��Ŀ����
10��ij��ѧ��ȤС��������ͼװ�ý������������ϳɺͷ����ʵ��̽������ش���������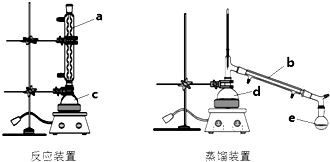
��1��д���ϳ����������Ļ�ѧ����ʽCH3COOH+CH3CH2OH
 CH3COOCH2CH3+H2O��
CH3COOCH2CH3+H2O����2������b�����������ܣ�ͼ���������������õ���a����a��b��c��d��e����
��3��Ϊ��������������IJ��ʿɲ�ȡ�Ĵ�ʩ��CH3COOCH2CH3��ʱ������룻��Ӧ�¶Ȳ��˹��ߣ�����CH3COOHCH3CH2OH�Ļӷ�����Ӧ�������ˮ������Ũ�������ˮ����������ƽ��������Ӧ�����ƶ�����߲���
��4������0.5h���ȷ�Ӧ����Ӧװ��c�дֲ�Ʒת����d�н�������
| ���� | 98.3%Ũ���� | �������� | ���� | �Ҵ� | ���� | ˮ |
| �е� | 338�棬 | 77.1�� | 118�� | 78.5�� | 34.6�� | 100�� |
���� ��1���Ҵ������ᷴӦ��������������ˮ��
��2����ͼ��֪���������ƣ�ͼ�������������������������ã�
��3��������������IJ��ʣ���ʹƽ�������ƶ���
��4���ɱ����е����ݿ�֪���Ҵ������������ķе�ӽ���
��� �⣺��1���ϳ����������Ļ�ѧ����ʽΪCH3COOH+CH3CH2OH CH3COOCH2CH3+H2O��
CH3COOCH2CH3+H2O��
�ʴ�Ϊ��CH3COOH+CH3CH2OH CH3COOCH2CH3+H2O��
CH3COOCH2CH3+H2O��
��2������b�����������ܣ�ͼ���������������õ���a���ʴ�Ϊ�������ܣ�a��
��3��������������IJ��ʿɲ�ȡ�Ĵ�ʩΪ��CH3COOCH2CH3��ʱ������룻��Ӧ�¶Ȳ��˹��ߣ�����CH3COOH CH3CH2OH�Ļӷ�����Ӧ�������ˮ������Ũ�������ˮ����������ƽ��������Ӧ�����ƶ�����߲��ʣ�
�ʴ�Ϊ����CH3COOCH2CH3��ʱ������룻��Ӧ�¶Ȳ��˹��ߣ�����CH3COOH CH3CH2OH�Ļӷ�����Ӧ�������ˮ������Ũ�������ˮ����������ƽ��������Ӧ�����ƶ�����߲��ʣ�
��4���ɱ����е����ݿ�֪���Ҵ������������ķе�ӽ����������õ������������У����п��ܺ���CH3CH2OH ���ʣ��ʴ�Ϊ��CH3CH2OH��
���� ���⿼����������ᴿ��ʵ����ƣ�Ϊ��Ƶ���㣬�����л���ĺϳ�ʵ�顢���������ᴿ������ƽ���ƶ�Ϊ���Ĺؼ������ط�����Ӧ�������Ŀ��飬��Ŀ�ѶȲ���

| A�� | ŨH2SO4�������ԣ�ϡH2SO4�������� | |
| B�� | ����ŨH2SO4������ˮ�ԣ����Կ���������� | |
| C�� | ϡH2SO4��ͭ����Ӧ����ŨH2SO4�ڼ��������¿���ͭ��Ӧ | |
| D�� | Ũ����������������ۻ� |
| A�� | �ŵ�ʱ����Һ��H+��PbO2�缫Ǩ�� | |
| B�� | �ŵ�ʱ����·��ת��0.2mol����ʱPb�缫������С20.7g | |
| C�� | ���ʱ���������Һ��pH���� | |
| D�� | ���ʱ�������ĵ缫��ӦʽΪ��PbSO4+2H2O-2e-=PbO2+4H++SO42- |
| A�� | ���� | B�� | ����ˮ����� | C�� | ���������� | D�� | ˮ�� |
| A�� | Cl-�Ľṹʾ��ͼ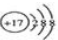 | B�� | �Ȼ�����ӵĵ���ʽ��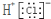 | ||
| C�� | ̼-12ԭ��${\;}_{5}^{12}C$ | D�� | HClO�ĽṹʽH-O-Cl |
| A�� | MgO��SiC | B�� | Fe��S | C�� | CO2 ��SiO2 | D�� | ��ɱ����� |
| Ԫ�ش��� | �����Ϣ |
| T | T �ĵ���������ˮ���ҷ�Ӧ������ǿ������Һ�к������ֵ�������ͬ������������ |
| X | X ��ԭ�����������������ڲ������������ |
| Y | �ڵ������ڽ���Ԫ���У�Y �ļ����Ӱ뾶��С |
| Z | T��X��Z ��ɵ� 36 ���ӵĻ����� A �Ǽ�������������Ҫ�ɷ� |
��1������ Z Ԫ�������ڱ��е�λ�õ������ڵڢ�A �壺
��2��T��X��Y Ԫ�ؼ����Ӱ뾶�ɴ�С��˳��Ϊ��O2-��Na+��Al3+�������ӷ��ţ�
��3����֪ 1mol ������ A �� Na2SO3����Һ�з�����Ӧʱ��ת�� 2mol ���ӣ�д���÷�Ӧ�����ӷ���ʽClO-+SO32-=Cl-+SO42-��
��4����������Ԫ�ص���̬ԭ��ʧȥ�����һ������������������ΪE����ͼ��ʾ��

��ͬ�����ڣ�����ԭ������������Eֵ�仯��������������ԭ�������ĵ�����E ֵ����
�ڸ�����ͼ�ṩ����Ϣ�����ƶ�E����E�����������������=������
