��Ŀ����
�������й�ʵ������У��������� ������ţ���
A����������ƽ��ȡ11.70gʳ��
B������Ͳ��ȡ12.36mL����
C������ʽ�ζ�����ȡ21.20mL 0.10mol/L��H2SO4��Һ
D����200mL����ƿ����500mL 0.1mol/L��NaCl��Һ
E���ⶨ��Һ��pHʱ���ýྻ������IJ�����պȡ��Һ������������ˮ��ʪ����pH��ֽ�ϣ��������ɫ���Ա�
���������ʵ���Ũ��Ϊa mol/L�ı�����ȥ�ⶨV mL NaOH��Һ�����ʵ���Ũ�ȣ�����д���пհף�
��1����ʽ�ζ���������ˮϴ����Ӧ�ý��еIJ����� ��
��2����ͼ1����ʽ�ζ�����Һ���ڵζ�ǰ��Ķ�����c��NaOH��= mol/L
��3�����ڵζ�ǰ�ζ��ܼ��첿���������ݣ��ζ���ζ��ܼ��첿��������ʧ����ⶨ��NaOH���ʵ���Ũ�� ���ƫ��ƫС������Ӱ�족����
����ͼ2���������������Ƶĵζ�����a��b���������������գ�
��4������a���� ��Һ�ζ� ��Һ��
��5��P�������Ϊ ����������ʵ���Ũ��Ϊ mol/L��
A����������ƽ��ȡ11.70gʳ��
B������Ͳ��ȡ12.36mL����
C������ʽ�ζ�����ȡ21.20mL 0.10mol/L��H2SO4��Һ
D����200mL����ƿ����500mL 0.1mol/L��NaCl��Һ
E���ⶨ��Һ��pHʱ���ýྻ������IJ�����պȡ��Һ������������ˮ��ʪ����pH��ֽ�ϣ��������ɫ���Ա�
���������ʵ���Ũ��Ϊa mol/L�ı�����ȥ�ⶨV mL NaOH��Һ�����ʵ���Ũ�ȣ�����д���пհף�

��1����ʽ�ζ���������ˮϴ����Ӧ�ý��еIJ�����
��2����ͼ1����ʽ�ζ�����Һ���ڵζ�ǰ��Ķ�����c��NaOH��=
��3�����ڵζ�ǰ�ζ��ܼ��첿���������ݣ��ζ���ζ��ܼ��첿��������ʧ����ⶨ��NaOH���ʵ���Ũ��
����ͼ2���������������Ƶĵζ�����a��b���������������գ�
��4������a����
��5��P�������Ϊ
���㣺�к͵ζ�
ר�⣺ʵ����
��������������ƽ����Ͳ��ֻ�ܹ���ȷ��0.1���ζ����ܹ���ȷ��0.01������һ�����ʵ���Ũ�ȵ���ҺҪѡ����ʵ�����ƿ��pH��ֽ������ʪ��
��1���ζ���ʹ��ǰӦ��ϴ��
��2���к͵ζ��������NaOH�����ʵ�����ȣ�
��3�����ݹ�ϵʽc�����⣩��V�����⣩=c��������V��������
��4������a��pH���ߣ��ҿ�ʼpH=1����Ϊ��ζ������b��pH���ͣ��ҿ�ʼpH=13����Ϊ��ζ��
��5��P��ʱpH=7����Һ�����ԣ���pH=1�����������Ũ�ȣ�
��1���ζ���ʹ��ǰӦ��ϴ��
��2���к͵ζ��������NaOH�����ʵ�����ȣ�
��3�����ݹ�ϵʽc�����⣩��V�����⣩=c��������V��������
��4������a��pH���ߣ��ҿ�ʼpH=1����Ϊ��ζ������b��pH���ͣ��ҿ�ʼpH=13����Ϊ��ζ��
��5��P��ʱpH=7����Һ�����ԣ���pH=1�����������Ũ�ȣ�
���
�⣺��A��������ƽֻ�ܹ���ȷ��0.1g����A����
B����Ͳ��ֻ�ܹ���ȷ��0.1mL����B����
C���ζ����ܹ���ȷ��0.01mL������ȡ������ʽ�ζ��ܣ���C��ȷ��
D������500mL ��ҺӦ��ʹ��500mL������ƿ����D����
E��pH��ֽ����������ˮ��ʪ��������ˮ��ʪ�൱�ڸ���Һϡ�ͣ���E����
�ʴ�Ϊ��C��
��1���ζ���ʹ��ǰӦ�ô���Һ��ϴ���ʴ�Ϊ���ñ�������ϴ2-3�Σ�
��2���к͵ζ��������NaOH�����ʵ�����ȣ���V2-V1��a=c ��NaOH��V���ʴ�Ϊ��
��
��3���ζ�ǰ��ʽ�ζ��ܼ��첿�������ݣ��ζ�����첿�ֳ�����Һ���ᵼ���������ƫ�������ƫ�ʴ�Ϊ��ƫ��
��4����ʼpH=1����pH=-lgc��H+������c��HCl��=0.1mol/L������a��pH���ߣ���pH=1����ΪNaOH�ζ�HCl���ʴ�Ϊ��NaOH��HCl��
��5������b��pH���ͣ��ҿ�ʼpH=13����ΪHCl�ζ�NaOH��P��ʱpH=7����Һ�����ԣ���P������Ϊ��15��7�����ʴ�Ϊ����15��7����0.1��
B����Ͳ��ֻ�ܹ���ȷ��0.1mL����B����
C���ζ����ܹ���ȷ��0.01mL������ȡ������ʽ�ζ��ܣ���C��ȷ��
D������500mL ��ҺӦ��ʹ��500mL������ƿ����D����
E��pH��ֽ����������ˮ��ʪ��������ˮ��ʪ�൱�ڸ���Һϡ�ͣ���E����
�ʴ�Ϊ��C��
��1���ζ���ʹ��ǰӦ�ô���Һ��ϴ���ʴ�Ϊ���ñ�������ϴ2-3�Σ�
��2���к͵ζ��������NaOH�����ʵ�����ȣ���V2-V1��a=c ��NaOH��V���ʴ�Ϊ��
| a(V2-V1) |
| V |
��3���ζ�ǰ��ʽ�ζ��ܼ��첿�������ݣ��ζ�����첿�ֳ�����Һ���ᵼ���������ƫ�������ƫ�ʴ�Ϊ��ƫ��
��4����ʼpH=1����pH=-lgc��H+������c��HCl��=0.1mol/L������a��pH���ߣ���pH=1����ΪNaOH�ζ�HCl���ʴ�Ϊ��NaOH��HCl��
��5������b��pH���ͣ��ҿ�ʼpH=13����ΪHCl�ζ�NaOH��P��ʱpH=7����Һ�����ԣ���P������Ϊ��15��7�����ʴ�Ϊ����15��7����0.1��
���������⿼������к͵ζ�ʵ�飬��Ŀ�ѶȲ�����ȷʵ���ԭ�������衢�����Լ�ע�����ʵ������������ɽ��ע��ͼ��������к͵Ĺ�ϵ��P��pH=7Ϊ�����״��㣮

