��Ŀ����
2��ij�¶�ʱ����һ���ݻ�Ϊ2L���ܱ������У�X��Y��Z�������ʵ����ʵ�����ʱ��ı仯������ͼ1��ʾ������ͼ�����ݣ�����д���пհף�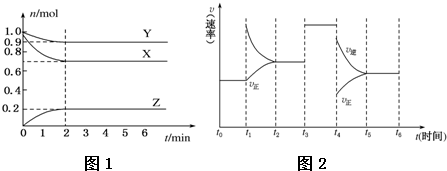
��1���÷�Ӧ�Ļ�ѧ����ʽΪ3X+Y?2Z��
��2����Ӧ��ʼ��2min������Z��ƽ����Ӧ����Ϊ0.05 mol•L-1•min-1��
��3����X��Y��Z��Ϊ���壬��Ӧ�ﵽƽ��ʱ��
��ѹǿ�ǿ�ʼʱ��0.9����
������ʱ�������������СΪԭ����$\frac{1}{2}$�����ﵽƽ��ʱ���������¶Ƚ����ߣ����������������Ƚ���������÷�Ӧ������ӦΪ���ȷ�Ӧ������ȡ������ȡ�����
��4����������Ӧ��2min���t1��t6�ڷ�Ӧ�����뷴Ӧʱ��ͼ����ͼ2����ÿһʱ�̾��ı�һ��Ӱ�췴Ӧ���ʵ����أ���B
A����t1ʱ������ѹǿ
B����t3ʱ�����˴���
C����t4ʱ�������¶�
D��t2��t3ʱX��ת������ߣ�
���� ��1���������ʵ����仯���ߣ���֪X��Y���ʵ�����С��Ϊ��Ӧ���Ӧ��û����ȫ��Ӧ�����Ը÷�ӦΪ���淴Ӧ��Z���ʵ������ӣ�Ϊ������ٸ��������ʵ����仯��֮�ȵ����������֮�ȣ���д����ѧ����ʽ��
��2������2min��Z���ʵ����仯���������������֪��Ũ�ȱ仯����ͨ�������֪������Z��ʾ�ķ�Ӧ���ʣ�
��3���������ѹǿ֮�ȵ�����������ʵ���֮�ȣ��������ʼʱ����������ʵ�����ƽ������������ʵ������õ��𰸣�
�������С��ԭ����0.5������ѹǿ��Ϊԭ����2����ƽ���������������С�ķ����ƶ������ݷ�Ӧ����ʽ��֪��ƽ�������ƶ���
��4������ͼ���жϸ�ʱ��ƽ���ƶ�����
��� �⣺��1����ͼ���֪X��YΪ��Ӧ�ZΪ�����X��Y��Z�Ļ�ѧ������֮�ȵ������ǵ����ʵ����仯֮��Ϊ0.3��0.1��0.2=3��1��2�����Է���ʽΪ3X+2Y?2Z���ʴ�Ϊ��3X+2Y?2Z��
��2��v��Z��=$\frac{0.2mol}{2L��2min}$=0.05 mol•L-1•min-1���ʴ�Ϊ��0.05 mol•L-1•min-1��
��3���ٸ���ͼ���п�֪����ʼʱ���������ʵ���Ϊ��2mol��ƽ������������ʵ���Ϊ��0.9mol+0.7mol+0.2mol=1.8mol��$\frac{{P}_{ƽ��}}{{P}_{��ʼ}}$=$\frac{{n}_{ƽ��}}{{n}_{��ʼ}}$=$\frac{1.8mol}{2mol}$�T0.9��
�ʴ�Ϊ��0.9��
�ڸ��ݷ���ʽ����С�����ƽ�������ƶ����������¶Ƚ����ߣ��ʸ÷�ӦΪ���ȷ�Ӧ���ʴ�Ϊ�����ȣ�
��4������ͼ��t1ʱ�������������Ũ�ȣ�ƽ�������ƶ���t3ʱ�����˴�����ƽ�ⲻ�ƶ���t4ʱӦΪ��Сѹǿ��ƽ�������ƶ�����Bѡ����ȷ���ʴ�Ϊ��B��
���� ����ͨ��ͼ�������ʵ����ʵ����ı仯�������黯ѧƽ����ƶ�����ѧ��Ӧ���ʼ��㣬�ۺϿ���ѧ����֪ʶ���ۺ�Ӧ��������

 �������¿��ÿ�ʱ��ҵϵ�д�
�������¿��ÿ�ʱ��ҵϵ�д� Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�
Ӣ�żƻ�ͬ����ʱ��Чѵ��ϵ�д�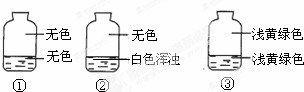 ��ʢ����������������ƿ�У��ֱ�ע��Լ���֮һ������Һ�岢���۲���ͼ����¼�������жϸ�ƿע���Һ��������ʲô����������
��ʢ����������������ƿ�У��ֱ�ע��Լ���֮һ������Һ�岢���۲���ͼ����¼�������жϸ�ƿע���Һ��������ʲô����������| A�� | AgNO3��Һ��NaOH��Һ��ˮ | B�� | NaOH��Һ��AgNO3��Һ��ˮ | ||
| C�� | ˮ��AgNO3��Һ��NaOH��Һ | D�� | AgNO3��Һ��ˮ��NaOH��Һ |
