��Ŀ����
16�� ̫���ܵ�صķ�չ�Ѿ������˵�����������������ͭ������CIGS�Ȼ����ﱡĤ̫���ܵ���Լ���Ĥ��ϵ̫���ܵ�أ����������գ�
̫���ܵ�صķ�չ�Ѿ������˵�����������������ͭ������CIGS�Ȼ����ﱡĤ̫���ܵ���Լ���Ĥ��ϵ̫���ܵ�أ����������գ���1����ͭ���ӣ�Cu+����̬ʱ�ĵ����Ų�ʽΪ[Ar]3d10��
��2����Ϊ��������Ԫ�أ����ڵ�Ԫ��������壬����3��Ԫ�صĵ�һ������I1�Ӵ�С˳��Ϊ����Ԫ�ط��ű�ʾ��Br��As��Se����ԭ�ӽṹ�۵���Խ���As��Se��Brԭ�Ӱ뾶���μ�С��ԭ�Ӻ˶������ӵ�������������ǿ��Ԫ�صĵ�һ��������������Seԭ�����������Ų�Ϊ4s24p4����Asԭ�����������Ų�Ϊ4s24p3��p�����Ų����ڰ����״̬�����ݺ��ع���������֪�������״̬���ȶ�������AsԪ�صĵ�һ�����ܱ�Se��
��3������Ԫ�ش���ͬһ�������Ԫ�ؾ���ȱ�����ԣ��۵��������ڼ۲����������仯�������� �й¶Ե��ӵķ��ӻ��������ɼӺ����BF3����NH3��Ӧ����BF3•NH3��BF3•NH3��Bԭ�ӵ��ӻ��������Ϊsp3��Nԭ�ӵ��ӻ��������Ϊsp3��B�� N֮���γ���λ ����
��4��������Ľṹ����ʯ�ṹ���ƣ��������ʯ������һ���Cԭ�ӻ���Siԭ����ͬ��ԭ�Ӳ��ɼ��������ͼ��ʾ�Ľ��ɰ��SiC���ṹ�����ɰ��������ԭ�Ӿ��壨������ͣ���SiC�ṹ�У�ÿ��Cԭ����Χ�����Cԭ����ĿΪ12��
���� ��1�����ݹ���ԭ��д����̬ͭ���ӣ�Cu+���ĵ����Ų�ʽ��ע��ͭԭ�ӱ����ͭ����ʱ��ʧȥ�������ӣ�
��2��ͬһ�����У�Ԫ�صĵ�һ����������ԭ�����������������������ƣ�����IIA��͵�VA��Ԫ�صĵ�һ�����ܴ���������Ԫ�أ�ԭ�Ӱ뾶ԽС��ԭ�Ӻ˶������ӵ�������Խ����Ԫ�ع���еĵ��Ӵ��ڰ�����ȫ����ȫ��ʱ��ԭ�����ȶ���
��3�����ݼ۲���ӶԻ�������ȷ�����ӻ���ʽ���ṩ�չ����ԭ�Ӻ��ṩ�µ��ӶԵ�ԭ��֮���γ���λ����
��4�����ɰ����ԭ�Ӿ��壬ÿ��̼ԭ������4����ԭ�ӣ�ÿ����ԭ������������3��̼ԭ�ӣ��ݴ��ж�ÿ��Cԭ����Χ�����Cԭ����Ŀ��
��� �⣺��1��ͭ��29��Ԫ�أ�ͭԭ��ʧȥһ�����ӱ����ͭ���ӣ�������ͭ���Ӻ�����28�����ӣ���̬ͭ���ӣ�Cu+���ĵ����Ų�ʽΪ��[Ar]3d10��
�ʴ�Ϊ��[Ar]3d10��
��2��As��Se��Br����ͬһ������ԭ������������������Ԫ���������ڵ�VA�塢��VIA�塢��VIIA�壬��VA��Ԫ�ش���������Ԫ�صĵ�һ�����ܣ�����3��Ԫ�صĵ�һ�����ܴӴ�С˳��ΪBr��As��Se�� As��Se��Brԭ�Ӱ뾶���μ�С��ԭ�Ӻ˶������ӵ�������������ǿ��Ԫ�صĵ�һ��������������Seԭ�����������Ų�Ϊ4s24p4����Asԭ�����������Ų�Ϊ4s24p3��p�����Ų����ڰ����״̬�����ݺ��ع���������֪�������״̬���ȶ�������AsԪ�صĵ�һ�����ܱ�Se��
�ʴ�Ϊ��Br��As��Se�� As��Se��Brԭ�Ӱ뾶���μ�С��ԭ�Ӻ˶������ӵ�������������ǿ��Ԫ�صĵ�һ��������������Seԭ�����������Ų�Ϊ4s24p4����Asԭ�����������Ų�Ϊ4s24p3��p�����Ų����ڰ����״̬�����ݺ��ع���������֪�������״̬���ȶ�������AsԪ�صĵ�һ�����ܱ�Se��
��3��BF3•NH3��Bԭ�Ӻ���3���� ��һ����λ����������۲��������4��Bԭ�Ӳ�ȡsp3�ӻ���Nԭ�Ӻ���3���� ��һ����λ����Nԭ�Ӳ�ȡsp3�ӻ����û������У�Bԭ���ṩ�չ����ԭ�ӡ�Nԭ���ṩ�µ��Ӷԣ�����B��Nԭ��֮���γ���λ�����ʴ�Ϊ��sp3��sp3����λ����
��4�����ɰ���н��ʯ�Ľṹ�ص㣬����ԭ�Ӿ��壬ÿ��̼ԭ������4����ԭ�ӣ�ÿ����ԭ������������3��̼ԭ�ӣ�����ÿ��Cԭ����Χ�����Cԭ����ĿΪ3��4=12���ʴ�Ϊ��ԭ�Ӿ��壻12��
���� ���⿼���Ϊ�ۺϣ�Ϊ�߿��������ͺ�Ƶ���㣬���ؿ���ѧ���ķ�����������Ŀ�漰��������Ų�����һ�����ܴ�С�ıȽϡ��ӻ���ʽ���жϵ�֪ʶ�㣬�ѵ�����λ�����жϣ�����ݾ����ṹ��Ϸḻ���������з�������ѶȽϴ�

 �Ķ��쳵ϵ�д�
�Ķ��쳵ϵ�д���2B-+Z2=B2+2Z-��2A2++B2=2A3++2B-��2XO4-+10Z-+16H+=2X2++5Z2+8H2O
����������Ӧ���ж����н����д�����ǣ�������
| A�� | Ҫ��ȥ���� A2+��Z-��B-�����Һ�е�A2+����������Z-��B-��Ӧ����Z2 | |
| B�� | ��ԭ��ǿ��˳��Ϊ��A2+��B-��Z-��X2+ | |
| C�� | X2+�� XO4- �Ļ�ԭ���B2�� B-���������� | |
| D�� | ����Һ�п��ܷ�����Ӧ��XO4-+5 A2++8H+=X2++5A3++4H2O |
| A�� | O2-1s22s22p4 | B�� | Ca 3d2 | ||
| C�� | Fe��3d54s3 | D�� | Si��1s22s22p63s23p2 |
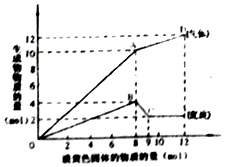 ij��Һ�п��ܺ���Fe2+��Mg2+��Cu2+��NH4+��Al3+�е�һ�ֻ��֣�������һ�ֵ���ɫ���岢������Һʱ���д̼�������ų��Ͱ�ɫ�������ɣ����뵭��ɫ��������ʵ����������꣩�������ij����Ͳ�����������ʵ����������꣩�Ĺ�ϵ����ͼ��ʾ������˵����ȷ���ǣ�������
ij��Һ�п��ܺ���Fe2+��Mg2+��Cu2+��NH4+��Al3+�е�һ�ֻ��֣�������һ�ֵ���ɫ���岢������Һʱ���д̼�������ų��Ͱ�ɫ�������ɣ����뵭��ɫ��������ʵ����������꣩�������ij����Ͳ�����������ʵ����������꣩�Ĺ�ϵ����ͼ��ʾ������˵����ȷ���ǣ�������| A�� | ��Һ��һ������Mg2+��NH4+��Al3+�����ܺ���Fe2+ | |
| B�� | ͼ��A�����������������Ϊ230�� | |
| C�� | ͼ��A��D֮�������д̼�����ζ������ | |
| D�� | ͼ��B��C֮�䷢���Ļ�ѧ��Ӧһ����������ԭ��Ӧ |
| A�� | 5�� | B�� | 6�� | C�� | 7�� | D�� | 8�� |
��1�������ƻ����Һǰ�Ƚ�����ˮ�������һ��ʱ�����ã���Ŀ����_����ˮ�е��ܽ������������_��
��2���ø÷�����õ�Na2S2O3•5H2O�����г�����һ���������ʣ�ij��ȤС�����������������ʵijɷֽ���̽���������Ǹ���Ӧ�����������Ľᾧˮ����
��������衿����1��������ֻ��Na2CO3���ʣ� ����2��������ֻ��Na2S���ʣ�
����3�������к�Na2S��Na2CO3�������ʣ�
���������ϡ���SO2+2H2S�T3S��+2H2O��
��Na2S2O3�����ԡ�������Һ�н��ȶ�������������Һ����Ѹ�ٷ�Ӧ��
Na2S2O3+H2SO4�TNa2SO4+S��+SO2��+H2O��
��H2S+CuSO4�TCuS������ɫ��+H2SO4
���ж���˼����ijͬѧȡ�����Ƶõľ�����������ϡH2SO4�У���������������ͨ��CuSO4��Һ�У�δ����ɫ�������ݴ���Ϊ����2������������Ϊ������Ƿ�����������������������������������˵�����ɣ�Na2S2O3��ϡH2SO4��Ӧ���ɵ�SO2��H2S������Ӧ��������H2S�ݳ���
����Ʒ�������ʵ�顿���ڼ���1������±��е�ʵ�鷽���������ۣ�������ѡ������ѡʵ���Լ���3mol•L-1 H2SO4��Һ��1mol•L-1 NaOH��Һ������KMnO4��Һ������NaHCO3��Һ��Ʒ����Һ������ʯ��ˮ��
| ʵ�鷽�� | ������ |