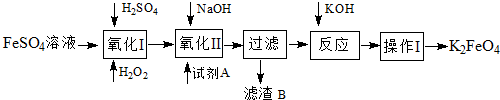
��Ŀ����
17��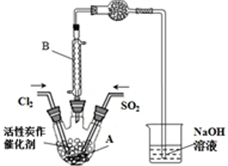 ��֪��SO2��g��+Cl2��g��+SCl2��g��?2SOCl2��g����H=-a kJ•mol-1
��֪��SO2��g��+Cl2��g��+SCl2��g��?2SOCl2��g����H=-a kJ•mol-1SO2Cl2��l��+SCl2��g��?2SOCl2��g����H=��b kJ•mol-1
��1����Ӧ��SO2��g��+Cl2��g��+SO2?Cl2��l����H=��-a-b��kJ•mol-1���ú�a��b�Ĵ���ʽ��ʾ�����÷�Ӧƽ�ⳣ������ʽΪK=$\frac{c��S{O}_{2}C{l}_{2}��}{c��S{O}_{2}��c��C{l}_{2}��}$
��2�������ȣ�SO2Cl2����һ���л��Ȼ�����Ҳ��﮵�������������ʣ�
ijѧϰС�����ݷ�Ӧ��SO2��g��+Cl2��g��?SO2Cl2��g��������Ʊ������ȵ�װ����ͼ���й���Ϣ�����ʾ��
| SO2Cl2 | Cl2 | SO2 | |
| �۵�/�� | -54.1 | -101 | -72.4 |
| �е�/�� | 69.1 | -34.6 | -10 |
| ���� | ��ˮ��������ˮ�� | / | / |
������Ũ������������Ϊԭ����ȡCl2����Ӧ�����ӷ���ʽΪ2MnO4-+16H++10Cl-=2Mn2++5Cl2��+8H2O��
��Ϊ�˱��ڻ����ķ�������߷�Ӧ���ת���ʣ�Aװ�õķ�Ӧ�������ѡ��B��ʵ�������A�л������뿪����÷�����G��
A������ B����ˮԡ C��������69.1��D����ˮԡ
E������ F����ȡ G������ H���ؽᾧ
�ܷ����������õ�SO2Cl2�м�ˮ�����ְ����������õõ���ɫ��Һ��
������SO2Cl2��H2O��Ӧ���ڷ�������ԭ��Ӧ��дSO2Cl2��NaOH��Һ��Ӧ�Ļ�ѧ����ʽSO2Cl2+4NaOH=Na2SO4+2NaCl+2H2O
������Ӧ�����ĵ����������Ϊ896mL����ת��Ϊ��״����SO2�����������õ������Ļ�����3.3g��������ȵIJ���Ϊ61.1%��������λ��Ч���֣���
���� ��1���ɢ�SO2��g��+Cl2��g��+SCl2��g��?2SOCl2��g����H=-a kJ•mol-1
��SO2Cl2��l��+SCl2��g��?2SOCl2��g����H=��b kJ•mol-1
��ϸ�˹���ɿ�֪����-�ڵõ�SO2��g��+Cl2��g��?SO2Cl2��g����KΪ������Ũ����֮���뷴Ӧ��Ũ����֮���ıȣ�
��2����BΪ�����ܣ�ͼ������װ�ã���֪�÷�ӦΪ���ȷ�Ӧ������ܿɷ�ֹˮ��������A�У�
��Ũ������������Ϊԭ����ȡCl2�������Ȼ��ء��Ȼ��̡�������ˮ��
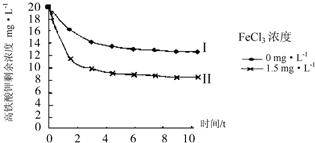
��Ϊ���ȷ�Ӧ�������¶�ƽ�������ƶ�����ϱ������ݿ�֪�������е㲻ͬ��
��SO2Cl2��NaOH��Һ��Ӧ��Ϊ��������ԭ��Ӧ�����������ơ��Ȼ��ƺ�ˮ��
��n��Cl2��=$\frac{0.896L}{22.4L/mol}$=0.04mol����SO2��g��+Cl2��g��?SO2Cl2��g����֪����������SO2Cl2Ϊ0.04mol��135g/mol=5.4g���Դ˼�����ʣ�
��� �⣺��1���ɢ�SO2��g��+Cl2��g��+SCl2��g��?2SOCl2��g����H=-a kJ•mol-1
��SO2Cl2��l��+SCl2��g��?2SOCl2��g����H=��b kJ•mol-1
��ϸ�˹���ɿ�֪����-�ڵõ�SO2��g��+Cl2��g��?SO2Cl2��g����
���H=��-a-b��kJ•mol-1���÷�Ӧƽ�ⳣ������ʽΪK=$\frac{c��S{O}_{2}C{l}_{2}��}{c��S{O}_{2}��c��C{l}_{2}��}$��
�ʴ�Ϊ����-a-b��kJ•mol-1��$\frac{c��S{O}_{2}C{l}_{2}��}{c��S{O}_{2}��c��C{l}_{2}��}$��
