��Ŀ����
5����һ�������������Na2CO3��Na2SO4��CuSO4��CaCl2��KCl �Ȼ����ɣ�Ϊ�˼����������������ʣ���������ʵ�飮�ٽ���������ˮ�������õ���ɫ����Һ����������Һ�еμ����ᱵ��Һ���а�ɫ�������ɣ��۹��ˣ�����������ϡ�����У����ֳ���ȫ���ܽ⣮
��1�����жϣ����������п϶�����Na2CO3���϶�û��Na2SO4��CuSO4��CaCl2�����ܺ���KCl��
��2���Կ����е����ʣ��ɲ�����ɫ��Ӧ �����飬������и����ʣ�������������ɫ�ܲ�Ƭ����dz��ɫ���森
���� ������ͭ��Һ����ɫ�ģ�����ɫ�����ˮ�õ�����ɫ��Һ���ʹ�����һ������������ͭ��
��̼�����������ᱵ��Ӧ����̼�ᱵ�������������������ᱵ��Ӧ�������ᱵ������
��̼�ᱵ���������ᣬ�����ᱵ������ϡ���ᣬ�������ʵ����ʽ��з��������ɽ��
��� �⣺��ȡ����������Ʒ����ˮ�����õ���ɫ��Һ����������ͭ��ҺΪ��ɫ��˵��һ����������ͭ��
��������Һ�еμ����ᱵ��Һ���а�ɫ�������ɣ�������ɸ��������ʣ����γɵij�������Ϊ̼�ᱵ�����ᱵ�����Ի�����п��ܺ�̼���ƻ������ƣ�Ҳ�����Ƕ��ߵĻ���
���ڰ�ɫ�����м���������ϡ���ᣬ���ֳ��������ܽ⣬̼�ᱵ���������ᣬ�����ᱵ������ϡ���ᣬ����ȫ���ܽ⣬˵������Ϊ̼�ᱵ����һ������̼���ƣ����������ƣ���̼���ƿ����Ȼ����γɳ���������Ʒ����ˮ�õ���ɫ��Һ���ʻ������һ�������Ȼ��ƣ��Ȼ����Ƿ������ȷ����
��1���������������������п϶�����Na2CO3���϶�û��CuSO4��CaCl2�����ܺ���KCl��
�ʴ�Ϊ����1��Na2CO3��Na2SO4��CuSO4��CaCl2��KCl��
��2�������Ȼ��صĴ��ڣ����Խ�����ɫ��������ɫ�ܲ�Ƭ����dz��ɫ���森
�ʴ�Ϊ����ɫ��Ӧ������ɫ�ܲ�Ƭ����dz��ɫ���森
���� ����Ϊ�����ƶ��⣬�����������ʵ����ʽ������ṩ����Ϣ���н��ץס��������г�������ȷ�Ӧ�����ǽ���ͻ�ƿڣ���Ŀ�ϼ�

 ��������һ���þ�ϵ�д�
��������һ���þ�ϵ�д� Сѧ��10����Ӧ����ϵ�д�
Сѧ��10����Ӧ����ϵ�д�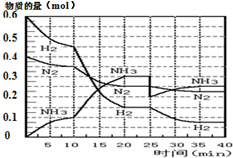 ���ĺϳ�ԭ��Ϊ��N2��g��+3H2��g��?2NH3��g������H=-92.4KJ•mol-1������500�桢20MPaʱ����N2��H2����һ���ݻ�Ϊ2L���ܱ������з�����Ӧ����Ӧ�����и����ʵ����ʵ����仯��ͼ��
���ĺϳ�ԭ��Ϊ��N2��g��+3H2��g��?2NH3��g������H=-92.4KJ•mol-1������500�桢20MPaʱ����N2��H2����һ���ݻ�Ϊ2L���ܱ������з�����Ӧ����Ӧ�����и����ʵ����ʵ����仯��ͼ���ش��������⣺
��1��10min����NH3��ʾ��ƽ����Ӧ����0.005mol/��L��min����
��2����10��20min�ڣ�NH3Ũ�ȱ仯��ԭ�������A��
A�����˴��� B����С�������
C�������¶� D������NH3���ʵ���
��3����1��ƽ���ʱ�䷶ΧΪ��20-25min����1��ƽ�⣺ƽ�ⳣ��K1=$\frac{��\frac{0.3mol}{2L}��^{2}}{��\frac{0.25mol}{2L}����{\frac{0.15mol}{2L}��}^{3}}$�������ݵı���ʽ����
��4���ڷ�Ӧ������25minʱ��
�����߷����仯��ԭ�����0.1molNH3
