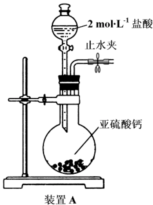
��Ŀ����
12����ʢ��100mLNH4Al��SO4��2��Һ���ձ��еμ�NaOH��Һ���������ʵ�����NaOH��Һ����仯ʾ��ͼ���£�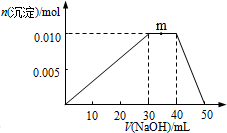
����˵����ȷ���ǣ�������
| A�� | NH4Al��SO4��2�ĵ��뷽��ʽΪ��NH4Al��SO4��2?NH4++Al3++2SO42- | |
| B�� | m�㷴Ӧ�����ӷ���ʽ��NH4++OH-�TNH3•H2O | |
| C�� | ��Ӧ������SO42-��Ũ��ʼ�ձ��ֲ��� | |
| D�� | ��ͼ���֪����NaOH��Һ��Ũ��Ϊ0.1mol•L-1 |
���� ��ʢ��100mLNH4Al��SO4��2��Һ���ձ��еμ�NaOH��Һ����0��30mL����Һ��Al3+������������������������������30mL��40mL��NH4+�����������ӷ�Ӧ����NH3•H2O��40mL��50mL��Al��OH��3�����������ӷ�Ӧ����ƫ��������ӣ�
A��NH4Al��SO4��2��ǿ����ʣ�����Һ����ȫ���룻
B��m��NH4+�����������ӷ�Ӧ����NH3•H2O��
C����������ӵ����ʵ������䣬��Һ���������
D���������ɵ�Al��OH��3�����ʵ������������ĵ��������Ƶ����ʵ�����Ũ�ȣ�
��� �⣺��ʢ��100mLNH4Al��SO4��2��Һ���ձ��еμ�NaOH��Һ����0��30mL����Һ��Al3+������������������������������30mL��40mL��NH4+�����������ӷ�Ӧ����NH3•H2O��40mL��50mL��Al��OH��3�����������ӷ�Ӧ����ƫ��������ӣ�
A��NH4Al��SO4��2��ǿ����ʣ�����Һ����ȫ���룬��NH4Al��SO4��2�ĵ��뷽��ʽΪ��NH4Al��SO4��2=NH4++Al3++2SO42-����A����
B��m��NH4+�����������ӷ�Ӧ����NH3•H2O���䷴Ӧ�����ӷ���ʽΪNH4++OH-=NH3•H2O����B��ȷ��
C����������Ӳ��μӷ�Ӧ����������ӵ����ʵ������䣬��������������Һ�ļ��룬��Һ���������������������ӵ�Ũ�ȼ�С����C����
D����ͼ��֪�����ɵ�Al��OH��3�����ʵ���Ϊ0.01mol��Al3++3OH-=Al��OH��3�������������Ƶ����ʵ���Ϊ0.03mol�������Ϊ0.03L������NaOH��Һ��Ũ��Ϊ1mol•L-1����D����
��ѡB��
���� ���⿼�����й����ӷ���ʽ�ļ��㣬��Ŀ�漰���뷽��ʽ�����ӷ�Ӧ�����ʵ���Ũ�ȵļ���ȣ���Ŀ�Ѷ��еȣ�ע�����ͼ���з�Ӧ���̣������ڿ���ѧ���ķ��������ͼ���������

 ��У����ϵ�д�
��У����ϵ�д�| A�� | ����Ƶ͵ĵط����� | |
| B�� | ��ʪë����պ���ռ�ˮ��ë����ס�ڱdz��� | |
| C�� | �۲����˳�糷�� | |
| D�� | ��ʪë����պ��ϡ����ˮ��ë����ס�ڱdz��� |
| A�� | 1molCO2������Ϊ44g/mol | |
| B�� | CO2��Ħ������Ϊ44g | |
| C�� | CO2��Ħ����������CO2����Է������� | |
| D�� | NA��CO2��������CO2����Է�����������ֵ����ͬ��NAΪ�����ӵ������� |
| A�� | ��״���£�22.4LH2SO4���е�ԭ����Ϊ7NA | |
| B�� | 16g������16g�������е���ԭ������ΪNA | |
| C�� | ���³�ѹ�£�14gN2���е�����Ϊ14NA | |
| D�� | 28gCO��22.4LCO2������̼ԭ������� |
| A�� | ˮ | B�� | ú�� | C�� | ɳ�� | D�� | ��ĭ����� |
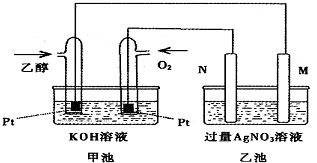 �������ؾ��ǵ绯ѧװ�ã�����ͼ���ӣ��ҳ��е������缫�ֱ���ʯī�缫�����缫����֪��������M��N�����缫�������������٣�������˵����ȷ���ǣ�������
�������ؾ��ǵ绯ѧװ�ã�����ͼ���ӣ��ҳ��е������缫�ֱ���ʯī�缫�����缫����֪��������M��N�����缫�������������٣�������˵����ȷ���ǣ�������| A�� | M�缫�IJ�����ʯī | |
| B�� | ���ҳ���ijһ�缫��������4.32gʱ�������ϼ׳���������Ϊ224mL | |
| C�� | �ڴ˹����У��׳���OH-��ͨ�Ҵ���һ���ƶ� | |
| D�� | �ڴ˹����У��ҳ���Һ�е��Ӵ�M�缫��N�缫�ƶ� |
 ��B
��B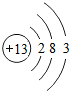 ���������Ӱ뾶��Na+��Al3+�������ӷ��ţ���
���������Ӱ뾶��Na+��Al3+�������ӷ��ţ���