��ϰ��ϵ�д�
 ��ս�п�����ϵ�д�
��ս�п�����ϵ�д�
�����Ŀ
2007��10��24�գ��ҹ������з��ĵ�һ��̽�����ǡ��϶�һ�š�˳�����գ����϶�һ�š�����Ҫ����֮һ��̽��������
He�����ݿ�ѧ��Ԥ�⣬�����������������������ֵ�
He��ÿ�ٶ�
He�˾۱����ͷų��������൱��Ŀǰ����һ�����ĵ����������й���
He��˵���У�����ȷ���ǣ�������
| 3 |
| 2 |
| 3 |
| 2 |
| 3 |
| 2 |
| 3 |
| 2 |
| A��������Ϊ1 |
| B��������Ϊ2 |
| C��������Ϊ3 |
D��ԭ�ӽṹʾ��ͼ�� |
��VL����MgSO4��K2SO4�Ļ����Һ�ֳ����ȷݣ�һ�ݼ��뺬amolNaOH����Һ��ǡ��ʹþ������ȫ����Ϊ������þ����һ�ݼ��뺬bmolBaCl2����Һ��ǡ��ʹ�����������ȫ����Ϊ���������ԭ�����Һ�м����ӵ�Ũ��Ϊ��������
A��
| ||
B��
| ||
C��
| ||
D��
|
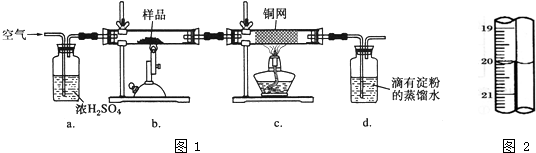

 ��ͼ��ijѧУʵ���Ҵӻ�ѧ�Լ��̵���ص�Ũ�����Լ���ǩ�ϵIJ������ݣ����ø�Ũ��������200mL 1.0mol/L��ϡ���ᣮ�ɹ�ѡ�õ������У�
��ͼ��ijѧУʵ���Ҵӻ�ѧ�Լ��̵���ص�Ũ�����Լ���ǩ�ϵIJ������ݣ����ø�Ũ��������200mL 1.0mol/L��ϡ���ᣮ�ɹ�ѡ�õ������У