ������CO2��H2O�����³�ѹ�£������е�CO2����ˮ���ﵽƽ��ʱ����Һ��pH=5.60��c(H2CO3)=1.5��10-5
mol��L-1��������ˮ�ĵ��뼰H2CO3�ĵڶ������룬��H2CO3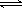 HCO3-��H+��ƽ�ⳣ��K1=
������֪��10-5.60=2.5��10-6��
HCO3-��H+��ƽ�ⳣ��K1=
������֪��10-5.60=2.5��10-6��
�ȳ����£�0.1 mol��L-1NaHCO3��Һ��pH����8������Һ��c(H2CO3) c(CO32-)(�����������������)��
��8�֣�
��124.2 ��2�֣� ��C3H8��5O2��3CO2��4H2O��2�֣� ��4.2��10-7�� mol��L-1 ����2�֣�
�ȣ���2�֣�
����������1���ɸ�˹���ɿ�֪���ɢټ�ȥ�ڿɵã�C3H8(g)== CH3CH=CH2(g)+H2(g) �ã���H=124.2kJ��mol-1��
��2��ע���ط�Ӧ��ʵ���DZ����ȼ�շ�Ӧ��ֻ��ȥ������ȼ�����������ɣ�C3H8��5O2=3CO2��4H2O
��3��K=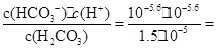 4.2��10-7mol��L-1
4.2��10-7mol��L-1
��4��NaHCO3��Һ��pH����8��˵��HCO3����ˮ��̶ȴ��ڵ���̶�:
HCO3- CO32����H��
HCO3����H2O
CO32����H��
HCO3����H2O H2CO3��OH��
H2CO3��OH��
��֪����Һ��c(H2CO3)>c(CO32-)

 HCO3-+H+��ƽ�ⳣ��K1=
HCO3-+H+��ƽ�ⳣ��K1= CO32-+H+��HCO3-+H2O
CO32-+H+��HCO3-+H2O H2CO3+OH-��
H2CO3+OH-��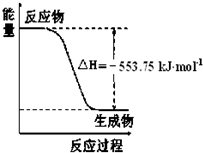 ��1����˹������Ϊ�����ܻ�ѧ������һ����ɻ��������ɣ��������̵���
��1����˹������Ϊ�����ܻ�ѧ������һ����ɻ��������ɣ��������̵���