��Ŀ����
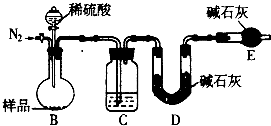
��״���£���a L SO2��Cl2��ɵĻ������ͨ��100mL 0.2mol?L-1Fe2��SO4��3��Һ�У���ַ�Ӧ����Һ���ػ�ɫ��dz����Ӧ�����Һ�м���������BaCl2��Һ�������ó������ˡ�ϴ�ӡ��������أ�������Ϊ23.3g�������й��ڸù��̵��ƶϲ���ȷ���ǣ�������
| A������������������ʵ���Ϊ0.04mol |
| B�����������SO2���ʵ���Ϊ0.04mol |
| C�����õij���Ϊ0.1mol BaSO4 |
| D��a��ȡֵ��ΧΪ0.896��a��1.792 |
���㣺��ѧ����ʽ���йؼ���
ר�⣺������
������SO2��Cl2��ɵĻ������ͨ��Fe2��SO4��3��Һ�У�����������������ǿ�������ӣ������������������Ӧ��SO2+Cl2+2H2O=2HCl+H2SO4����ַ�Ӧ����Һ���ػ�ɫ��dz��˵����������������������2Fe3++SO2+H2O=SO42-+2Fe2++2H+������������ȫ��Ӧ����Ӧ�����Һ������BaCl2��Һ�����ɵ�23.3g����ΪBaSO4��������Ԫ���غ��֪n��BaSO4��=3n[Fe2��SO4��3]+n��SO2�����ݴ˼���n��SO2���������������������������ݷ���ʽ��֪n��Cl2����n��SO2�����ݴ˽��
���
�⣺SO2��Cl2��ɵĻ������ͨ��Fe2��SO4��3��Һ�У�����������������ǿ�������ӣ������������������Ӧ��SO2+Cl2+2H2O=2HCl+H2SO4����ַ�Ӧ����Һ���ػ�ɫ��dz��˵����������������������2Fe3++SO2+H2O=SO42-+2Fe2++2H+������������ȫ��Ӧ����Ӧ�����Һ������BaCl2��Һ�����ɵ�23.3g����ΪBaSO4�������ʵ���Ϊ
=0.1mol��������Ԫ���غ��֪n��BaSO4��=3n[Fe2��SO4��3]+n��SO2����n��SO2��=0.1mol-0.1L��0.2mol/L��3=0.04mol��
A�������������������������ݷ���ʽ��֪n��Cl2����n��SO2��=0.04mol����A����
B��������������֪�����������SO2���ʵ���Ϊ0.04mol����B��ȷ��
C�������������֪�����õij���Ϊ0.1mol BaSO4����C��ȷ��
D��������������֪��V��SO2��=0.04mol��22.4L/mol=0.896L�������������������������ݷ���ʽ��֪n��Cl2����n��SO2������n��Cl2����0.04mol����V��Cl2����0.04mol��22.4L/mol=0.896L����a��ȡֵ��ΧΪ0.896��a��1.792����D��ȷ��
��ѡA��
| 23.3g |
| 233g/mol |
A�������������������������ݷ���ʽ��֪n��Cl2����n��SO2��=0.04mol����A����
B��������������֪�����������SO2���ʵ���Ϊ0.04mol����B��ȷ��
C�������������֪�����õij���Ϊ0.1mol BaSO4����C��ȷ��
D��������������֪��V��SO2��=0.04mol��22.4L/mol=0.896L�������������������������ݷ���ʽ��֪n��Cl2����n��SO2������n��Cl2����0.04mol����V��Cl2����0.04mol��22.4L/mol=0.896L����a��ȡֵ��ΧΪ0.896��a��1.792����D��ȷ��
��ѡA��
���������⿼��������йؼ��㣬��Ŀ�Ѷ��еȣ����������жϷ����ķ�Ӧ�ǹؼ������ضԻ�ѧ������⼼����˼ά�������飬ע�������غ�˼����еļ��㣮

��ϰ��ϵ�д�
 �����������һ��һ��ϵ�д�
�����������һ��һ��ϵ�д�
�����Ŀ
���������ε�ϡ��Һ���ֱ���a mol?L-1 NaX��Һ��b mol?L-1 NaY��Һ������˵������ȷ���ǣ�������
| A����a=b��pH��NaX����pH��NaY��������ͬŨ��ʱ������HX��HY |
| B����a=b�������c��X-��=c��Y-��+c��HY��������ͬŨ��ʱ������HX��HY |
| C����a��b�����c��X-��=c��Y-��������Ƴ���Һ��c��HX����c��HY��������ͬŨ��ʱ������HX��HY |
| D��������Һ�������ϣ����c��X-��+c��Y-��+c��HX��+c��HY��=0.1mol?L-1������Ƴ�a+b=0.2mol?L-1 |
�����й�ʵ��װ�ý��е���Ӧʵ�飬�ܴﵽʵ��Ŀ���ǣ�������

| A����ͼlװ�����ʵ������ȡ�������� |
| B����ͼ2��ʾװ�ÿ���ȡ������CO2���� |
| C����ͼ3��ʾװ�ÿɷ���������Ȼ�̼��Һ��ˮ |
| D����ͼ4װ���Ʊ�Fe��OH��2���ܽϳ�ʱ��۲�����ɫ |
HAΪһԪ���ᣬ��0.1mol?L-1 NaA��Һ�У�����˵����ȷ���ǣ�������
| A�������£�����Һ��pH=7 |
| B���ʵ����ȸ���Һ����Һ��PHֵ���� |
| C��c��Na+����c��A-����c��H+����c��OH-�� |
| D��c��Na+��+c��H+��=c��A-��+c��OH-�� |
�������������칹�������ʽΪC5H10O2��Ϊ����ͬ���칹�干��������
| A��7�� | B��8�� | C��9�� | D��10�� |

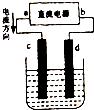 ͼ�ǵ��CuCl2��Һ��װ�ã�����c��dΪʯī�缫��
ͼ�ǵ��CuCl2��Һ��װ�ã�����c��dΪʯī�缫��