��Ŀ����
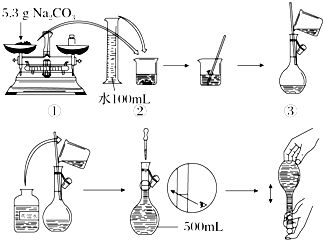 ��Na2CO3?10H2O���壬����0.2mol?L-1��Na2CO3��Һ480mL��
��Na2CO3?10H2O���壬����0.2mol?L-1��Na2CO3��Һ480mL����1��Ӧ��ȡNa2CO3?10H2O�����������
��2���������в�����������Һ��Ũ�Ȳ�����Ӱ�죬�������Ҫ��
��Na2CO3?10H2O����ʧȥ�˲��ֽᾧˮ
���á���������ij��������������壨ʹ�����룩
��̼���ƾ��岻�������л����Ȼ���
�ܳ���̼���ƾ���ʱ������������
������ƿδ������ʹ��
��������������ҺŨ��ƫ�ߵ���
��3�����в����У�����ƿ�����߱��Ĺ�����
A������һ�����ȷŨ�ȵı���Һ
B��������Һ
C����������ƿ������µ����������Һ��
D��ȷϡ��ijһŨ�ȵ���Һ
E�����������ܽ��������
��4��ijͬѧ���ù���Na2CO3��������Na2CO3��Һ�Ĺ�����ͼ��ʾ������Ϊ��ͬѧ�Ĵ�������
A.1����B.2��C.3����D.4����
���㣺����һ�����ʵ���Ũ�ȵ���Һ
ר�⣺ʵ����
��������1������n=cv��������Na2CO3�����ʵ���������Na2CO3?10H2O�����ʵ�������Na2CO3�����ʵ���������m=nM����Na2CO3?10H2O��������
��2������c=
�������������ʵ����ʵ��������Һ�������Ӱ���жϣ�
��3������ƿ������һ�����ȷŨ�ȵı���Һ�Ķ���������
��4��������Na2CO3���������������ʱӦƽ�ӿ̶��ߣ�
��2������c=
| n |
| V |
��3������ƿ������һ�����ȷŨ�ȵı���Һ�Ķ���������
��4��������Na2CO3���������������ʱӦƽ�ӿ̶��ߣ�
���
�⣺��1��ʵ������Ҫ0.2mol/L��Na2CO3��Һ480mL����������ƿ�Ĺ��û��480mL��ֻ����500mL����ƿ����500mLNa2CO3��Һ��ҪNa2CO3�����ʵ���Ϊ��0.5L��0.2mol/L=0.1mol��Na2CO3?10H2O�����ʵ���Ϊ0.1mol��Na2CO3?10H2O������Ϊ��0.1mol��286g/mol=28.6g��
�ʴ�Ϊ��28.6g��
��2����̼���ƾ���ʧȥ�˲��ֽᾧˮ�����ʵ�����ƫ�����ʵ����ʵ���ƫ����������ҺŨ��ƫ�ߣ�
���á���������ij��������������壬���ʵ�����ƫ�ͣ����ʵ����ʵ���ƫ�ͣ���������ҺŨ��ƫ�ͣ�
��̼���ƾ��岻�������л����Ȼ��ƣ����ʵ�����ƫ�ͣ����ʵ����ʵ���ƫ�ͣ���������ҺŨ��ƫ�ͣ�
�ܳ���̼���ƾ���ʱ�����������⣬���ʵ�����ƫ�����ʵ����ʵ���ƫ����������ҺŨ��ƫ�ߣ�
������ƿδ�������ʹ�ã����ʵ����ʵ�������Һ����������ı䣬����������ҺŨ����Ӱ�죻
�ʴ�Ϊ���٢ܣ��ڢۣ���
��3������ƿ��Ϊ���������������ڴ�����ܽ⣬Ҳ���ܲ��������������ݻ���Һ����������ܼ��ȣ��ʴ�Ϊ��BCE��
��4������Ҫ��Na2CO3���������m=nM=cVM=0.2mol/L��0.5L��106g/mol=10.6g��������5.3g������ʱ��Ӧ�����ӣ�������̶���ƽ�ӣ���ѡB��
�ʴ�Ϊ��28.6g��
��2����̼���ƾ���ʧȥ�˲��ֽᾧˮ�����ʵ�����ƫ�����ʵ����ʵ���ƫ����������ҺŨ��ƫ�ߣ�
���á���������ij��������������壬���ʵ�����ƫ�ͣ����ʵ����ʵ���ƫ�ͣ���������ҺŨ��ƫ�ͣ�
��̼���ƾ��岻�������л����Ȼ��ƣ����ʵ�����ƫ�ͣ����ʵ����ʵ���ƫ�ͣ���������ҺŨ��ƫ�ͣ�
�ܳ���̼���ƾ���ʱ�����������⣬���ʵ�����ƫ�����ʵ����ʵ���ƫ����������ҺŨ��ƫ�ߣ�
������ƿδ�������ʹ�ã����ʵ����ʵ�������Һ����������ı䣬����������ҺŨ����Ӱ�죻
�ʴ�Ϊ���٢ܣ��ڢۣ���
��3������ƿ��Ϊ���������������ڴ�����ܽ⣬Ҳ���ܲ��������������ݻ���Һ����������ܼ��ȣ��ʴ�Ϊ��BCE��
��4������Ҫ��Na2CO3���������m=nM=cVM=0.2mol/L��0.5L��106g/mol=10.6g��������5.3g������ʱ��Ӧ�����ӣ�������̶���ƽ�ӣ���ѡB��
���������⿼����һ�����ʵ���Ũ����Һ�����ƹ����еļ���������������ڻ�������Ŀ���ѶȲ���

��ϰ��ϵ�д�
 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д�
�����Ŀ
 773Kʱ���������ʵ�����H2O��CO����1L���ܱ������У�������Ӧ��H2O��g��+CO��g��?H2��g��+CO2��g����H��0����CO��ת������ͼ��ʾ������˵������ȷ���ǣ�������
773Kʱ���������ʵ�����H2O��CO����1L���ܱ������У�������Ӧ��H2O��g��+CO��g��?H2��g��+CO2��g����H��0����CO��ת������ͼ��ʾ������˵������ȷ���ǣ�������| A��t1ʱCO2��Ũ�ȴ���t2ʱCO2��Ũ�� |
| B��773Kʱ���÷�Ӧ�Ļ�ѧƽ�ⳣ��Ϊ0.75 |
| C��773Kʱ��������ƽ����ϵ���ٳ���CO����ѧƽ�ⳣ����С |
| D��������ƽ����ϵ���ȣ���ѧƽ�ⳣ����С |
��һС�齹̿��xgO2ͬʱ����װ��ѹ�������ܱ������У�ѹǿΪP0������һ�������³�ַ�Ӧ��ָ���ԭ�¶ȣ�ѹǿΪP1����P0��P1��2P0������뽹̿������mΪ��������
A��m��
| ||
B��m��
| ||
C��m��
| ||
D��m=
|
 ��֪��һ��̼ԭ�����������ǻ�ʱ�Զ���ˮ�γ�ȩ��������
��֪��һ��̼ԭ�����������ǻ�ʱ�Զ���ˮ�γ�ȩ�������� ����A��F�����л��������ͼ��ʾ��ת����ϵ���ݴ�����������⣺
����A��F�����л��������ͼ��ʾ��ת����ϵ���ݴ�����������⣺