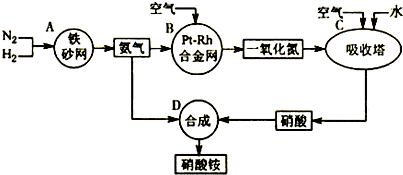
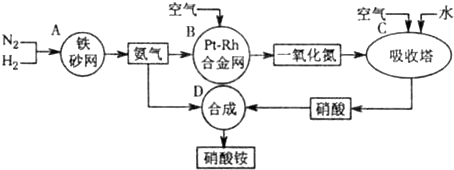
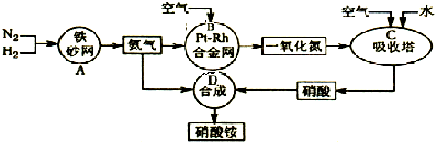
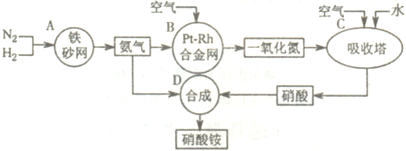
��Ŀ����
��ҵ��������淋�����ͼ����ͼ����ش�
��1����֪ ��N2(g)+3H2(g)![]() 2NH3(g) ��H����92 kJ?mol��1��
2NH3(g) ��H����92 kJ?mol��1��
����500�桢2.02��107Pa��������������һ�ܱ������г���1molN2��3molH2,��ַ�Ӧ�ų�������______(�<����>����=��)92.4kJ��������________ _________��
��Ϊ��Ч���������ת���ʣ�ʵ���������˲�ȡ�Ĵ�ʩ��____________
A.�����¶� B.���ʺϴ������Ե��ʵ����� C.����ѹǿ
D.����ѹǿ E.ѭ�����úͲ��ϲ��䵪�� F.��ʱ�Ƴ���
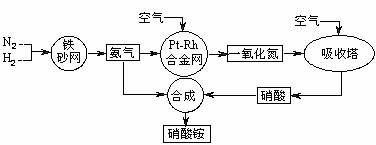
��2�� ��֪����Ͻ���δԤ��Ҳ�ᷢ�ȡ�д�����������Ļ�ѧ����ʽ��_____________________________________________ _____________________��
�÷�Ӧ�Ļ�ѧƽ�ⳣ������ʽK=___________�����¶�����ʱ��Kֵ____������С����Ӱ�죩��
��1���٣� ��CEF
��2��4NH3+5O2![]() 4NO+6H2O K=
4NO+6H2O K=![]() ��С
��С

��ϰ��ϵ�д�
�����Ŀ