��Ŀ����
ij�л���X�Ľṹ��ʽΪ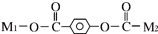 ������M1��M2����δ֪���ֵĽṹ��������һ�������¿��Է�������ת����
������M1��M2����δ֪���ֵĽṹ��������һ�������¿��Է�������ת����
��֪��RCOOH
RCH2OH��RX
RCOOH ��ش��������⣺
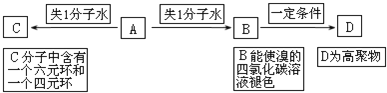
��1��A�ķ���ʽΪ ��C��D�ķ�Ӧ����Ϊ ��
��2��B������������Һ��Ӧ�Ļ�ѧ����ʽΪ ��
��3��д��C���п��ܵĽṹ��ʽ ��
��4��C�����з�Ӧ�ɵõ�H������ʽΪC6H8O4�������ں���Ԫ������
��ȷ��C�Ľṹ��ʽΪ ����ӦG��H�Ļ�ѧ����ʽΪ ��
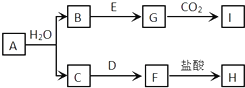
��E�ж���ͬ���칹�壬��д����ͬʱ��������Ҫ���E��ͬ���칹�壺
��������������E��ȫ��ͬ��
��1H�˴Ź�������ʾ��3����ڲ�ͬ�Ļ�ѧ������ ��
 ������M1��M2����δ֪���ֵĽṹ��������һ�������¿��Է�������ת����
������M1��M2����δ֪���ֵĽṹ��������һ�������¿��Է�������ת����
��֪��RCOOH
| ��ԭ |
| ��NaCN |
| ��H3O+ |
��1��A�ķ���ʽΪ
��2��B������������Һ��Ӧ�Ļ�ѧ����ʽΪ
��3��д��C���п��ܵĽṹ��ʽ
��4��C�����з�Ӧ�ɵõ�H������ʽΪC6H8O4�������ں���Ԫ������

��ȷ��C�Ľṹ��ʽΪ
��E�ж���ͬ���칹�壬��д����ͬʱ��������Ҫ���E��ͬ���칹�壺
��������������E��ȫ��ͬ��
��1H�˴Ź�������ʾ��3����ڲ�ͬ�Ļ�ѧ������
���㣺�л���ĺϳ�
ר�⣺�л���Ļ�ѧ���ʼ��ƶ�
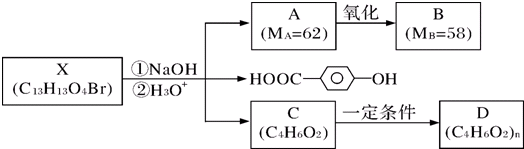
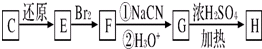
�������л���X�ķ���ʽΪC13H13O4Br�������Ͷ�Ϊ
=7�������ṹ��ʽ ����֪M1��M2�����к��в����Ͷ�Ϊ1�Ļ��ţ���C��D�ķ���ʽ��֪��Ӧ�Ƿ����Ӿ۷�Ӧ����C����̼̼˫�����Ȼ�������֪AΪ��Ԫ����Դ������ˮ����-Br��ˮ�⣬Aת������B��-CH2OHת��Ϊ-CHO����Է���������С4��Ҳ֤��A����2���ǻ���A����Է�������Ϊ62��ȥ��2��-OH��ʣ�������ԭ������Ϊ62-34=28��ֻ��Ϊ2��Cԭ�ӡ�4��Hԭ�ӣ���AΪHOCH2CH2OH��BΪOHC-CHO����4����C������Ϣ�л�ԭ�õ�E��E�к���̼̼˫�����ǻ����������ˣ���E���巢���ӳɷ�Ӧ�õ�F��F��NaCN������ȡ����Ӧ��ˮ��õ�G��G�к���2��-COOH��1��-OH���������ˣ���G����������Ӧ�õ�H�������ں���Ԫ������G�и�-COOHҲ�������ˣ���C��̼̼˫���������ˣ���CΪCH2=CH-CH2-COOH��EΪCH2=CHCH2CH2OH��FΪBrCH2CHBrCH2CH2OH��GΪ
