��Ŀ����
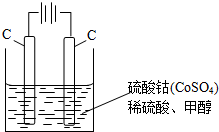
��ͼ1��ʵ����ģ��¹���ѧ�����ϣ��ȼ�շ��ⶨ�л�����ɵ�װ�ã�����ͭ����������750������ʹ�л�������������ȫ������ΪCO2��H2O���Իش��������⣮
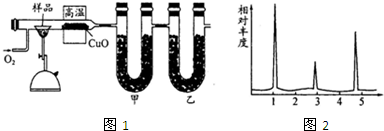
��1����װ����ʢ�ŵ������� ��
��2��ʵ�鿪ʼʱ��Ҫ��ͨ��������Ȼ���ټ��ȣ�Ŀ���� ��Ϊ��С������װ���л������ӵ�װ�ü��Լ��� ����ԭͼ����������ʵ������Ҫ��õ������ǣ�
��3����4.6gij�л���A����ʵ�飬��ü���������5.4g������������8.8g����������и�Ԫ�ص�ԭ�Ӹ������� ��ijѧ���ڴ˻������������·�����
��A���ܵĽṹΪ ��д�ṹ��ʽ����
��A�ĺ˴Ź���������ͼ2����A�Ĺ���������Ϊ ��

��1����װ����ʢ�ŵ�������
��2��ʵ�鿪ʼʱ��Ҫ��ͨ��������Ȼ���ټ��ȣ�Ŀ����
| ʵ��ǰ | ʵ��� |
| | |
��A���ܵĽṹΪ
��A�ĺ˴Ź���������ͼ2����A�Ĺ���������Ϊ
���㣺�ⶨ�л�����ӵ�Ԫ�����
ר�⣺ʵ�������
��������1������ˮ������������ˮ�Ȼ��ƣ������ʲ������ն�����̼���壻
��2��װ�����п�������Ӱ���л��ﱻ������Ӧ�����ž���Ҫ��֤��������ӵ������ֱ����л���ȼ�����ɵ�ˮ�Ͷ�����̼���������ܿ������е�ˮ�Լ�������̼�ĸ��ţ�
��3��Ũ��������Ϊˮ����������ʯ������Ϊ������̼���������Դ˿�ȷ���л�����C��Hԭ�Ӹ�����ֵ������Ԫ�أ��������غ�ȷ����ԭ�ӵ������ó�ԭ�Ӹ����ȣ������ʽ�������ݽṹ����������д��ѧʽ��
��2��װ�����п�������Ӱ���л��ﱻ������Ӧ�����ž���Ҫ��֤��������ӵ������ֱ����л���ȼ�����ɵ�ˮ�Ͷ�����̼���������ܿ������е�ˮ�Լ�������̼�ĸ��ţ�
��3��Ũ��������Ϊˮ����������ʯ������Ϊ������̼���������Դ˿�ȷ���л�����C��Hԭ�Ӹ�����ֵ������Ԫ�أ��������غ�ȷ����ԭ�ӵ������ó�ԭ�Ӹ����ȣ������ʽ�������ݽṹ����������д��ѧʽ��
���
�⣺��1���л���ȼ�����ɵ�ˮ�Լ�������̼�����գ�Ӧ����ʹ�ø��������ˮ�������ն�����̼������ˮ��������ˮ�Ȼ��ƣ��ʴ�Ϊ����ˮ�Ȼ��ƣ�
��2��ʵ�鿪ʼʱ��װ�����п�����Ϊ��ʹ�л���������Ҫ��ͨ���������ų�װ���еĿ�����Ȼ���ټ��ȣ�ʵ��װ���У�Ϊ�ܿ������е�ˮ�Լ�������̼�ĸ��ţ�����װ�ú�Ҫ��һ�����м�ʯ�ҵĸ���ܣ�����������������ӵ�������Ϊ���ɵ�ˮ�Ͷ�����̼���������ʴ�Ϊ���ų�װ���еĿ�����ʹ�л���������������װ�ú�����ʢ�м�ʯ�ҵ�U�ι� ����Ʒ���������ס��ҵ��������ס��ҵ�������
����Ʒ���������ס��ҵ��������ס��ҵ�������
��3���������֪n��H2O��=
=0.3mol��n��CO2��=
=0.2mol�����������غ㣬ȷ������������������5.4g+8.8g-4.6g=9.6g��0.3mol��������ԭ���غ��֪�л����к���n��O��=0.3mol+0.2mol��2-0.3mol��2=0.1mol��
���л�����n��C����n��H����n��O��=0.2mol��0.6mol��0.1mol=2��6��1���ʴ�Ϊ��n��C����n��H����n��O��=2��6��1��
�ٸ������и�Ԫ�ص�ԭ�Ӹ�����Ϊn��C����n��H����n��O��=2��6��1���л���ķ���ʽΪC2H6O���ṹ��ʽΪ��CH3CH2OH��CH3OCH3���ʴ�Ϊ��CH3CH2OH��CH3OCH3��
���л���A�ķ���ʽΪC2H6O��A�ĺ˴Ź���������ͼ2�����������壬��A�Ľṹ��ʽΪ��CH3CH2OH������������Ϊ���ǻ����ʴ�Ϊ�����ǻ���
��2��ʵ�鿪ʼʱ��װ�����п�����Ϊ��ʹ�л���������Ҫ��ͨ���������ų�װ���еĿ�����Ȼ���ټ��ȣ�ʵ��װ���У�Ϊ�ܿ������е�ˮ�Լ�������̼�ĸ��ţ�����װ�ú�Ҫ��һ�����м�ʯ�ҵĸ���ܣ�����������������ӵ�������Ϊ���ɵ�ˮ�Ͷ�����̼���������ʴ�Ϊ���ų�װ���еĿ�����ʹ�л���������������װ�ú�����ʢ�м�ʯ�ҵ�U�ι�
 ����Ʒ���������ס��ҵ��������ס��ҵ�������
����Ʒ���������ס��ҵ��������ס��ҵ���������3���������֪n��H2O��=
| 5.4g |
| 18g/mol |
| 8.8g |
| 44g/mol |
���л�����n��C����n��H����n��O��=0.2mol��0.6mol��0.1mol=2��6��1���ʴ�Ϊ��n��C����n��H����n��O��=2��6��1��
�ٸ������и�Ԫ�ص�ԭ�Ӹ�����Ϊn��C����n��H����n��O��=2��6��1���л���ķ���ʽΪC2H6O���ṹ��ʽΪ��CH3CH2OH��CH3OCH3���ʴ�Ϊ��CH3CH2OH��CH3OCH3��
���л���A�ķ���ʽΪC2H6O��A�ĺ˴Ź���������ͼ2�����������壬��A�Ľṹ��ʽΪ��CH3CH2OH������������Ϊ���ǻ����ʴ�Ϊ�����ǻ���
�����������ۺϿ����л���ķ��ӵ�ȷ������Ŀ�Ѷ��еȣ�ע��������غ�ĽǶȼ����л���ķ���ʽ��

��ϰ��ϵ�д�
�����Ŀ
�ڱ�״���£�����3.01��1023�����ӵ����������У���ռ���ԼΪ11.2L���ǣ�������
| A��N2 |
| B��C |
| C��H2O |
| D��S |
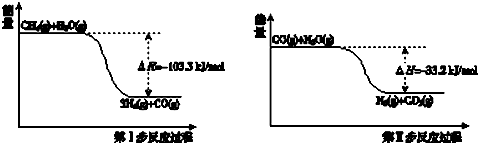
 ��Ҫ��������и�С�⣮
��Ҫ��������и�С�⣮