��Ŀ����
6���������һ�ֳ��ݰ�ȫ��������ͨ�����·����ϳɣ�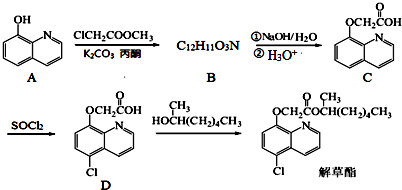
��1��������D�еĺ��������ŵ�����Ϊ�Ȼ����Ѽ���
��2��������B�Ľṹ��ʽΪ
 ����C��D�ķ�Ӧ������ȡ����Ӧ��
����C��D�ķ�Ӧ������ȡ����Ӧ����3��д��ͬʱ��������������C��һ��ͬ���칹��Ľṹ��ʽ
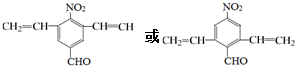 ��
�����ܷ���������Ӧ���Ӻ���1�������������Ϻ�������
�����к���4�ֲ�ͬ��ѧ��������
��4����֪��CH3CHO$��_{��}^{Cl_{2}}$ClCH2CHO
��д���Ա��Ӻͱ�ȩΪԭ���Ʊ��߷��ӻ�����X���ṹ��ʽ��ͼ
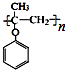 ���ĺϳ�·�����̣����Լ�����ѡ�����ϳ�·������ͼʾ�����£�
���ĺϳ�·�����̣����Լ�����ѡ�����ϳ�·������ͼʾ�����£�CH3CHO$��_{��������}^{O_{2}}$CH3COOH$��_{Ũ���ᡢ��}^{CH_{3}CH_{2}OH}$CH3COOCH2CH3��
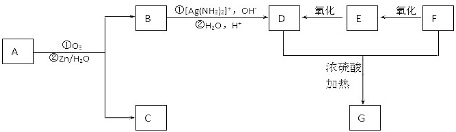
���� ��1������D�Ľṹ��ʽ����֪���еĺ���������Ϊ�Ȼ����Ѽ���
��2���Ա�A��C�ṹ��B�ķ���ʽ����֪A���ǻ���Hԭ�ӱ�-CH2COOCH3ȡ������B���Ա�C��D�ṹ��֪C�б�����Hԭ�ӱ�Clȡ������D��
��3��C��ͬ���칹�������㣺���ܷ���������Ӧ��˵������ȩ�������Ӻ���1�������������Ϻ��������������к���4�ֲ�ͬ��ѧ�������⣬������2��-CH=CH2��-CHO���������ڶ�λ����2��-CH=CH2����-CHO�����������߶Գƣ�
��4���ϳ� ��Ҫ����
��Ҫ���� ��������
��������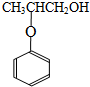 ������ȥ��Ӧ�õ�����
������ȥ��Ӧ�õ����� ������
������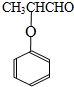 �����������ӳɷ�Ӧ�õ������A��B��ת����֪��
�����������ӳɷ�Ӧ�õ������A��B��ת����֪��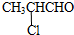 �뱽�ӷ���ȡ����Ӧ�õ�
�뱽�ӷ���ȡ����Ӧ�õ� ��CH3CH2CHO���������ȷ�Ӧ�õ�
��CH3CH2CHO���������ȷ�Ӧ�õ�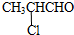 ��
��
��� �⣺��1������D�Ľṹ��ʽ����֪���еĺ���������Ϊ�Ȼ����Ѽ���
�ʴ�Ϊ���Ȼ����Ѽ���
��2���Ա�A��C�ṹ��B�ķ���ʽ����֪A���ǻ���Hԭ�ӱ�-CH2COOCH3ȡ������B����B�Ľṹ��ʽΪ�� ���Ա�C��D�ṹ��֪C�б�����Hԭ�ӱ�Clȡ������D������ȡ����Ӧ��
���Ա�C��D�ṹ��֪C�б�����Hԭ�ӱ�Clȡ������D������ȡ����Ӧ��
�ʴ�Ϊ�� ��ȡ����Ӧ��
��ȡ����Ӧ��
��3��C��ͬ���칹�������㣺���ܷ���������Ӧ��˵������ȩ�������Ӻ���1�������������Ϻ��������������к���4�ֲ�ͬ��ѧ�������⣬������2��-CH=CH2��-CHO���������ڶ�λ����2��-CH=CH2����-CHO�����������߶Գƣ�����������ͬ���칹��Ϊ�� ��
��
�ʴ�Ϊ�� ��
��
��4���ϳ� ��Ҫ����
��Ҫ���� ��������
�������� ������ȥ��Ӧ�õ�����
������ȥ��Ӧ�õ����� ������
������ �����������ӳɷ�Ӧ�õ������A��B��ת����֪��
�����������ӳɷ�Ӧ�õ������A��B��ת����֪��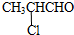 �뱽�ӷ���ȡ����Ӧ�õ�
�뱽�ӷ���ȡ����Ӧ�õ� ��CH3CH2CHO���������ȷ�Ӧ�õ�
��CH3CH2CHO���������ȷ�Ӧ�õ�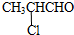 ���ϳ�·������ͼΪ��
���ϳ�·������ͼΪ�� ��
��
�ʴ�Ϊ�� ��
��
���� ���⿼���л���ĺϳɡ������Žṹ���л���Ӧ���͡���������ͬ���칹����д�ȣ�ע�����ת������������Ϣ��ƺϳ�·�ߣ��Ƕ��л���ѧ�������ۺϿ��飬�Ϻõؿ���ѧ������������֪ʶǨ������������

| ʵ�� | A���Լ� | B���Լ� | C��Һ�� |
| I | ����ʳ��ˮ | ��ʯ | H2O |
| II | Ũ��ˮ | NaOH���� | CCl4 |
| III | ���� | H2O | |
| IV | NaOH��Һ | þ���Ͻ� | H2O |

��1������E�������ǣ�������ƿ�ͷ�Һ©�����ѹǿ�㶨��ʹҺ����˳�����¡�������Һ��ĵ�������������
��2��ʵ��I��ƿB�з�����Ӧ�Ļ�ѧ����ʽ��CaC2+2H2O��Ca��OH��2+C2H2�������������徭�鴿���ȼ�������ǻ�������������Ũ�ҵĺ��̣�
��3��ʵ���ʣ���NH3�����մ��������¸���β������װ������ͼ2�����ܷ�ֹ��������be��
��4��ʵ����ʵ��Ŀ������֤�����ڳ�ʪ�����лᷢ��������ʴ����A���Լ���ѡ��ac������ţ���
a��NaOH��Һ b��C2H5OH c��NaCl��Һ d��ϡ����
��֤�������ڳ�ʪ�����лᷢ��������ʴ��������C�ܵ�Һ��������D��Һ���½���
��5��д��ʵ��IV�з�����Ӧ�����ӷ���ʽ2Al+2H2O+2OH-=2AlO2-+3H2����������ʱC��Һ�����D��Һ�棬������þ���Ͻ���þ������������ƫ�ߣ��ƫ�ߡ�����ƫ�͡�����Ӱ�족����
| A�� | ���ʵķ����ᴿ����֮һΪ��ɸ�֡����磺����--�������ᴿ����Һ--���˷��룬�����Ͼ����������������ӵ�ֱ����Сѡ����к��ʿ��ġ�ɸ�ӡ� | |
| B�� | ����ƿ����Ͳ�͵ζ����϶�����ʹ���¶ȣ�����ƿ�ޡ�0���̶ȣ���Ͳ�͵ζ����С�0���̶ȣ�ʹ��ʱ�ζ���ˮϴ������ϴ��������ƿˮϴ������ϴ | |
