��Ŀ����
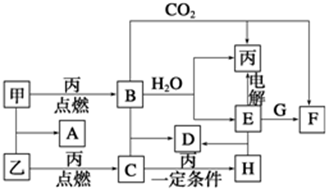
11���絼���Ǻ����������Һ����������С��������������Һ�絼�ʱ仯����ȷ���ζ���Ӧ���յ㣮��ͼ��ijͬѧ��0.1mol/L KOH��Һ�ֱ�ζ������Ϊ20mL��Ũ�Ⱦ�Ϊ0.1mol/L��HCl��CH3COOH��Һ�ζ�����ʾ��ͼ�������Һ����仯���Բ��ƣ��������й��жϲ���ȷ���� ��������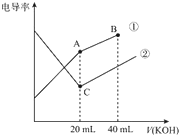
| A�� | ���ߢٴ���0.1 mol/L KOH��Һ�ζ�CH3COOH��Һ�ĵζ����ߣ����ߢڴ���0.1 mol/L KOH��Һ�ζ�HCl��Һ�ĵζ����� | |
| B�� | ����ͬ�¶��£�C��ˮ�����c��H+������A��ˮ�����c��H+�� | |
| C�� | ��A�����Һ���У�c��CH3COO-��+c��OH-��-c��H+��=0.05 mol/L | |
| D�� | ��B�����Һ���У�c��K+����c��OH-����c��CH3COO-����c��H+�� |
���� A����Һ�絼��������Ũ�ȳ����ȣ���ͬŨ�ȵĴ����HCl������絼��С��HCl��
B����������ˮ���룬ǿ��ǿ���β��ٽ�Ҳ������ˮ���룬���������ӵ��δٽ�ˮ���룻
C��A����Һ������ΪCH3COOK����Һ�д��ڵ���غ㣬���ݵ���غ��c��CH3COO-��+c��OH-��-c��H+��=c��K+��������Ϻ���Һ�������������Ũ�ȼ�С��
D��B����Һ������Ϊ�����ʵ���Ũ�ȵ�KOH��CH3COOK��CH3COO-ˮ������OH-��KOH��������OH-����������غ��жϣ�
��� �⣺A����Һ�絼��������Ũ�ȳ����ȣ���ͬŨ�ȵĴ����HCl������絼��С��HCl������ͼ֪��δ��KOH��Һʱ���絼�ʢڣ��٣������ߢٴ���0.1 mol/L KOH��Һ�ζ�CH3COOH��Һ�ĵζ����ߣ����ߢڴ���0.1 mol/L KOH��Һ�ζ�HCl��Һ�ĵζ����ߣ���A��ȷ��
B����������ˮ���룬ǿ��ǿ���β��ٽ�Ҳ������ˮ���룬���������ӵ��δٽ�ˮ���룬A����Һ������Ϊ����أ��ٽ�ˮ���룬C����Һ������ΪKCl��KCl���ٽ�Ҳ������ˮ���룬��C��ˮ�����c��H+��С��A��ˮ�����c��H+������B����
C��A����Һ������ΪCH3COOK����Һ�д��ڵ���غ㣬���ݵ���غ��c��CH3COO-��+c��OH-��-c��H+��=c��K+��������Ϻ���Һ�������һ������������Ũ�Ƚ�Ϊԭ����һ�룬����c��CH3COO-��+c��OH-��-c��H+��=c��K+��=0.05 mol/L����C��ȷ��
D��B����Һ������Ϊ�����ʵ���Ũ�ȵ�KOH��CH3COOK��CH3COO-ˮ������OH-��KOH��������OH-������c��OH-����c��CH3COO-������������غ��c��K+����c��OH-����c��CH3COO-����c��H+������D��ȷ��
��ѡB��
���� �������������Һ�����ж�Ϊ���忼������Ũ�ȴ�С�Ƚϣ�Ϊ��Ƶ���㣬��ȷ��Һ�����ʳɷּ��������ǽⱾ��ؼ���ע�����غ�������غ����ȷ���ã��״�ѡ����C���ܶ�ͬѧ����������Һ����仯���´�����Ŀ�ѶȲ���

 �߽�������ϵ�д�
�߽�������ϵ�д�
��������롿
����������ijɷֿ���ֻ��SO3һ�֣�
����������ijɷֿ��ܺ���SO2��O2���֣�
����������ijɷֿ��ܺ���SO3��SO2��O2���֣�
��ʵ��̽����
ʵ����������ԣ�
��֪ʵ�����ʱ������ͭ��ȫ�ֽ⣮
��1��������װ̽��ʵ���װ�ã����������ҵķ��������ӿڵ�����˳��Ϊ���١��������ޡ��ݡ��ۡ��ܡ�����ߡ��ڣ���ӿ���ţ���
��2����ʵ�����ʱB����Ͳû���ռ���ˮ����֤���������ȷ��
��3��������ʵ��С����и�ʵ�飬���ڼ���ʱ���¶Ȳ�ͬ��ʵ����������������Ҳ��ͬ���������£�
| ʵ��С�� | ��ȡCuSO4������/g | װ��C���ӵ�����/g | ��Ͳ��ˮ���������ɱ�״������������/mL |
| һ | 6.4 | 2.56 | 448 |
| �� | 6.4 | 2.56 | 224 |
��һС�飺2CuSO4$\frac{\underline{\;\;��\;\;}}{\;}$2CuO+2SO2��+O2����
�ڶ�С�飺4CuSO4$\frac{\underline{\;\;��\;\;}}{\;}$4CuO+2SO2��+2SO3��+O2����
| A�� | 58.5 g���Ȼ��ƹ����к���NA���Ȼ��Ʒ��� | |
| B�� | ��״���£�5.6 Lһ��������5.6 L������Ϻ�ķ�������Ϊ0.5NA | |
| C�� | 0.1 mol•L-1����������Һ�к���������Ϊ0.1NA | |
| D�� | 5.6 g��������ϡ���ᷴӦʱʧȥ������Ϊ0.2NA |
| A�� | 1.6gCH4�к��еķ����� | |
| B�� | 100 mL1mol/LH2SO4�к��е�H+ �� | |
| C�� | 2.4gMg�к��е�ԭ���� | |
| D�� | ��״���£�22.4LCO�����к��еķ����� |
| A�� | 2�� | B�� | 3�� | C�� | 4�� | D�� | 5�� |
| A�� | ����֬������ͬϵ�������л����Ǽ��� | |
| B�� | 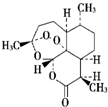 �������ǿ�ű����Чҩ���ṹ��ͼ��ʾ�������������������Ѽ���������ˮ���������ȷ¡���ͪ�ȣ��ܹ�����ˮ�ⷴӦ������ǿ������ | |
| C�� | ��֬��һ��������ˮ��ɸ�֬������ͣ���Ϊ������Ӧ | |
| D�� | ��X��Y�����л�����ۺ��ֱ�����ϣ�ֻҪ�����ʵ���֮�Ͳ��䣬��ȫȼ��ʱ��������������ˮ�����ʵ���Ҳ���䣬��XΪCH4����Y������CH3COOH |
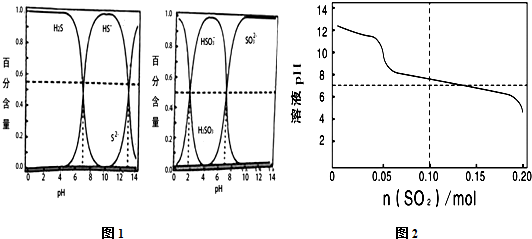
| A�� | ��ͼ1��һ����������������Kaֵ��֪H2SO3?2H++SO32- Ka��10-7 | |
| B�� | ��1L0.1 mol•L-1��Na2S��Һ�г���ͨ��SO2����ﵽ0.1 molʱ����Һ��H2S��HS-��HSO3-��SO32-��ͬʱһ�������� | |
| C�� | ��1L0.1 mol•L-1��Na2S��Һ�г���ͨ��SO2����ﵽ0.1 molʱ����Һ�д������¹�ϵ��c��H+��+c��Na+���Tc��OH-��+2c��S2-��+2c��SO32-��+c��HS- ��+c��HSO3-�� | |
| D�� | �ڷ�Ӧ�����е�pH����7.5���ң�֮����ܳ��ֵ���ɫ���� |