�ڴ�ڶ���ƽ��ʱ����ƽ���ƽ�ⳣ��K2���� K1������ڡ��������ڡ�����С�ڡ�����
��5���������¡��˹��̵������о��������ڳ��¡���ѹ�����������£�N2�ڴ�������������Fe2O3��TiO2��������ˮ�������з�Ӧ��
N2��g��+3H2O��1��?2NH3��g��+$\frac{3}{2}$O2��g������H=a kJ•mol-1
��һ���о�NH3���������¶ȵĹ�ϵ����ѹ�´ﵽƽ��ʱ��ò���ʵ���������±���
| T/K | 303 | 313 | 323 |
| NH3������/��10-6mol�� | 4.8 | 5.9 | 6.0 |
����֪��N2��g��+3H2��g��?2NH3��g����H=-92.4kJ•mol-1
2H2��g��+O2��g��=2H2O��l��=-571.6kJ•mol-1
�����µ�����ˮ��Ӧ���ɰ������������Ȼ�ѧ����ʽΪ��2N2��g��+6H2O��l��=4NH3��g��+3O2��g����H=+1536kJ•mol-1��
| A�� | v1��v2��K1��K2ͬʱ���� | B�� | v1��v2��K1��K2ͬʱ��С | ||
| C�� | v1��v2����K1����K2��С | D�� | v2��С��v1����K1����K2��С |
| A�� | һ����������ӵ�����ΪM/NA | |
| B�� | һ����������ӵ����ΪVm/NA | |
| C�� | ����Ħ������Ĵ�Сȡ����������ӱ����Ĵ�С | |
| D�� | ����˵��������ȷ |
 ��ͼ�ǿ��淴ӦX2+3Y2?2Z2 �ڷ�Ӧ�����еķ�Ӧ���ʣ�V����ʱ�䣨t���Ĺ�ϵ���ߣ�������������ȷ���ǣ�������
��ͼ�ǿ��淴ӦX2+3Y2?2Z2 �ڷ�Ӧ�����еķ�Ӧ���ʣ�V����ʱ�䣨t���Ĺ�ϵ���ߣ�������������ȷ���ǣ�������| A�� | t2ʱ����Ӧ������ | B�� | t2-t3����Ӧ���ڽ��� | ||
| C�� | t1ʱ��ֻ��������Ӧ | D�� | t2-t3�������ʵ�Ũ�Ȳ��ٷ����仯 |
| A�� | ��һ�������£���ӦCO��g��+2H2��g��?CH3OH��g����һ�ܱ������дﵽƽ�⣬�����������������������������ά��H2��Ũ�Ⱥ��������¶Ȳ��䣬ԭƽ����Ƚϴ���ƽ��ʱCO��ת���ʲ��� | |
| B�� | �����£�pH=12��NaOH��Һ��pH=2�Ĵ����Ϻ�ǡ����ȫ��Ӧ����������Һ����ı仯������Ӧ�����Һ�У�c��CH3COO-��+c��CH3COOH��=0.01mol/L | |
| C�� | pH��ͬ�Ģ�CH3COONa����NaHCO3����NaAlO2������Һ�е�c��Na+�����ۣ��ڣ��� | |
| D�� | ��֪ij�¶��£�Ksp��AgCl��=1.56��10-10��Ksp��Ag2CrO4��=1��10-12����0.001mol/L��AgNO3��Һ��ε���0.001mol/L��KCl��0.001mol/L K2CrO4��Һ�����Ȳ���Ag2CrO4���� |
| A�� | ú��ʯ�Ͷ��ǻ�����ֻ��̼��������Ԫ����� | |
| B�� | ú��������Һ�����������仯 | |
| C�� | ú�ĸ����ʯ�͵ķ������Ķ��ǻ�ѧ�仯 | |
| D�� | ��������Ȼ�̼��Һ�ɼ���ֱ�����ͺ��ѻ����� |
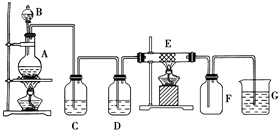 ��ͼΪ��ȡ���������Cl2����������������Ӧ��װ�ã�A��Cl2����װ�ã�C��DΪ���徻��װ�ã�EӲ�ʲ�������װ��ϸ��˿����FΪ����Ŀչ��ƿ���ձ�GΪβ������װ�ã��Իش�
��ͼΪ��ȡ���������Cl2����������������Ӧ��װ�ã�A��Cl2����װ�ã�C��DΪ���徻��װ�ã�EӲ�ʲ�������װ��ϸ��˿����FΪ����Ŀչ��ƿ���ձ�GΪβ������װ�ã��Իش