��Ŀ����
14��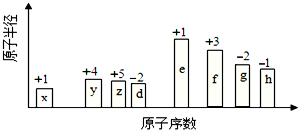 ��ԭ�������ĵ����İ��ֶ�����Ԫ�أ�����ĸX��ʾ��ԭ�Ӱ뾶����Դ�С��������ۻ�����۵ı仯��ͼ��ʾ��
��ԭ�������ĵ����İ��ֶ�����Ԫ�أ�����ĸX��ʾ��ԭ�Ӱ뾶����Դ�С��������ۻ�����۵ı仯��ͼ��ʾ�������жϳ���Ԫ�ػش����⣺
��1��f ��Ԫ�����ڱ���λ���ǵ������ڢ�A�壻g�����ӽṹʾ��ͼΪ
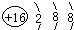 ��
����2����z��d��e��f ����Ԫ���У������Ӱ뾶���ɴ�С������˳��Ϊr��N3-����r��O2-����r��Na+����r��Al3+������
��ѧʽ��ʾ����ͬ�����Ƚ�g��h������������Ӧ��ˮ��������Խ�ǿ���ǣ�HClO4��
��3����x��zԪ�����һ����ԭ�ӹ��ۻ����д�������ʽ
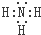 ��
����4����x��z��h����Ԫ�ع��ɵ��Σ���ˮ��Һ�����ԣ������ӷ���ʽ������ԭ��NH4++H2O?NH3��H2O+H+��
��5����֪1mol e �ĵ���������d2��ȼ�գ��ָ������£��ų�255.5kJ������д���÷�Ӧ���Ȼ�ѧ����ʽ��2Na��s��+O2��g��=Na2O2��s����H=-511kJ•mol-1��
���� ��ͼ�еĻ��ϼۡ�ԭ�Ӱ뾶�Ĵ�С��ԭ����������֪x��HԪ�أ�y��CԪ�أ�z��NԪ�أ�d��OԪ�أ�e��NaԪ�أ�f��AlԪ�أ�g��SԪ�أ�h��ClԪ�أ�
��1��f��AlԪ�أ���Ԫ�����ڱ���λ���ǵ������ڢ�A�壻S2-���Ӻ˵����Ϊ16�����������Ϊ18����3�����Ӳ㣬���������Ϊ2��8��8��
��2�����Ӳ�ṹ��ͬ�����ӣ��˵����Խ�����Ӱ뾶ԽС�����Ӳ�Խ�����Ӱ뾶Խ�ǽ�����Խǿ�����������ˮ���������Խǿ��
��3����H��NԪ�����һ����ԭ�ӹ��ۻ�����ΪNH3��
��4����H��N��Cl����Ԫ�ع��ɵ��Σ���ˮ��Һ�����ԣ�����ΪNH4Cl��笠�����ˮ����Һ�����ԣ�
��5������������ȼ�����ɹ������ƣ���Ӧ����ʽΪ��2Na+O2$\frac{\underline{\;��ȼ\;}}{\;}$Na2O2������2mol�Ʒ�Ӧ�ų��ų���������ע�����ʵľۼ�״̬�뷴Ӧ����д�Ȼ�ѧ����ʽ��
��� �⣺��ͼ�еĻ��ϼۡ�ԭ�Ӱ뾶�Ĵ�С��ԭ����������֪x��HԪ�أ�y��CԪ�أ�z��NԪ�أ�d��OԪ�أ�e��NaԪ�أ�f��AlԪ�أ�g��SԪ�أ�h��ClԪ�أ�
��1��f��AlԪ�أ���Ԫ�����ڱ���λ���ǵ������ڢ�A�壻S2-���ӽṹʾ��ͼΪ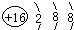 ��
��
�ʴ�Ϊ���������ڢ�A�壻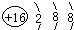 ��
��
��2�����Ӳ�ṹ��ͬ�����ӣ��˵����Խ�����Ӱ뾶ԽС�������Ӱ뾶��r��N3-����r��O2-����r��Na+����r��Al3+����
�ǽ�����S��Cl���ǽ�����Խǿ�����������ˮ���������Խǿ�������ԣ�HClO4��H2SO4��
�ʴ�Ϊ��r��N3-����r��O2-����r��Na+����r��Al3+����HClO4��
��3����H��NԪ�����һ����ԭ�ӹ��ۻ�����ΪNH3������ʽΪ��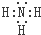 ��
��
�ʴ�Ϊ��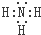 ��
��
��4����H��N��Cl����Ԫ�ع��ɵ��Σ���ˮ��Һ�����ԣ�����ΪNH4Cl��笠�����ˮ�⣺NH4++H2O?NH3��H2O+H+����Һ�����ԣ�
�ʴ�Ϊ��NH4++H2O?NH3��H2O+H+��
��5������������ȼ�����ɹ������ƣ���Ӧ����ʽΪ��2Na+O2$\frac{\underline{\;��ȼ\;}}{\;}$Na2O2��2mol�Ʒ�Ӧ�ų��ų�������Ϊ255.5kJ��2=511kJ����Ӧ�Ȼ�ѧ����ʽΪ��2Na��s��+O2��g��=Na2O2��s����H=-511kJ•mol-1��
�ʴ�Ϊ��2Na��s��+O2��g��=Na2O2��s����H=-511kJ•mol-1��
���� ���⿼��ṹ����λ�ù�ϵӦ�ã����ݻ��ϼ���ԭ�Ӱ뾶�ƶ�Ԫ���ǽ���ؼ���ע���Ԫ�������ɵ��������գ�

 �Ƹ������������ϵ�д�
�Ƹ������������ϵ�д�| A�� | ����������Һ | B�� | ϡ���� | C�� | ϡ���� | D�� | ��ˮ |
| A�� | �跨��̫����۽����������£�ʹˮ�ֽ�������� | |
| B�� | Ѱ�����⻯ѧ���ʣ�ʹˮ�ֽ����������ͬʱ�ͷ����� | |
| C�� | Ѱ�����⻯ѧ���ʣ����ڿ���������Դ�Էֽ�ˮ��ȡ���� | |
| D�� | ����ˮ���������������ǿ�ȼ�յ����ʣ���˿��о���ˮ���ֽ������£�ʹ���ܳ�Ϊ������Դ |
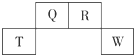 ������Ԫ��Q��R��T��W��Ԫ�����ڱ��е�λ������ͼ��ʾ������W��ԭ�Ӻ������������������������֮��Ϊ8��3������˵����ȷ���ǣ�������
������Ԫ��Q��R��T��W��Ԫ�����ڱ��е�λ������ͼ��ʾ������W��ԭ�Ӻ������������������������֮��Ϊ8��3������˵����ȷ���ǣ�������| A�� | Q������������Ӧˮ��������Դ���R������������Ӧˮ��������� | |
| B�� | T�����Ӱ뾶����W�����Ӱ뾶 | |
| C�� | Ԫ��T�γɵ��ʵĻ�ԭ�Դ���Ԫ��W�γɵ��ʵĻ�ԭ�� | |
| D�� | ��R������������Ӧˮ�����Ũ��Һ�е���T���ܽ⣬˵��δ������ѧ��Ӧ |
| A�� | 0.43 | B�� | 0.47 | C�� | 0.49 | D�� | 0.52 |
���̷� ������������ �۵⻯�� �ܱ��� �ݹ������ƣ�
| A�� | �٢ڢۢܢ� | B�� | �ۢ� | C�� | �٢ڢ� | D�� | �٢� |
| ��A | ��A | ��A | ��A | ��A | ��A | V��A | 0 | |
| 2 | �� | �� | ⑪ | |||||
| 3 | �� | �� | �� | �� | �� |
��2���٢ۢ�����Ԫ�ص����������ˮ�����У�������ǿ��NaOH���٢ۢ�����Ԫ�ص�ԭ�Ӱ뾶�ɴ�С��˳��ΪNa��Mg��Al��
��3����Ԫ�ص�ij������ͨ����������������д�������Ԫ�ص������ﷴӦ�Ļ�ѧ����ʽ2Na2O2+2H2O=4NaOH+O2����
| A�� | 2NA | B�� | NA | C�� | 1.5NA | D�� | 0.5 NA |
| A�� | AgNO3 | B�� | BaCl2 | C�� | NaOH | D�� | HCl |