��Ŀ����
15��һ���ѧ�о��ɹ�������ͭ�������CuMn2O4�����ڳ����´����������е�һ����̼�ͼ�ȩ��HCHO������1����һ�����ʵ���Ũ�ȵ�Cu��NO3��2 ��Mn��NO3��2 ��Һ�м���Na2CO3 ��Һ�����ó������������գ����Ƶ�CuMn2O4��
��д����̬Mnԭ�ӵļ۵����Ų�ʽΪ3d54S2��
��CO32-�Ŀռ乹����ƽ�������Σ���������������
��2����ͭ��������Ĵ��£�CO ������ΪCO2��HCHO ������ΪCO2 ��H2O��
�ٸ��ݵȵ�����ԭ����CO ���ӵĽṹʽΪC��O��
��CO2 ������C ԭ�ӹ�����ӻ�����Ϊsp�ӻ���
��1mol��ȩ��HCHO�������к��еĦҼ���ĿΪ3��6.02��1023������3NA����
��3����CuSO4��Һ�м������NH3�qH2O��Һ������[Cu ��NH3��4]2+�������ǿռ乹�ͣ�[Cu ��NH3��4]2+�Ľṹ����ʾ��ͼ��ʾΪ
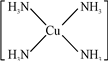 2+��
2+��
���� ��1����Mnԭ�Ӻ��������Ϊ25�����ڵ������ڢ�B�壻Cuԭ�Ӻ��������Ϊ29�������������ԭ����д��������Ų�ʽ��
�ڼ���Cԭ�Ӽ۲���Ӷ������µ��Ӷ���ȷ���ռ乹�ͣ�
��2����CO��N2��Ϊ�ȵ����壬���߽ṹ���ƣ�CO������Cԭ������ԭ��֮���γ�3�Թ��õ��Ӷԣ�
��CO2 ������Cԭ������2���Ҽ���û�йµ��Ӷԣ��ӻ������ĿΪ2��
��HCHO�����к���2��C-H����1��C=O˫���������к���3���Ҽ���
��3��[Cu ��NH3��4]2+����Cu2+��4��NH3������
��� �⣺��1����Mnԭ�Ӻ��������Ϊ25�����ڵ������ڢ�B�壬�۵����Ų�ʽΪ3d54s2���ʴ�Ϊ��3d54s2��
��CO32-��Cԭ�ӹµ��Ӷ���=$\frac{1}{2}$��4+2-2��3��=0���۲���Ӷ���=3+0=3���ռ乹��Ϊƽ�������Σ��ʴ�Ϊ��ƽ�������Σ�
��2����CO��N2��Ϊ�ȵ����壬���߽ṹ���ƣ�CO������Cԭ������ԭ��֮���γ�3�Թ��õ��Ӷԣ�CO�ṹʽΪC��O���ʴ�Ϊ��C��O��
��CO2 ������Cԭ������2���Ҽ���û�йµ��Ӷԣ��ӻ������ĿΪ2��Cԭ�Ӳ�ȡsp�ӻ����ʴ�Ϊ��sp��
��HCHO�����к���2��C-H����1��C=O˫���������к���3���Ҽ���1mol��ȩ��HCHO�������к��еĦҼ���ĿΪ3��6.02��1023����3 NA����
�ʴ�Ϊ��3��6.02��1023����3 NA����
��3��[Cu��NH3��4]2+����Cu2+��4��NH3�γ���λ������[Cu��NH3��4]2+�Ľṹ����ʾ��ͼ��ʾΪ�� 2+��
2+��
�ʴ�Ϊ 2+��
2+��
���� �����Ƕ����ʽṹ�����ʵĿ��飬�漰��������Ų����ӻ���ʽ��ռ乹���жϡ��ȵ����塢��ѧ���������ȣ�ע��Ի���֪ʶ���������գ�

| A�� | ��ѧ������ʹ�������ϣ�Ҳ����ʹԭ������ | |
| B�� | �ǽ���Ԫ�ص�ԭ��֮��ֻ���γɹ��ۼ� | |
| C�� | ��ѧ��Ӧ�����У���Ӧ������ڵĻ�ѧ�����ѣ���������л�ѧ���γ� | |
| D�� | ��ѧ����һ�������� |