��2����B�����ڱ�ʵ���е�����������SO2Cl2����ʹ�ӷ��IJ���SO2Cl2��������ͼ������װ�ã���Ӧ�ɳ�����������֪�÷�ӦΪ���ȷ�Ӧ������ܵ������Ƿ�ֹˮ��������A��SO2Cl2����ˮ�ⷴӦ������SO2��Cl2���壬��ֹ��Ⱦ������
�ʴ�Ϊ������SO2Cl2����ʹ�ӷ��IJ���SO2Cl2���������ţ���ֹˮ��������A��SO2Cl2����ˮ�ⷴӦ������SO2��Cl2���壬��ֹ��Ⱦ������
����Ũ������������Ϊԭ����ȡCl2����Ӧ�����ӷ���ʽΪ2MnO4-+16H++10Cl-=2Mn2++5Cl2��+8H2O��
�ʴ�Ϊ��2MnO4-+16H++10Cl-=2Mn2++5Cl2��+8H2O��
��Ϊ���ȷ�Ӧ�������¶�ƽ�������ƶ�����ѡ���ˮԡ����ϱ������ݿ�֪�������е㲻ͬ����ѡ��������룬�ʴ�Ϊ��B��G��
��SO2Cl2��NaOH��Һ��Ӧ�Ļ�ѧ����ʽΪSO2Cl2+4NaOH=Na2SO4+2NaCl+2H2O���ʴ�Ϊ��SO2Cl2+4NaOH=Na2SO4+2NaCl+2H2O��
��n��Cl2��=$\frac{0.896L}{22.4L/mol}$=0.04mol����SO2��g��+Cl2��g��?SO2Cl2��g����֪����������SO2Cl2Ϊ0.04mol��135g/mol=5.4g�����õ������Ļ�����3.3g��������ȵIJ���Ϊ$\frac{3.3g}{5.4g}$��100%=61.1%���ʴ�Ϊ��61.1%��
���� ���⿼�����ʵ��Ʊ�ʵ�鼰��ѧƽ����㣬Ϊ��Ƶ���㣬���ո�˹����Ӧ�á��Ʊ�ԭ��������Ӧ�á���������Ϊ���Ĺؼ������ط�����ʵ�������Ŀ��飬ע��������ԭ��Ӧ��Ӧ�ã���Ŀ�ѶȲ���

1mol���л���������������������Һ��ϲ����ȣ���ַ�Ӧ�����������������Ƶ����ʵ���Ϊa�������Ǵ��ǻ����������������Ƶķ�Ӧ����ͬ������Һ���ɵõ��Ĺ���������������ĸ����ʯ�ҹ��ȣ��������������Ƶ����ʵ���Ϊb����a��b�ֱ��ǣ�������
��֪��RCH2COONa+NaOH��CaO��$\stackrel{��}{��}$RCH3+Na2CO3��
| A�� | 5mol��10mol | B�� | 6mol��2mol | C�� | 8mol��4mol | D�� | 8mol��2mol |
| A�� | Na+��Mg2+��Cl-��SO42- | B�� | Cu2+��Cl-��NO3-��OH- | ||
| C�� | Ca2+��Na+��CO32-��NO3- | D�� | K+��Fe3+��NO3-��SCN- |
��1��д��Fԭ�ӵ���Χ�����Ų�ʽ3d64s2��
| Ԫ �� | M | F | |
| ������ ��kJ•mol-1�� | I1 | 717 | 759 |
| I2 | 1509 | 1561 | |
| I3 | 3248 | 2957 | |
��3��F��M��������Ϊ25����Ԫ�صIJ��ֵ��������������ұ����Ƚ���Ԫ�ص�I2��I3��֪����̬M2+��ʧȥһ�����ӱ���̬F2+��ʧȥһ�������ѣ��Դˣ���Ľ����ǣ�Mn2+��3d��������Ų�Ϊ����״̬���ȶ���
��4��H2S��CԪ�ص��⻯�����ʽΪH2C2������Ҫ�������ʱȽ����£�
| �۵�/K | �е�/K | ��״��ʱ��ˮ�е��ܽ�� | |
| H2S | 187 | 202 | 2.6 |
| H2C2 | 272 | 423 | ������Ȼ��� |
��5����ͼa��b�ֱ�ΪM��F����Ķѻ�ģ�ͣ�����M��ԭ�Ӱ뾶�ֱ�ΪRanm�����Ŀռ������ʣ������û���M$\frac{��}{3\sqrt{2}}$��

| A�� | �ڷŵ�ʱ�����������ķ�Ӧ��Pb+SO42-�TPbSO4+2e- | |
| B�� | �ڷŵ�ʱ���õ�صĸ���������Ǧ�� | |
| C�� | �ڳ��ʱ������������Ũ�Ȳ��ϱ�С | |
| D�� | �ڳ��ʱ�����������ķ�Ӧ��PbSO4+2e-�TPb+SO42- |
| A�� | 1 mol CO2��������44g | |
| B�� | 17 g NH3�������22.4 L | |
| C�� | 100 mL 1 mol/L NaOH��Һ�к���1 mol OH- | |
| D�� | ��״���£�11.2 L O2�к���6.02��1023�������� |