| C�� | ��ҵ�ϵ�ⱥ��ʳ��ˮʱ��Ϊ����ֹ�����֮�䷴Ӧ�������������ӽ���Ĥ���������������� | |
| D�� | �����£�Ũ�Ⱦ�Ϊ0.1 mol•L-1Na2S2O3��H2SO4��Һ����ȡ5 mL��10 mL��Na2S2O3��Һ�ֱ���10 mL��H2SO4��Һ��Ϸ�Ӧ������֤Na2S2O3Ũ�ȶԷ�Ӧ���ʵ�Ӱ�� |
| A�� | �з����ת�����ϣ��������̫���� | |
| B�� | ��װ���Ͼ�����ϩ������������ɻ�����Ⱦ | |
| C�� | ��ʯ�Ҷ�úȼ���γɵ�������������ʯ�� | |
| D�� | �з���Ч�����������ڼ������ٶԻ��ʵ�ʹ�� |
| A�� | ��ˮ�ܽ��Ȼ���������AgCl+2NH3•H2O=[Ag��NH3��2]++Cl-+2H2O | |
| B�� | CH3COOH��Һ��ȥˮ���е�CaCO3��CaCO3+2H+=Ca2++H2O+CO2�� | |
| C�� | ����K2Cr2O7��Һ����˫��ˮ��Cr2O72-+8H++5H2O2=2 Cr3++4O2��+9H2O | |
| D�� | ��NaHSO4��Ba��OH��2��Һ��������ԣ�H++SO42-+Ba2++OH-=BaSO4��+H2O |
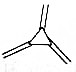 ������ϩ���������л��뵼����ʲ��ϵIJ��Ӽ�����ͬ���칹���к�������̼̼�����Ľṹ�� ��������
������ϩ���������л��뵼����ʲ��ϵIJ��Ӽ�����ͬ���칹���к�������̼̼�����Ľṹ�� ��������| A�� | 4�� | B�� | 5�� | C�� | 6�� | D�� | 7�� |
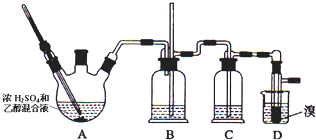
���ܴ��ڵ���Ҫ����Ӧ�У��Ҵ���Ũ����Ĵ�������l40����ˮ�������ѣ��й������б����£�
| �Ҵ� | 1��2-�������� | ���� | |
| ״̬ | ��ɫҺ�� | ��ɫҺ�� | ��ɫҺ�� |
| �ܶ�/g•cm-3 | 0.79 | 2.2 | 0.71 |
| �е�/�� | 78.5 | 132 | 34.6 |
| �۵�/�� | -130 | 9 | -1l6 |
��1��д��������ƿA�з�����Ӧ�Ļ�ѧ����ʽ
 ��
����2��Ϊ�˼���Aװ���и���Ӧ������ʱӦע��Ѹ�ٵذѷ�Ӧ�¶���ߵ�170�����ң�
��3����װ��C��Ӧ����NaOH��Һ����Ŀ�������շ�Ӧ�п������ɵ��������壻
��4���ж�Dװ���з�Ӧ�Ѿ�������������������ɫ��ȫ��ȥ��
��5����1��2-��������ֲ�Ʒ���ڷ�Һ©���м�ˮ�����ã�����Ӧ���²㣨��ϡ������¡�����
��6�������������������������ѣ���������ķ�����ȥ��
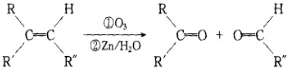
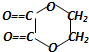 +2H2O����Ӧ������������Ӧ��D��̼�����Ʒ�Ӧ�Ļ�ѧ����ʽ��HOOCCOOH+2NaHCO3=NaOOCCOONa+2CO2��+2H2O��
+2H2O����Ӧ������������Ӧ��D��̼�����Ʒ�Ӧ�Ļ�ѧ����ʽ��HOOCCOOH+2NaHCO3=NaOOCCOONa+2CO2��+2H2O��