����֪M1��M2�����к��в����Ͷ�Ϊ1�Ļ��ţ���C��D�ķ���ʽ��֪��Ӧ�Ƿ����Ӿ۷�Ӧ����C����̼̼˫�����Ȼ�������֪AΪ��Ԫ����Դ������ˮ����-Br��ˮ�⣬Aת������B��-CH2OHת��Ϊ-CHO����Է���������С4��Ҳ֤��A����2���ǻ���A����Է�������Ϊ62��ȥ��2��-OH��ʣ�������ԭ������Ϊ62-34=28��ֻ��Ϊ2��Cԭ�ӡ�4��Hԭ�ӣ���AΪHOCH2CH2OH��BΪOHC-CHO����4����C������Ϣ�л�ԭ�õ�E��E�к���̼̼˫�����ǻ����������ˣ���E���巢���ӳɷ�Ӧ�õ�F��F��NaCN������ȡ����Ӧ��ˮ��õ�G��G�к���2��-COOH��1��-OH���������ˣ���G����������Ӧ�õ�H�������ں���Ԫ������G�и�-COOHҲ�������ˣ���C��̼̼˫���������ˣ���CΪCH2=CH-CH2-COOH��EΪCH2=CHCH2CH2OH��FΪBrCH2CHBrCH2CH2OH��GΪ ��HΪ
��HΪ �����л���X�Ľṹ��ʽΪ��
�����л���X�Ľṹ��ʽΪ�� ���ݴ˽��
���ݴ˽��
| 2��13+2-13-1 |
| 2 |
 ����֪M1��M2�����к��в����Ͷ�Ϊ1�Ļ��ţ���C��D�ķ���ʽ��֪��Ӧ�Ƿ����Ӿ۷�Ӧ����C����̼̼˫�����Ȼ�������֪AΪ��Ԫ����Դ������ˮ����-Br��ˮ�⣬Aת������B��-CH2OHת��Ϊ-CHO����Է���������С4��Ҳ֤��A����2���ǻ���A����Է�������Ϊ62��ȥ��2��-OH��ʣ�������ԭ������Ϊ62-34=28��ֻ��Ϊ2��Cԭ�ӡ�4��Hԭ�ӣ���AΪHOCH2CH2OH��BΪOHC-CHO����4����C������Ϣ�л�ԭ�õ�E��E�к���̼̼˫�����ǻ����������ˣ���E���巢���ӳɷ�Ӧ�õ�F��F��NaCN������ȡ����Ӧ��ˮ��õ�G��G�к���2��-COOH��1��-OH���������ˣ���G����������Ӧ�õ�H�������ں���Ԫ������G�и�-COOHҲ�������ˣ���C��̼̼˫���������ˣ���CΪCH2=CH-CH2-COOH��EΪCH2=CHCH2CH2OH��FΪBrCH2CHBrCH2CH2OH��GΪ
����֪M1��M2�����к��в����Ͷ�Ϊ1�Ļ��ţ���C��D�ķ���ʽ��֪��Ӧ�Ƿ����Ӿ۷�Ӧ����C����̼̼˫�����Ȼ�������֪AΪ��Ԫ����Դ������ˮ����-Br��ˮ�⣬Aת������B��-CH2OHת��Ϊ-CHO����Է���������С4��Ҳ֤��A����2���ǻ���A����Է�������Ϊ62��ȥ��2��-OH��ʣ�������ԭ������Ϊ62-34=28��ֻ��Ϊ2��Cԭ�ӡ�4��Hԭ�ӣ���AΪHOCH2CH2OH��BΪOHC-CHO����4����C������Ϣ�л�ԭ�õ�E��E�к���̼̼˫�����ǻ����������ˣ���E���巢���ӳɷ�Ӧ�õ�F��F��NaCN������ȡ����Ӧ��ˮ��õ�G��G�к���2��-COOH��1��-OH���������ˣ���G����������Ӧ�õ�H�������ں���Ԫ������G�и�-COOHҲ�������ˣ���C��̼̼˫���������ˣ���CΪCH2=CH-CH2-COOH��EΪCH2=CHCH2CH2OH��FΪBrCH2CHBrCH2CH2OH��GΪ ��HΪ
��HΪ �����л���X�Ľṹ��ʽΪ��
�����л���X�Ľṹ��ʽΪ�� ���ݴ˽��
���ݴ˽�����
�⣺�л���X�ķ���ʽΪC13H13O4Br�������Ͷ�Ϊ
=7�������ṹ��ʽ ����֪M1��M2�����к��в����Ͷ�Ϊ1�Ļ��ţ���C��D�ķ���ʽ��֪��Ӧ�Ƿ����Ӿ۷�Ӧ����C����̼̼˫�����Ȼ�������֪AΪ��Ԫ����Դ������ˮ����-Br��ˮ�⣬Aת������B��-CH2OHת��Ϊ-CHO����Է���������С4��Ҳ֤��A����2���ǻ���A����Է�������Ϊ62��ȥ��2��-OH��ʣ�������ԭ������Ϊ62-34=28��ֻ��Ϊ2��Cԭ�ӡ�4��Hԭ�ӣ���AΪHOCH2CH2OH��BΪOHC-CHO��
����֪M1��M2�����к��в����Ͷ�Ϊ1�Ļ��ţ���C��D�ķ���ʽ��֪��Ӧ�Ƿ����Ӿ۷�Ӧ����C����̼̼˫�����Ȼ�������֪AΪ��Ԫ����Դ������ˮ����-Br��ˮ�⣬Aת������B��-CH2OHת��Ϊ-CHO����Է���������С4��Ҳ֤��A����2���ǻ���A����Է�������Ϊ62��ȥ��2��-OH��ʣ�������ԭ������Ϊ62-34=28��ֻ��Ϊ2��Cԭ�ӡ�4��Hԭ�ӣ���AΪHOCH2CH2OH��BΪOHC-CHO��
��1��AΪHOCH2CH2OH������ʽΪC2H6O2��C��D�ķ�Ӧ����Ϊ�Ӿ۷�Ӧ���ʴ�Ϊ��C2H6O2���Ӿ۷�Ӧ��
��2��B������������Һ��Ӧ�Ļ�ѧ����ʽΪ��OHC-CHO+4Ag��NH3��2OH
H4NOOC-COONH4+2H2O+4Ag��+6NH3��
�ʴ�Ϊ��OHC-CHO+4Ag��NH3��2OH
H4NOOC-COONH4+2H2O+4Ag��+6NH3��
��3��C����̼̼˫�����Ȼ�����C�ķ���ʽ��֪��C���п��ܵĽṹ��ʽ�У�CH2=CH-CH2-COOH��CH3-CH=CH-COOH��CH2=C��CH3��-COOH��
�ʴ�Ϊ��CH2=CH-CH2-COOH��CH3-CH=CH-COOH��CH2=C��CH3��-COOH��
��4��C������Ϣ�л�ԭ�õ�E��E�к���̼̼˫�����ǻ����������ˣ���E���巢���ӳɷ�Ӧ�õ�F��F��NaCN������ȡ����Ӧ��ˮ��õ�G��G�к���2��-COOH��1��-OH���������ˣ���G����������Ӧ�õ�H�������ں���Ԫ������G�и�-COOHҲ�������ˣ���C��̼̼˫���������ˣ���CΪCH2=CH-CH2-COOH��EΪCH2=CHCH2CH2OH��FΪBrCH2CHBrCH2CH2OH��GΪ ��HΪ
��HΪ �����л���X�Ľṹ��ʽΪ��
�����л���X�Ľṹ��ʽΪ�� ��
��
��ȷ��C�Ľṹ��ʽΪCH2=CH-CH2-COOH����ӦG��H�Ļ�ѧ����ʽΪ ��
��
�ʴ�Ϊ��CH2=CH-CH2-COOH�� ��
��
��EΪCH2=CHCH2CH2OH��E�ж���ͬ���칹�壬ͬʱ��������Ҫ��������������E��ȫ��ͬ�����к���ȩ�������Ǻ����Ѽ�����״����1H�˴Ź�������ʾ��3����ڲ�ͬ�Ļ�ѧ����������ͬʱ����Ҫ���ͬ���칹��Ϊ�� �ȣ�
�ȣ�
�ʴ�Ϊ�� �ȣ�
�ȣ�
| 2��13+2-13-1 |
| 2 |
 ����֪M1��M2�����к��в����Ͷ�Ϊ1�Ļ��ţ���C��D�ķ���ʽ��֪��Ӧ�Ƿ����Ӿ۷�Ӧ����C����̼̼˫�����Ȼ�������֪AΪ��Ԫ����Դ������ˮ����-Br��ˮ�⣬Aת������B��-CH2OHת��Ϊ-CHO����Է���������С4��Ҳ֤��A����2���ǻ���A����Է�������Ϊ62��ȥ��2��-OH��ʣ�������ԭ������Ϊ62-34=28��ֻ��Ϊ2��Cԭ�ӡ�4��Hԭ�ӣ���AΪHOCH2CH2OH��BΪOHC-CHO��
����֪M1��M2�����к��в����Ͷ�Ϊ1�Ļ��ţ���C��D�ķ���ʽ��֪��Ӧ�Ƿ����Ӿ۷�Ӧ����C����̼̼˫�����Ȼ�������֪AΪ��Ԫ����Դ������ˮ����-Br��ˮ�⣬Aת������B��-CH2OHת��Ϊ-CHO����Է���������С4��Ҳ֤��A����2���ǻ���A����Է�������Ϊ62��ȥ��2��-OH��ʣ�������ԭ������Ϊ62-34=28��ֻ��Ϊ2��Cԭ�ӡ�4��Hԭ�ӣ���AΪHOCH2CH2OH��BΪOHC-CHO����1��AΪHOCH2CH2OH������ʽΪC2H6O2��C��D�ķ�Ӧ����Ϊ�Ӿ۷�Ӧ���ʴ�Ϊ��C2H6O2���Ӿ۷�Ӧ��
��2��B������������Һ��Ӧ�Ļ�ѧ����ʽΪ��OHC-CHO+4Ag��NH3��2OH
| �� |
�ʴ�Ϊ��OHC-CHO+4Ag��NH3��2OH
| �� |
��3��C����̼̼˫�����Ȼ�����C�ķ���ʽ��֪��C���п��ܵĽṹ��ʽ�У�CH2=CH-CH2-COOH��CH3-CH=CH-COOH��CH2=C��CH3��-COOH��
�ʴ�Ϊ��CH2=CH-CH2-COOH��CH3-CH=CH-COOH��CH2=C��CH3��-COOH��
��4��C������Ϣ�л�ԭ�õ�E��E�к���̼̼˫�����ǻ����������ˣ���E���巢���ӳɷ�Ӧ�õ�F��F��NaCN������ȡ����Ӧ��ˮ��õ�G��G�к���2��-COOH��1��-OH���������ˣ���G����������Ӧ�õ�H�������ں���Ԫ������G�и�-COOHҲ�������ˣ���C��̼̼˫���������ˣ���CΪCH2=CH-CH2-COOH��EΪCH2=CHCH2CH2OH��FΪBrCH2CHBrCH2CH2OH��GΪ
 ��HΪ
��HΪ �����л���X�Ľṹ��ʽΪ��
�����л���X�Ľṹ��ʽΪ�� ��
����ȷ��C�Ľṹ��ʽΪCH2=CH-CH2-COOH����ӦG��H�Ļ�ѧ����ʽΪ
 ��
���ʴ�Ϊ��CH2=CH-CH2-COOH��
 ��
����EΪCH2=CHCH2CH2OH��E�ж���ͬ���칹�壬ͬʱ��������Ҫ��������������E��ȫ��ͬ�����к���ȩ�������Ǻ����Ѽ�����״����1H�˴Ź�������ʾ��3����ڲ�ͬ�Ļ�ѧ����������ͬʱ����Ҫ���ͬ���칹��Ϊ��
 �ȣ�
�ȣ��ʴ�Ϊ��
 �ȣ�
�ȣ�
���������⿼���л�����ƶϺͺϳɣ��������X�ĽṹƬ�Ρ�ˮ�����ṹ����Է������������ƶϣ���Ҫ�������չ����ŵ�������ת������Ŀ�ۺϽ�ǿ���Ѷ��еȣ�

��ϰ��ϵ�д�
�����Ŀ
���Ƕ�ʮһ���͵ġ����ǽ���������ҵ�����Խ��ʯΪԭ�ϣ������ķ�Ӧ�ǣ�
��Ӧ��aTiO2+bCl2+cC
aTiCl4+cCO ��Ӧ��TiCl4+2Mg
Ti+2MgCl2���ڷ�Ӧ�������˵������ȷ���ǣ�������
��Ӧ��aTiO2+bCl2+cC
| 1173K |
| 1220K��1420K |
| A����Ӧ���е�TiO2�������� |
| B��TiCl4�ڷ�Ӧ�����ǻ�ԭ����ڷ�Ӧ������������ |
| C��a=b=c=2 |
| D��ÿ����9.6 g Ti����Ӧ���й�ת��0.8 mol���� |
����˵����ȷ���ǣ�������
| A��pH=2�Ŀ�����c��H+����pH=3������ˮ��c��H+����10�� |
| B�������ͬ��pH��ͬ�Ĵ����������ȫ�ܽ������þ�ۣ�������ʱ�� |
| C��pH=10��NaOH��Һ��pH=12��NaOH����Һ�������Ϻ���Һ��pH=11 |
| D����V1LpH=11��NaOH��Һ��V2LpH=3��HA��Һ��Ϻ������ԣ���V1��V2 |
����ʱ�����и���������ָ����Һ��һ���ܴ���������ǣ�������
| A��pH=0����Һ�У�Na+��Fe2+��NO3-��ClO- | ||
| B��c��Fe3+��=0.1mol/L����Һ�У�K+��Ba2+��SO42-��SCN- | ||
C��
| ||
| D����ˮ�����c��H+��=1��10-14 mol/L����Һ�У�Ca2+��K+��Cl-��HCO3- |