| A�� | C��Mg��Li | B�� | Li��C��Mg | C�� | C��Si��Mg | D�� | C��O��Mg |
| ѡ�� | ʵ����� | ʵ��Ŀ�� |
| A | ���ȵ�NaOH��Һ�е��뱥��FeCl3��Һ | �Ʊ�Fe��OH��3���� |
| B | ��SO2ͨ��KMnO4��Һ | ��֤SO2��Ư���� |
| C | ������Fe3+��MgCl2��Һ�м�������MgCO3��ĩ�����ȡ����貢���� | ��ȥMgCl2��Һ��������Fe3+ |
| D | ��0.1mol•L-1��Na2SO4��Һ����BaCl2��Һ�������г����������ٵμ�0.1mol•L-1��Na2CO3��Һ | �Ƚ�BaCO3��BaSO4�ܶȻ��Ĵ�С |
| A�� | A | B�� | B | C�� | C | D�� | D |
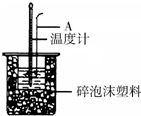 ������ͼװ�òⶨ�к��ȵ�ʵ�鲽�����£�
������ͼװ�òⶨ�к��ȵ�ʵ�鲽�����£�����ȡ50mL 0.25mol/L H2SO4��Һ����С�ձ��У������¶ȣ�
����ȡ50mL 0.55mol/L NaOH��Һ�������¶ȣ�
�۽�NaOH��Һ����С�ձ��У���Ͼ��Ⱥ�������Һ�¶ȣ���ش�
��1����ͼ��ʾ������A�������ǻ��β����������
��2��NaOH��Һ�Թ�����ԭ��ȷ�����ᱻ��ȫ�кͣ�
��3������NaOH��Һ����ȷ������B������ĸ����
A���ز������������� B��һ��Ѹ�ټ��� C�������μ���
��4��ʹ������NaOH��Һ��Ͼ��ȵ���ȷ�������û��β��������������
��5������Һ���ܶȾ�Ϊ1g•cm-3���кͺ���Һ�ı�����c=4.18J•��g•�棩-1�������ʵ������д�����к��ȵ��Ȼ�ѧ����ʽH2SO4��aq��+2NaOH��aq��Na2SO4��aq��+2H2O��l����H=-113.7kJ•mol-1
| �¶� ʵ����� | ��ʼ�¶�t1/�� | ��ֹ�¶� t2/�� | �¶Ȳ�ƽ��ֵ ��t2-t1��/�� | ||
| H2SO4 | NaOH | ƽ��ֵ | |||
| 1 | 25.0 | 25.2 | 28.5 | ||
| 2 | 24.9 | 25.1 | 28.3 | ||
| 3 | 25.5 | 26.5 | 31.8 | ||
| 4 | 25.6 | 25.4 | 29.0 | ||
a��ʵ��װ�ñ��¡�����Ч����
b���ֶ�ΰ�NaOH��Һ����ʢ�������С�ձ���
c�����¶ȼƲⶨNaOH��Һ��ʼ�¶Ⱥ�ֱ�ӲⶨH2SO4��Һ���¶�
��7����������ȷ����ȡ���Һ������¶ȣ����϶�ȡ�¶����ݣ�����¼��ֱ�������½���ȡ���ֵ��
| A�� | ����ʧȥ�ĵ���������� | |
| B�� | ��������С�ĵ���������� | |
| C�� | 2p���������������2s����������� | |
| D�� | ���������������˶��ĵ���������� |
| A�� | CO2��H2O2 | B�� | C2H4��CH4 | C�� | C60��C2H4 | D�� | NH3��HCl |
| A�� | ԭ������A��B | B�� | ��������A-��B2+ | C�� | ���Ӱ뾶A-��B2+ | D�� | ԭ�Ӱ뾶A��B |
| A�� | ���ۡ���ά�ء���֬�����ڸ߷��ӻ����� | |
| B�� | ����ζ�����ʲ�һ���������� | |
| C�� | ��Ȼ������ˮ������ղ����Ϊ������ | |
| D�� | ��֬ˮ��õ��Ĵ��DZ����� |