��Ŀ����
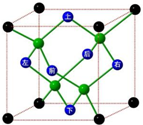 ��������ˮ���ӵĿռ����з�ʽ����ʯ�������ƣ���ͼ��
��������ˮ���ӵĿռ����з�ʽ����ʯ�������ƣ���ͼ����1��һ��ˮ���Ӻ�
��2��ÿ��������ƽ��ռ��
��3��ʵ���ñ��������������Ϊ18.5kJ/mol���������ۻ���Ϊ5.0kJ/mol����˵�����ۻ���ˮ�����
��4������������߳�Ϊa cm��������ܶ���
���㣺�����ļ���,�����ijɼ����
ר�⣺��ѧ���뾧��ṹ
��������1��ˮ��������ԭ�ӵļ۵��Ӷ���Ϊ
=4������������H-O���������������Թµ��Ӷԣ�ͭ������ˮ�γ�ˮ��ͭ���ӣ��ݴ˴��⣻
��2�����ݾ����ṹ�����þ�̯�����㾧����ռ�е�ˮ��������ˮ����֮����ڵ�������ͷ���֮���������������ʯ��̼̼֮���ǹ��ۼ����ݴ˴��⣻
��3��Һ̬ˮ�����������
��4��������=
�����ܶȣ�
| 6+2 |
| 2 |
��2�����ݾ����ṹ�����þ�̯�����㾧����ռ�е�ˮ��������ˮ����֮����ڵ�������ͷ���֮���������������ʯ��̼̼֮���ǹ��ۼ����ݴ˴��⣻
��3��Һ̬ˮ�����������
��4��������=
| m |
| V |
���
�⣺��1��ˮ��������ԭ�ӵļ۵��Ӷ���Ϊ
=4������������H-O���������������Թµ��Ӷԣ�ͭ������ˮ�γ�ˮ��ͭ����Ϊ��[Cu��H2O��4]2+��
�ʴ�Ϊ��2��[Cu��H2O��4]2+��
��2�����ݾ����ṹ�����þ�̯�����㾧����ռ�е�ˮ������Ϊ4+8��
+6��
=8��ˮ����֮����ڵ�������ͷ���֮���������������ʯ��̼̼֮���ǹ��ۼ����ʴ�Ϊ��8���ƻ�����������ͬ�����ʯ�ڻ���Ҫ�ƻ����ۼ��������ڻ���Ҫ�ƻ�������ͷ��Ӽ�����������
��3�����������������Ϊ18.5kJ?mol-1�������ۻ���Ϊ5.0kJ?mol-1��˵�����ۻ�ΪҺ̬ˮʱֻ���ƻ���һ�������������Һ̬ˮ�����������
�ʴ�Ϊ�����֣�
��4����Ϊ�ھ�������8��ˮ���ӣ����Ը�����=
���Լ����ܶ�Ϊ
g?cm-3=
g?cm-3���ʴ�Ϊ��
��
| 6+2 |
| 2 |
�ʴ�Ϊ��2��[Cu��H2O��4]2+��
��2�����ݾ����ṹ�����þ�̯�����㾧����ռ�е�ˮ������Ϊ4+8��
| 1 |
| 8 |
| 1 |
| 2 |
��3�����������������Ϊ18.5kJ?mol-1�������ۻ���Ϊ5.0kJ?mol-1��˵�����ۻ�ΪҺ̬ˮʱֻ���ƻ���һ�������������Һ̬ˮ�����������
�ʴ�Ϊ�����֣�
��4����Ϊ�ھ�������8��ˮ���ӣ����Ը�����=
| m |
| V |
| ||
| a3 |
| 144 |
| a3NA |
| 144 |
| a3NA |
������������Ҫ�����˷��Ӿ���Ľṹ������������ļ��㣬�е��Ѷȣ�����ʱҪע������������������ʵ�Ӱ�죮

��ϰ��ϵ�д�
�����Ŀ
�������ӷ���ʽ��ȷ���ǣ�������
A���ö��Ե缫��ⱥ���Ȼ�����Һ��2Cl-+2H+
| ||||
| B��̼�������Һ����������ʯ��ˮ��Ӧ��HCO3-+Ca2++OH-�TCaCO3��+H2O | ||||
| C���������е���AgNO3��Һ�������е���Ԫ�أ�Br-+Ag+�TAgBr�� | ||||
| D����������Һ��ͨ������CO2��2C6H5O-+CO2+H2O��2C6H5OH+CO32- |
 A��B��C��D�ǰ�ԭ��������С�������еĵڶ���������Ԫ�صĵ��ʣ�B��E��Ϊ��ɿ����ijɷ֣�F����ɫ��Ӧ�ʻ�ɫ����G�зǽ��������Ԫ�ص�ԭ�Ӹ�����Ϊ1��2��
A��B��C��D�ǰ�ԭ��������С�������еĵڶ���������Ԫ�صĵ��ʣ�B��E��Ϊ��ɿ����ijɷ֣�F����ɫ��Ӧ�ʻ�ɫ����G�зǽ��������Ԫ�ص�ԭ�Ӹ�����Ϊ1��2�� ��������Ԫ�أ�����A��B��C��DΪ����������Ԫ�أ�E��FΪ��������Ԫ�أ����ǵ�ԭ����������������������������Ϣ���ش����⣮
��������Ԫ�أ�����A��B��C��DΪ����������Ԫ�أ�E��FΪ��������Ԫ�أ����ǵ�ԭ����������������������������Ϣ���ش����⣮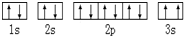 ��Υ����
������