| A�� | �Ʊ�Al��OH��3����Һ����1mol•L-1AlCl3��Һ�мӹ�����6mol•L-1NaOH��Һ | |
| B�� | �ᴿ���������������������������������������������м����������̼������Һ�����÷�Һ������ȥ�л����ˮ | |
| C�� | ������Һ���Ƿ���Fe3+��ȡ������������Һ�������м�������������ˮ���ٵμ�KSCN��Һ���۲�ʵ������ | |
| D�� | ̽��������H2O2�ֽ����ʵ�Ӱ�죺����ͬ�����£���һ�Թ��м���2mL5%H2O2��1mLH2O������һ�Թ��м���2mL 10%H2O2��1mLFeCl3��Һ���۲첢�Ƚ�ʵ������ |
��1��ͼ1��ʾCO��g����H2��g������CH3OH��g���ķ�Ӧ�����������ı仯������aδʹ�ô���������bʹ�ô�������д���������¸÷�Ӧ���Ȼ�ѧ����ʽ��CO��g��+2H2��g��=CH3OH��g����H=-91 kJ/mol��
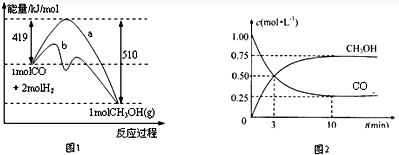
��2��Ϊ̽����ӦCO2��g��+3H2��g��?CH3OH��g��+H2O��g����ԭ���������Ϊ1L���ܱ�����A�У�����1molCO2��3molH2��һ�������·�����Ӧ�����CO2��CH3OH��g����Ũ����ʱ��仯��ͼ2��ʾ��
�ٴӷ�Ӧ��ʼ��ƽ�⣬H2��ƽ����Ӧ����v��H2��=0.225mol/��L��min����CO2��ת����Ϊ75%��
�ڸ��¶��£���Ӧ�ﵽƽ��ʱ�����й�ϵʽ��ȷ����a������ţ���
a��v����CO2��=v����H2O��g���� b��$\frac{{P}_{��ʼ}}{{P}_{ƽ��}}$=2 c��v����H2��=v����CO2��d��$\frac{{P}_{��ʼ}}{{P}_{ƽ��}}$=1��
�۸��¶��£���Ӧ��ƽ�ⳣ��=$\frac{16}{3}$��
���������1L���ܱ�����B�У�����2molCO2��6molH2������A��ͬ�������´ﵽƽ�⣬����������H2��ת���ʣ�A������B�������������������=������
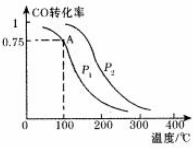
��3����ҵ����CO2��H2Ϊԭ�Ϻϳ��Ҵ���2CO2��g��+6H2��g��?CH3CH2OH��g��+3H2O��g������һ��ѹǿ�£���õ�ʵ���������±���
| �¶ȣ�K�� CO2ת���ʣ�%�� n��H2��/n��CO2�� | 500 | 600 | 700 | 800 |
| 1.5 | 45 | 33 | 20 | 12 |
| 2 | 60 | X | 28 | 15 |
| 3 | 83 | 62 | 37 | 22 |
�ڱ���X��ȡֵ��Χ��33��X��60��
| A�� | �������������������Ǣ�A�� | B�� | �߸�����18���� | ||
| C�� | ��A��ȫ���ǽ���Ԫ�� | D�� | ����Ԫ�������������Ǣ�B�� |
| A�� | 2NaCl�����ڣ�$\frac{\underline{\;���\;}}{\;}$2Na+Cl2�� | B�� | 6MgO+4Al$\frac{\underline{\;\;��\;\;}}{\;}$6Mg+2Al2O3 | ||
| C�� | Fe2O3+3CO$\frac{\underline{\;����\;}}{\;}$2Fe+3CO2 | D�� | 2Ag2O$\frac{\underline{\;\;��\;\;}}{\;}$4Ag+O2�� |
| A�� | 1mol H2O���е�ԭ����ΪNA | |
| B�� | 4gH2�����������4NA | |
| C�� | ���³�ѹ�£�11.2L O2�к��з�����Ϊ0.5NA | |
| D�� | 1L 0.1 mol•L-1NaCl��Һ�к�Na+��0.1NA |
| A�� | 150ml 1mol/L��NaCl��Һ | B�� | 150ml 3mol/LKCl��Һ | ||
| C�� | 75ml 2mol/L��NH4Cl��Һ | D�� | 50ml 1mol/lCuCl2��Һ |
 CO��SO2����Ҫ�Ĵ�����Ⱦ���壬���û�ѧ��Ӧԭ����������Ⱦ����Ҫ������
CO��SO2����Ҫ�Ĵ�����Ⱦ���壬���û�ѧ��Ӧԭ����������Ⱦ����Ҫ������